Embed presentation
Download to read offline




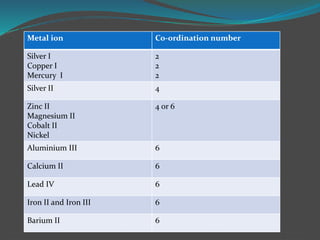


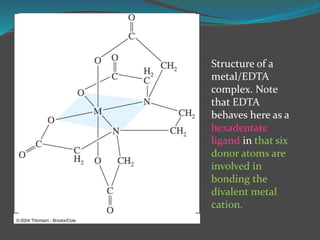




















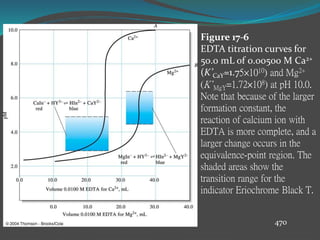






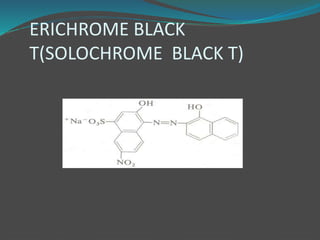












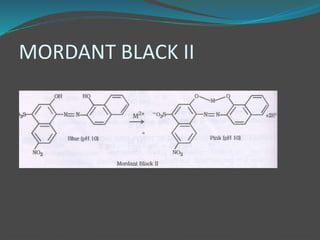






This document discusses complexometric titration, which involves titrating a metal ion with a complexing agent or chelating agent. It provides examples of different types of complexometric titrations including direct titration, back titration, and replacement titration. Assays for several substances using complexometric titration methods are described, such as magnesium sulfate using EDTA as the titrant, and calcium carbonate where the carbonate is dissolved using acid prior to titration.






















































