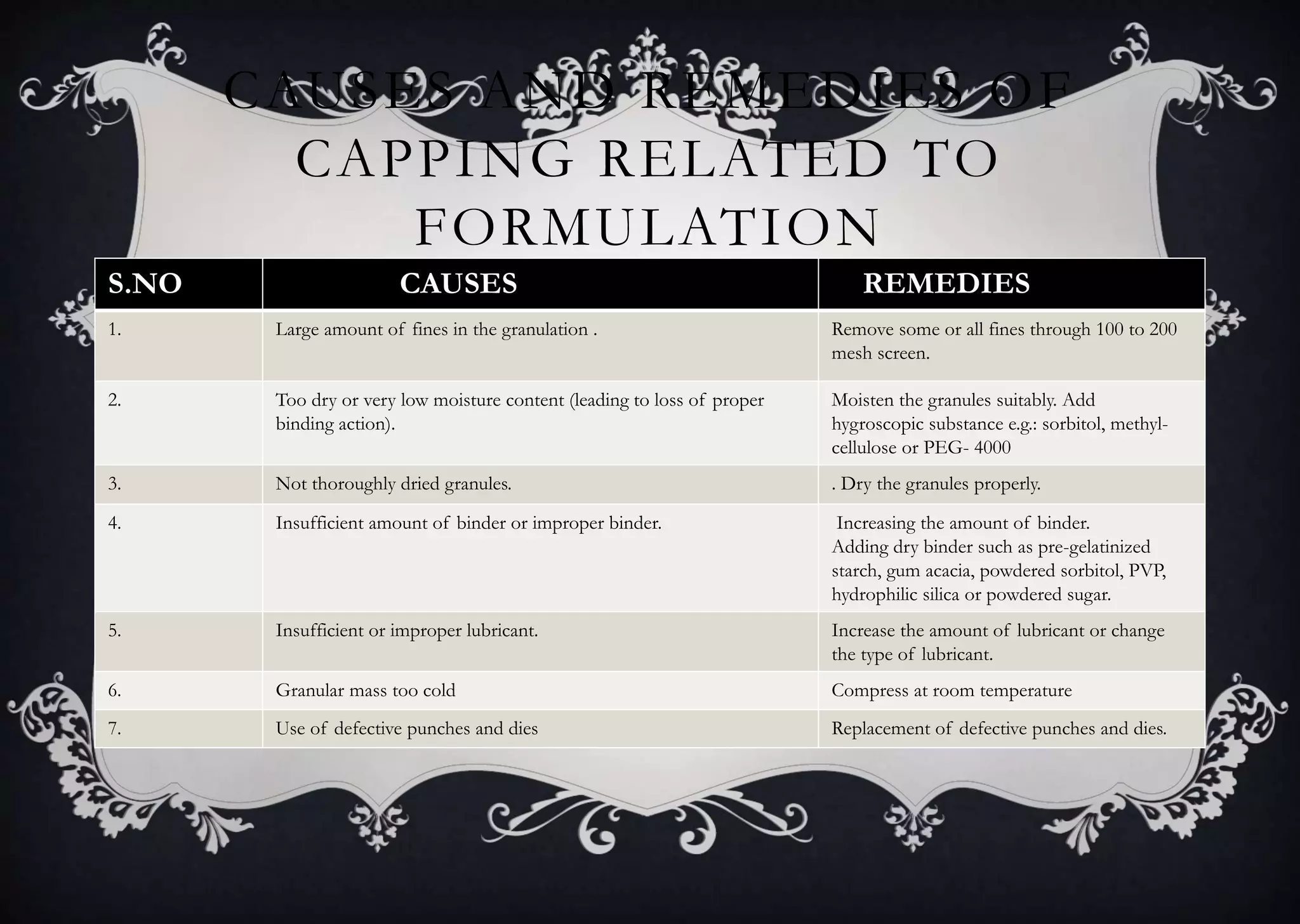
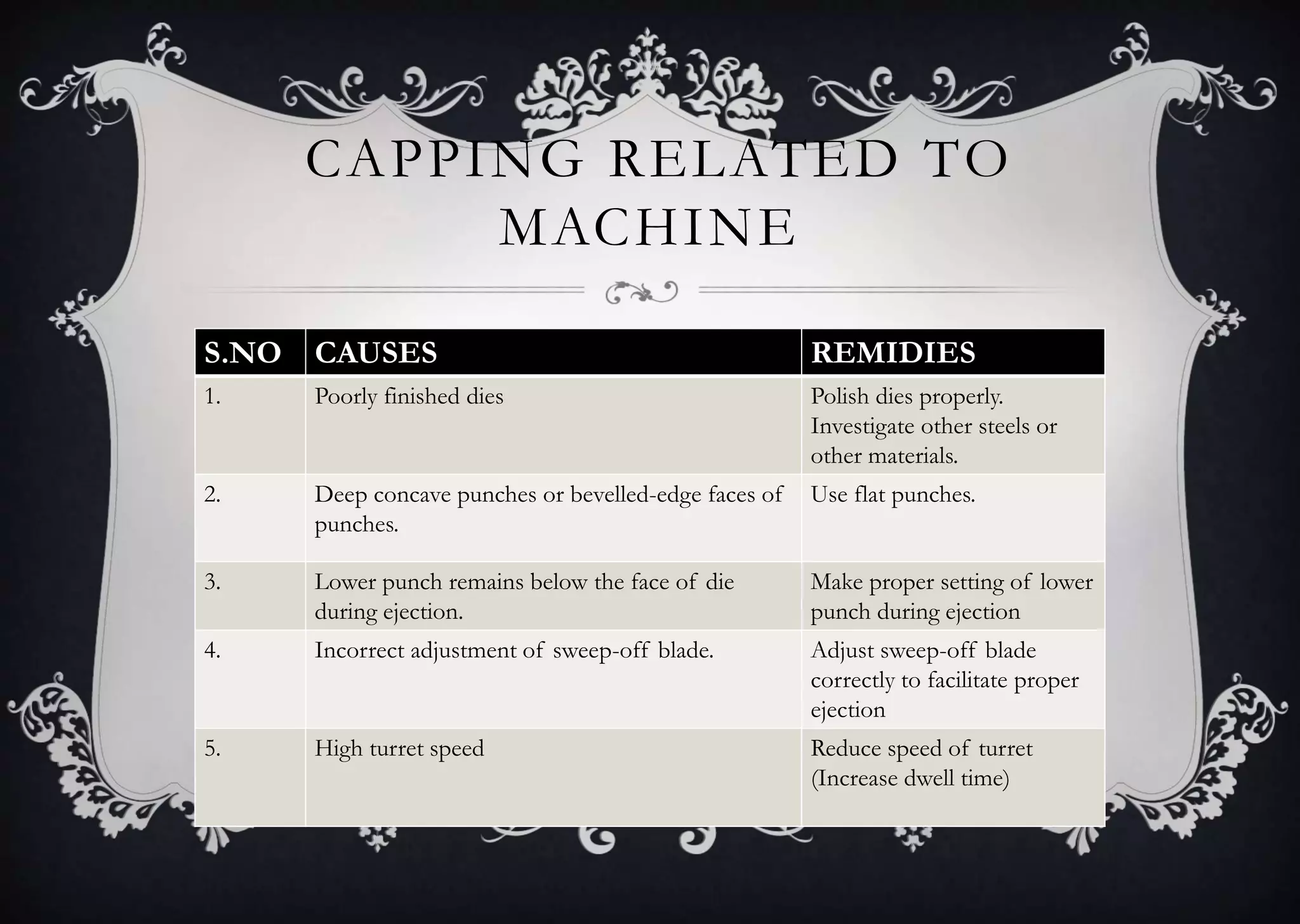
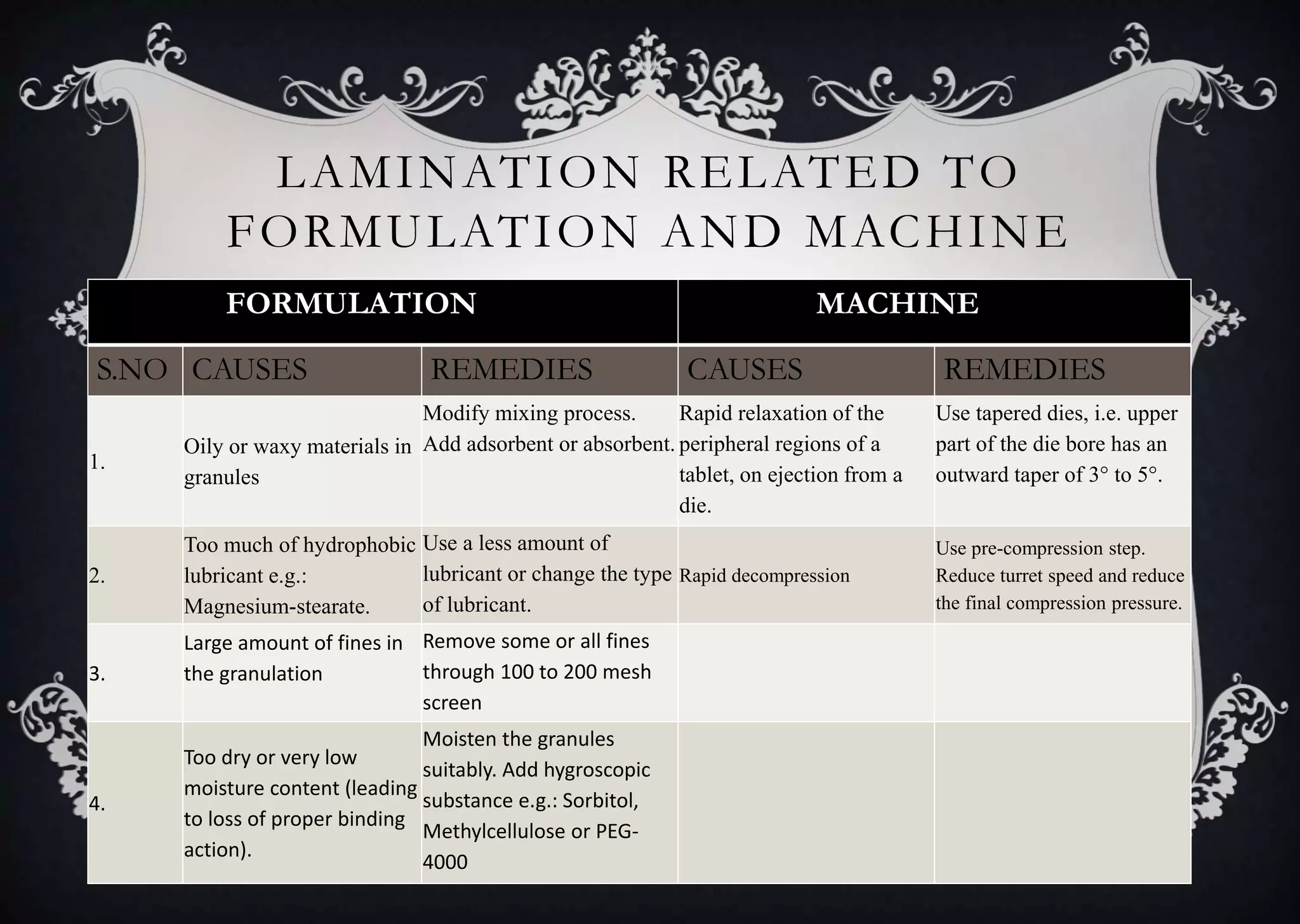
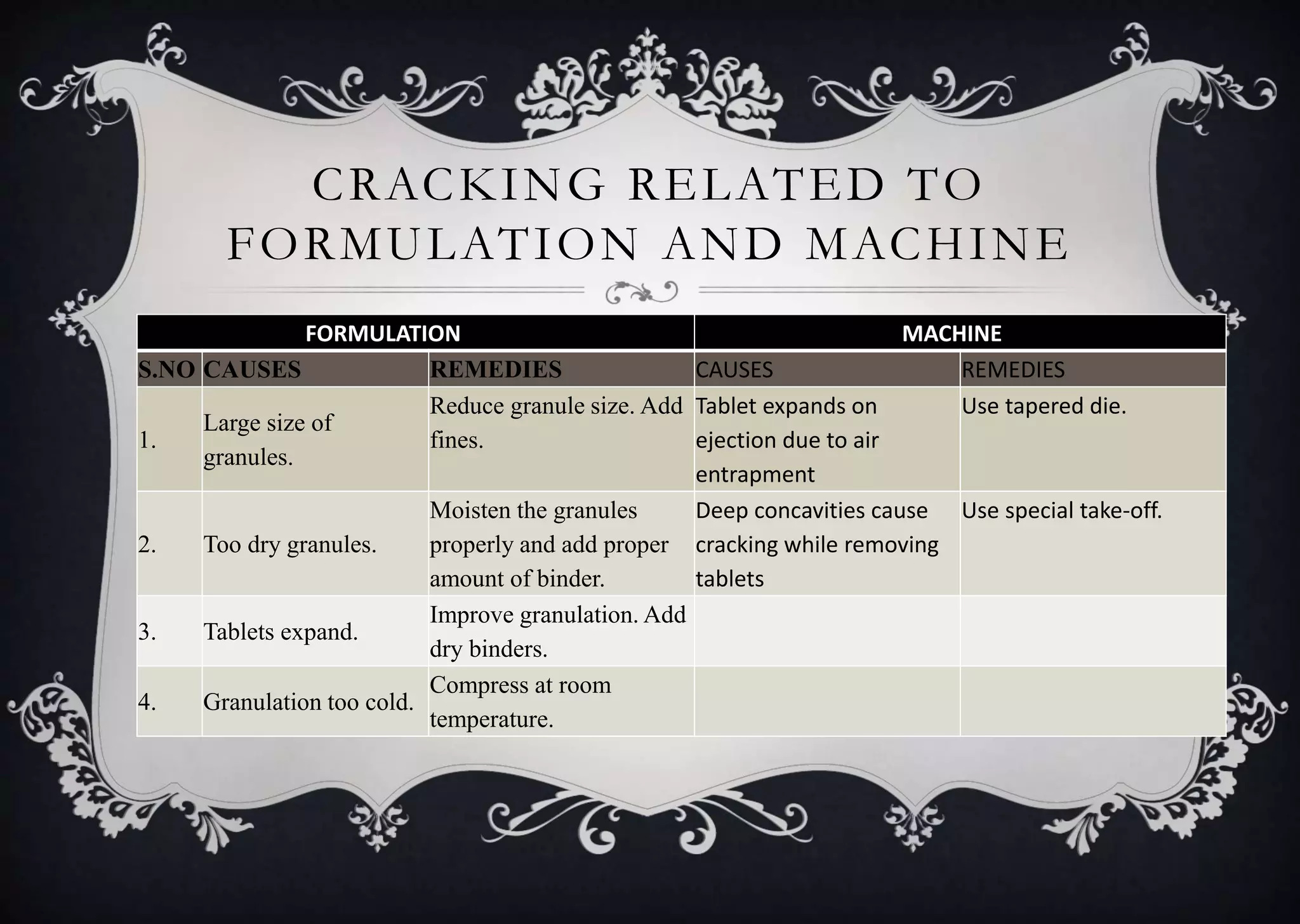
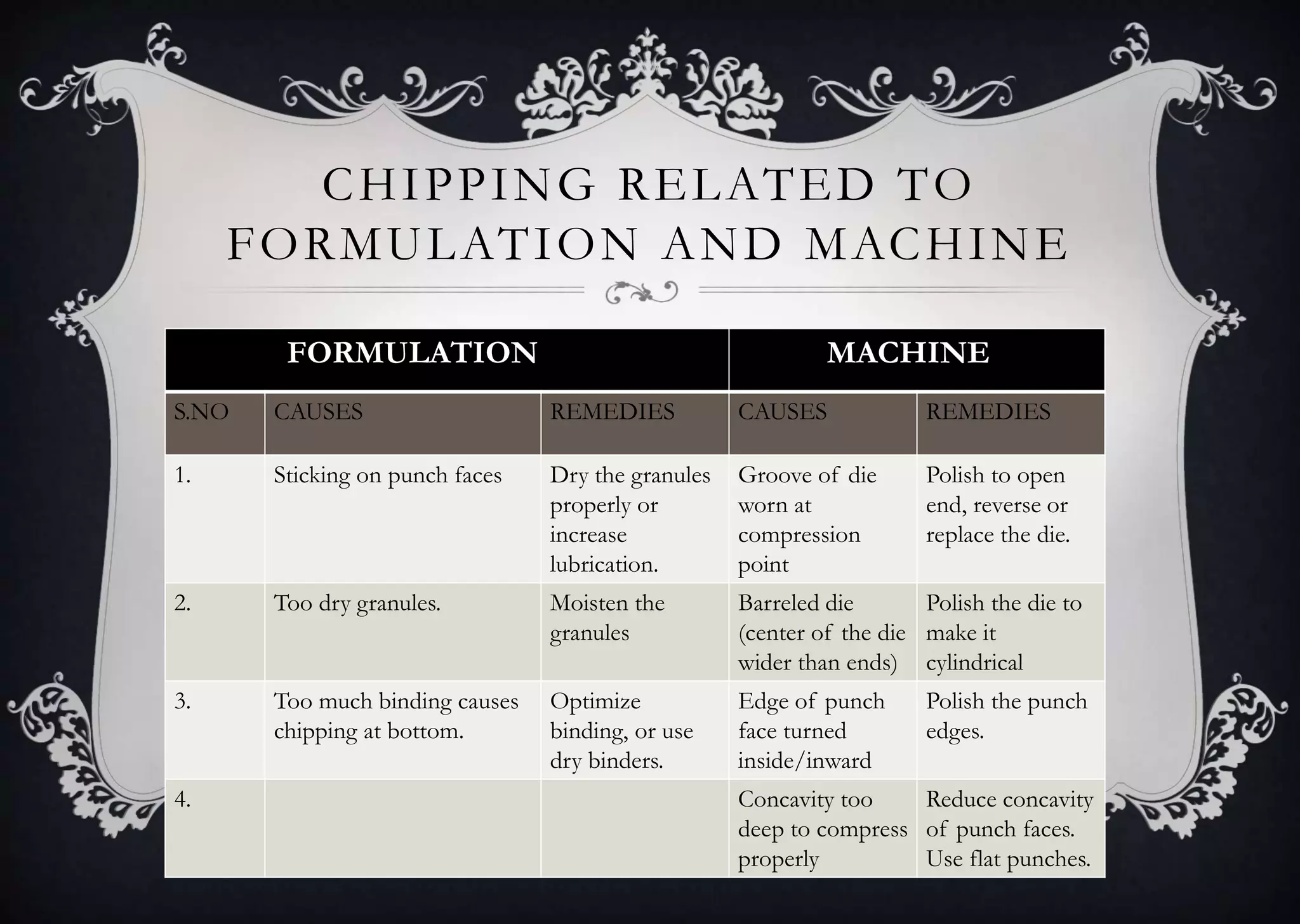
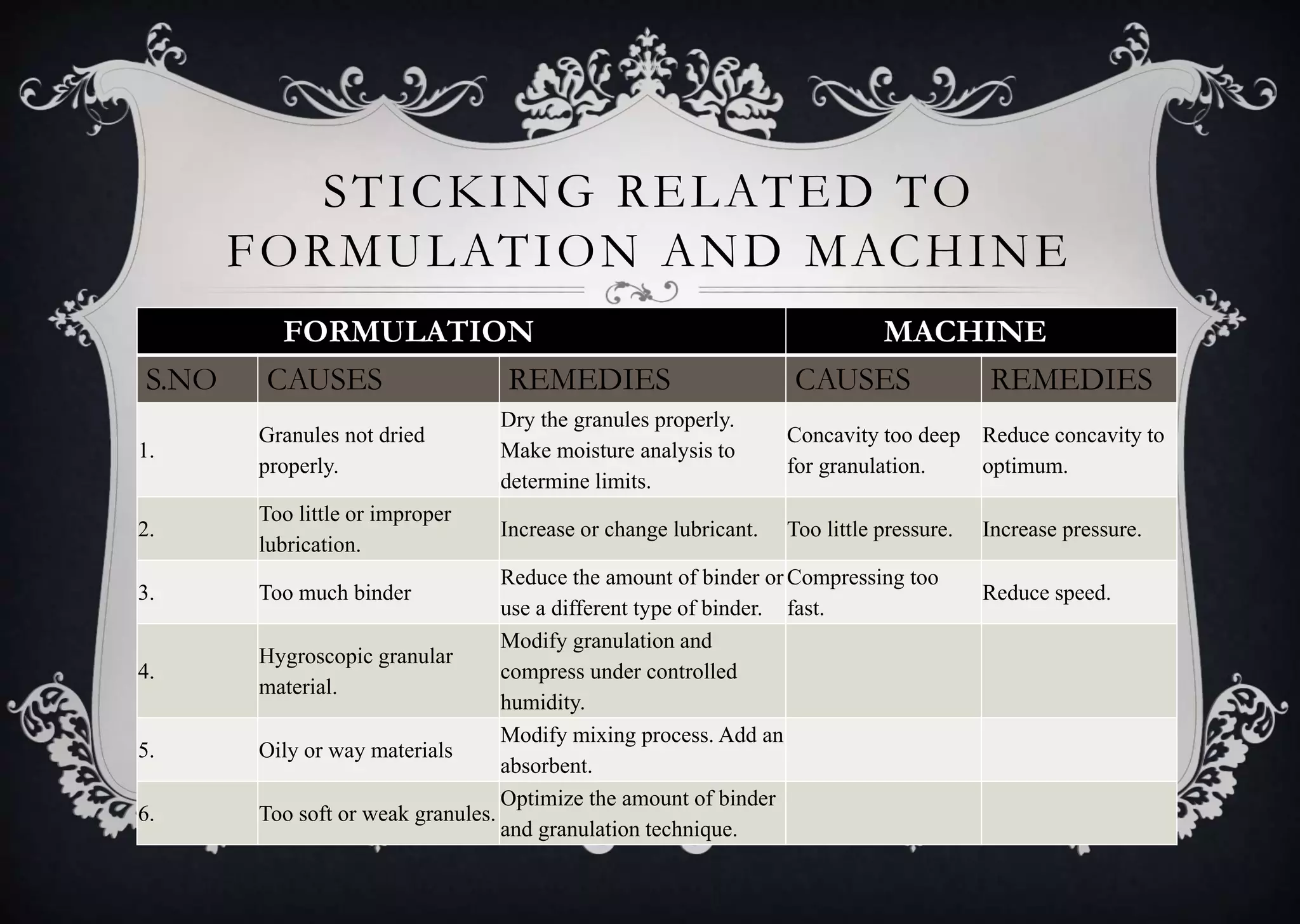
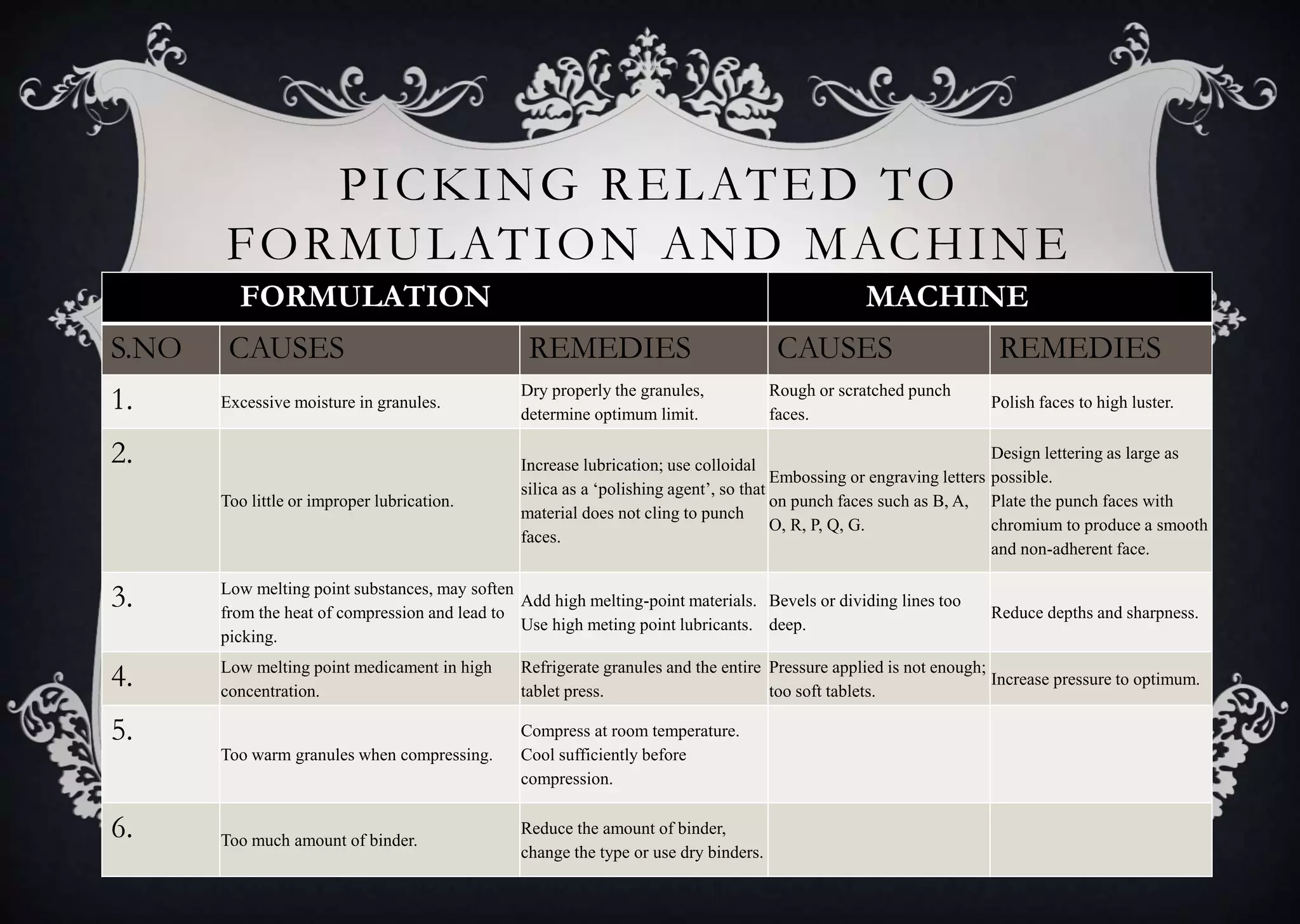
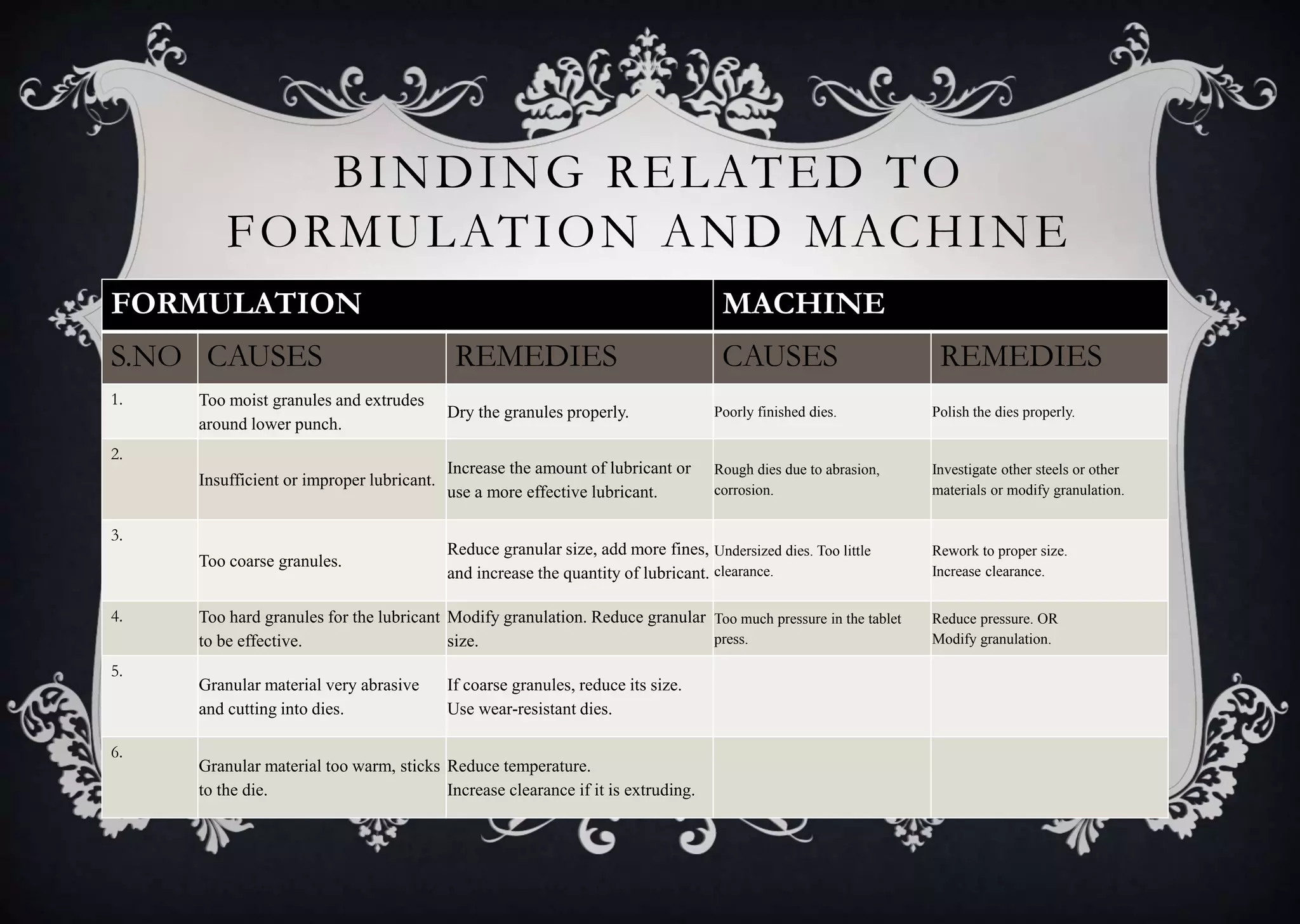
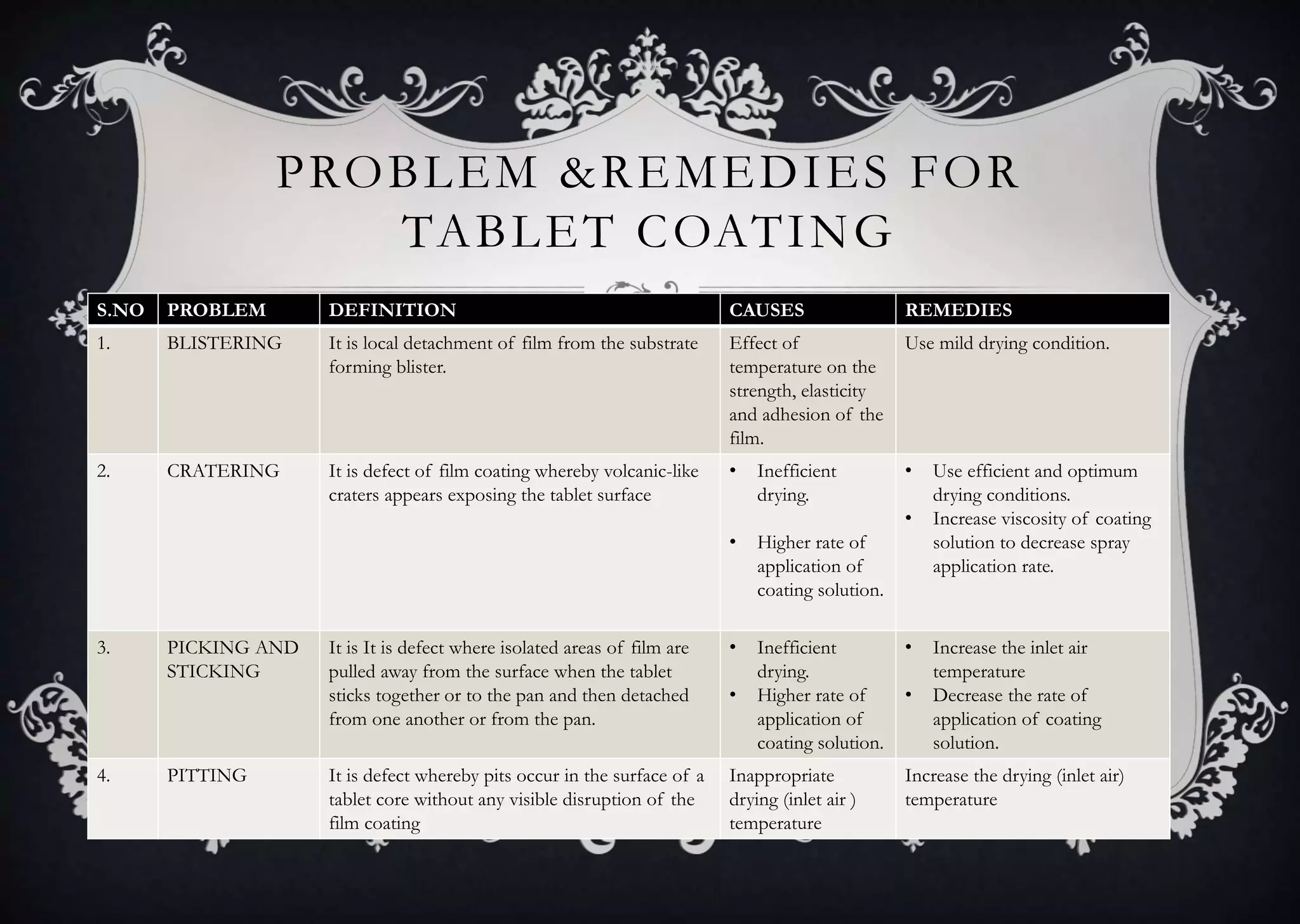
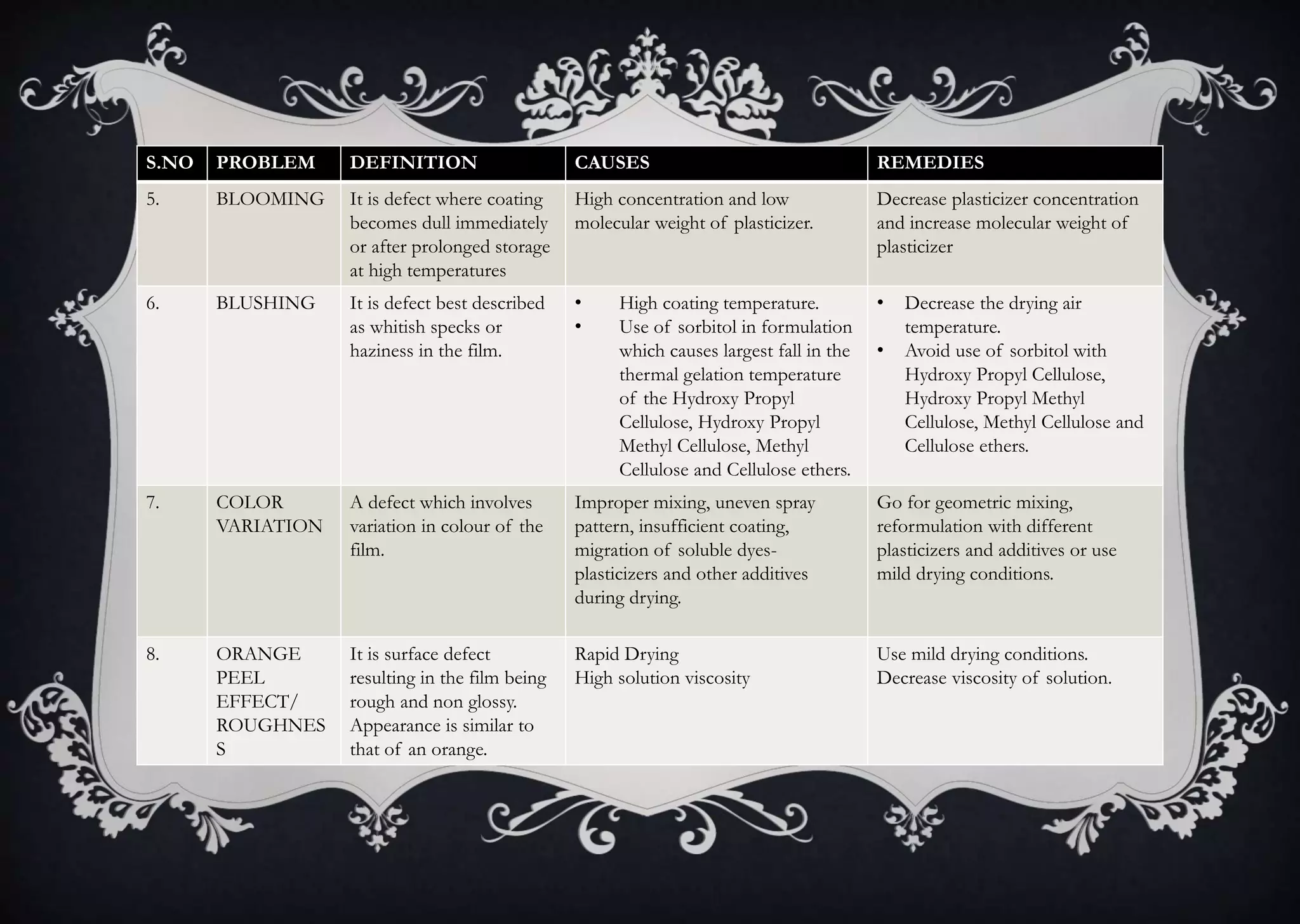
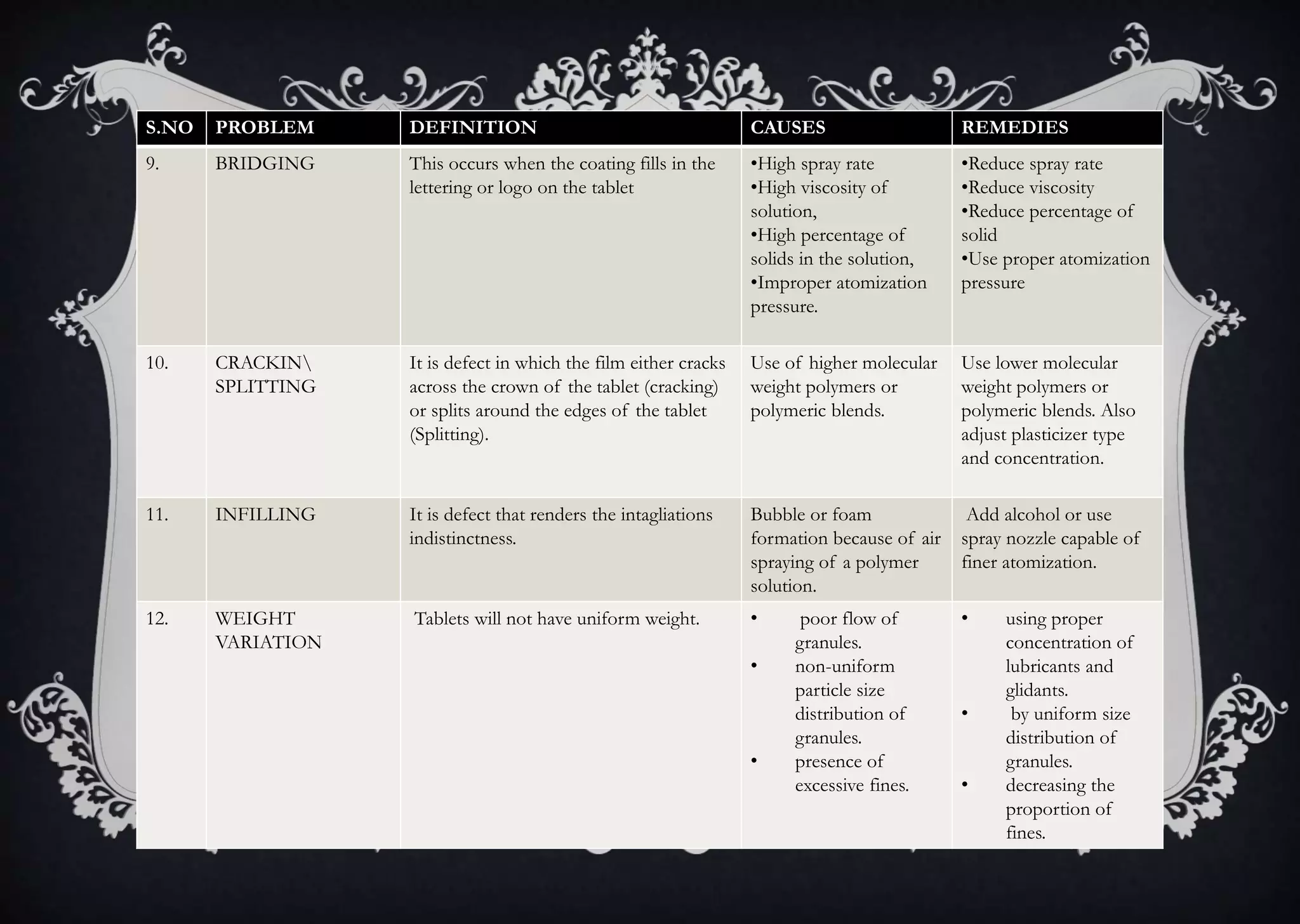
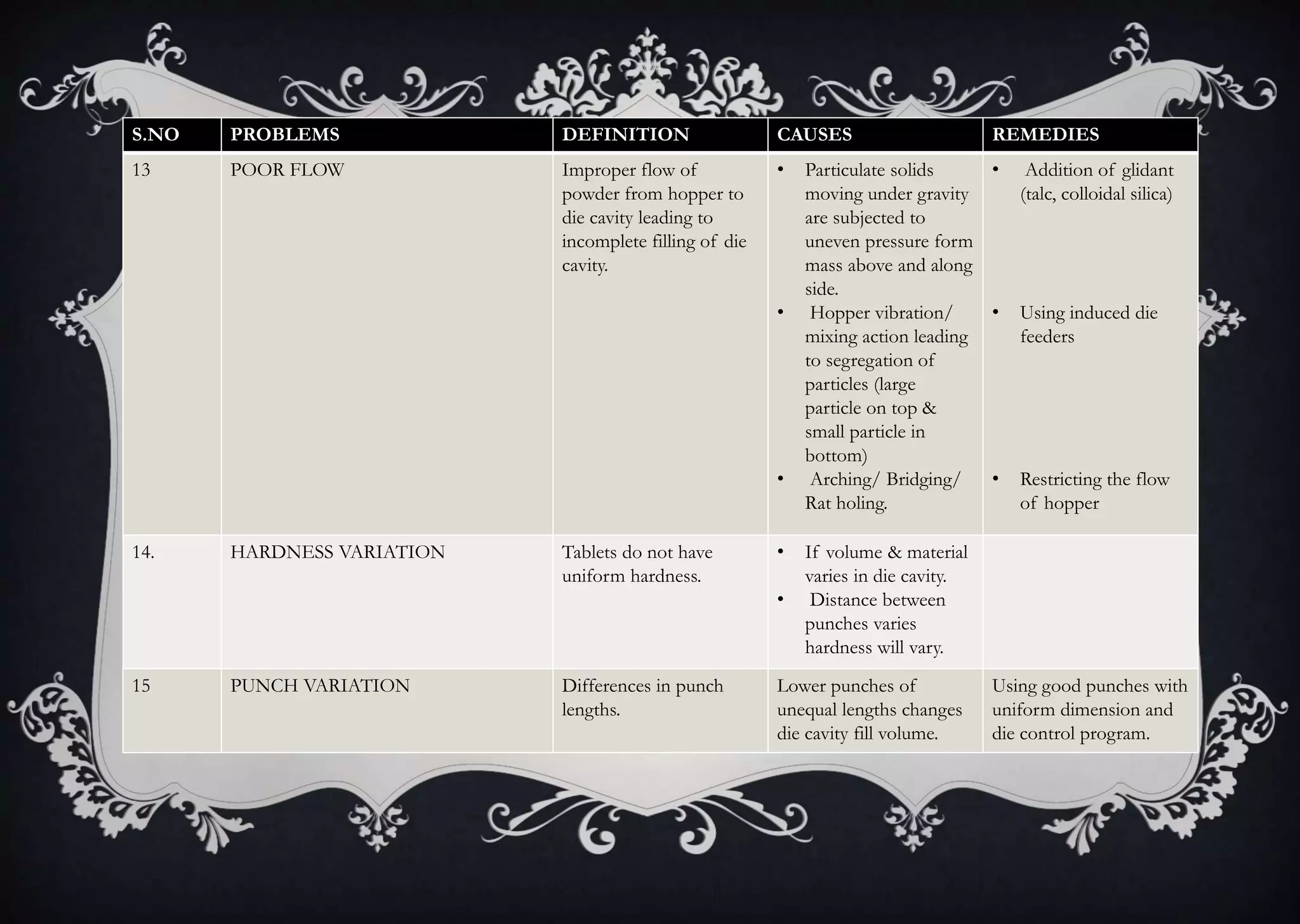
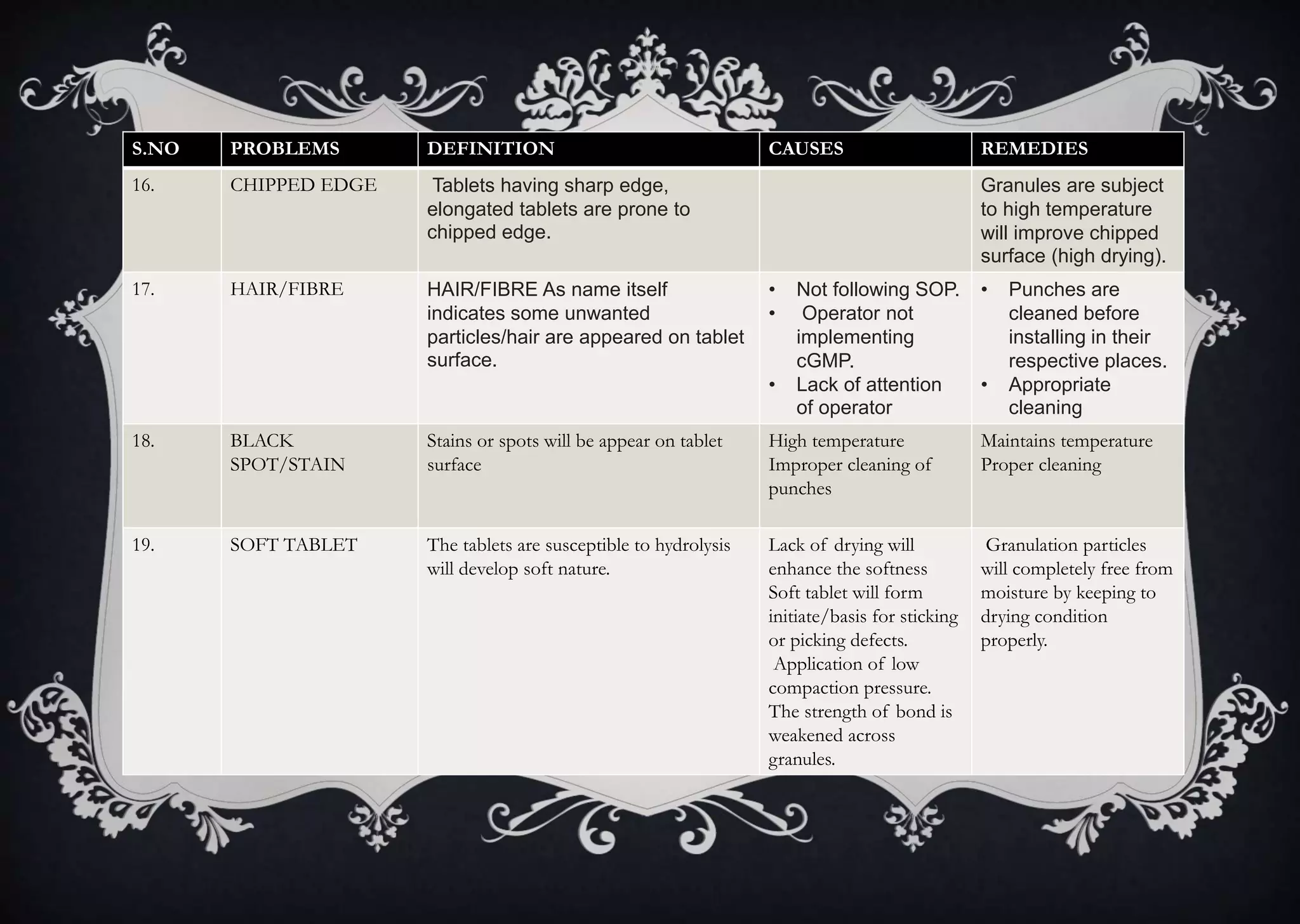
This document discusses various defects that can occur during the tableting process and their causes and remedies. It covers defects related to tableting, excipients, multiple factors, and the machine. Defects include capping, laminating, cracking, chipping, sticking, picking, binding, mottling, and double impressions. Causes can be related to formulation factors like moisture content, binder level, granule size, or machine factors like die and punch condition. Remedies involve modifying the formulation through changes to processing or composition, or adjusting machine settings and maintenance. The document provides detailed tables outlining specific causes and recommended remedies for different common defects.