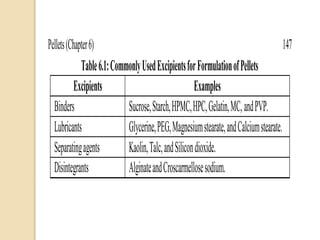
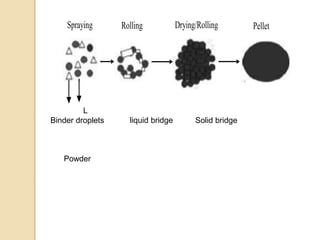
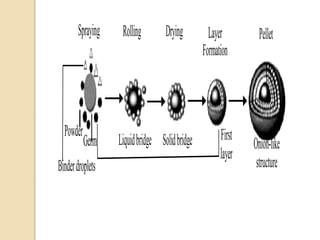
Pellets are small, spherical granules typically between 500-1500μm in size. They can be produced through various processes including extrusion-spheronization, suspension layering, and powder layering. Pellets offer benefits over other dosage forms like reduced variability in gastric emptying and plasma drug levels. Excipients used in pellet formulations include binders, fillers, lubricants, and coating agents. Pellets show advantages for swallowing and allow inclusion of incompatible drugs or varied release mechanisms in a single dosage form.