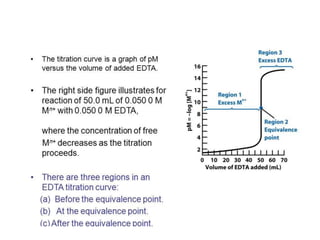
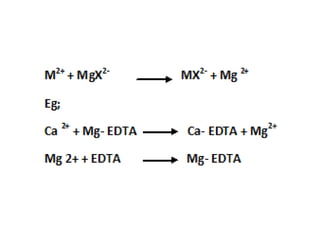
The document discusses different types of complexometric titration methods including direct titration, backtitration, replacement/substitution titration, and indirect titration. Direct titration involves directly titrating a metal ion solution with EDTA. Backtitration involves adding excess EDTA and back-titrating the excess with a second metal ion. Replacement/substitution titration involves quantitatively displacing one metal ion with the metal ion being determined. Indirect titration is used for anions by first precipitating them with a metal cation before titrating the metal with EDTA. The document also discusses titration curves in complexometric titration and EDTA as the most commonly used titrant.









![pM [negative logarithm of the free metal ion
concentration (-log Mn+)] is plotted against the volume
of EDTA solution added , a point of inflexion occurs at
the equivalence point.
Buffer plays an important role in complexometric
titrations , if solubility product of the metal hydroxide
is low, it may be precipitated if the hydroxyl ion
concentration is increased too much(alkaline pH) .
Also at lower pH values(acidic pH) when the
concentration of the metal ions is lower , the stability
constant of the complexes will not be so high.](https://image.slidesharecdn.com/typesofcomplexometrictitration-210301065259/85/Types-of-complexometric-titration-11-320.jpg)