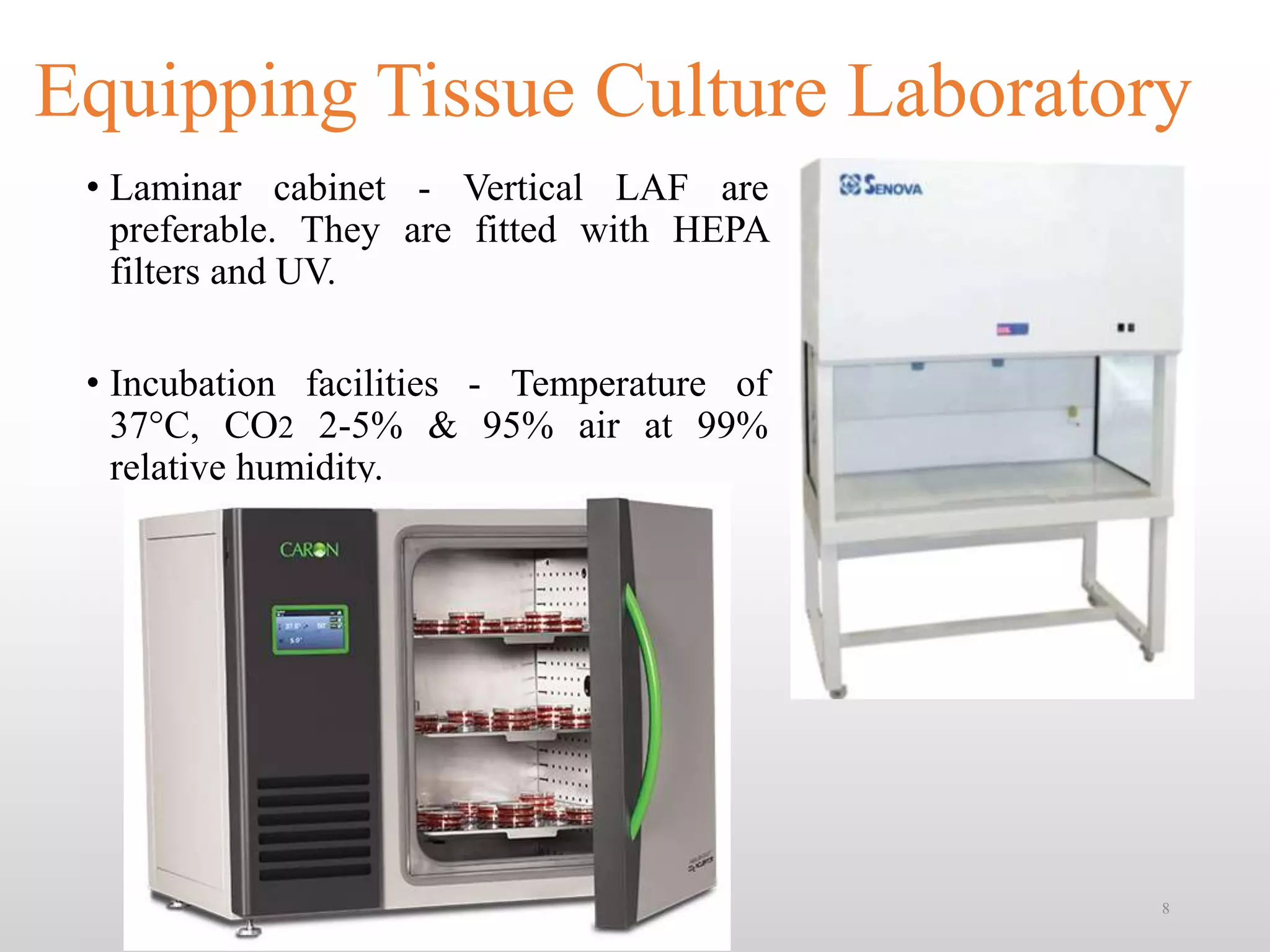
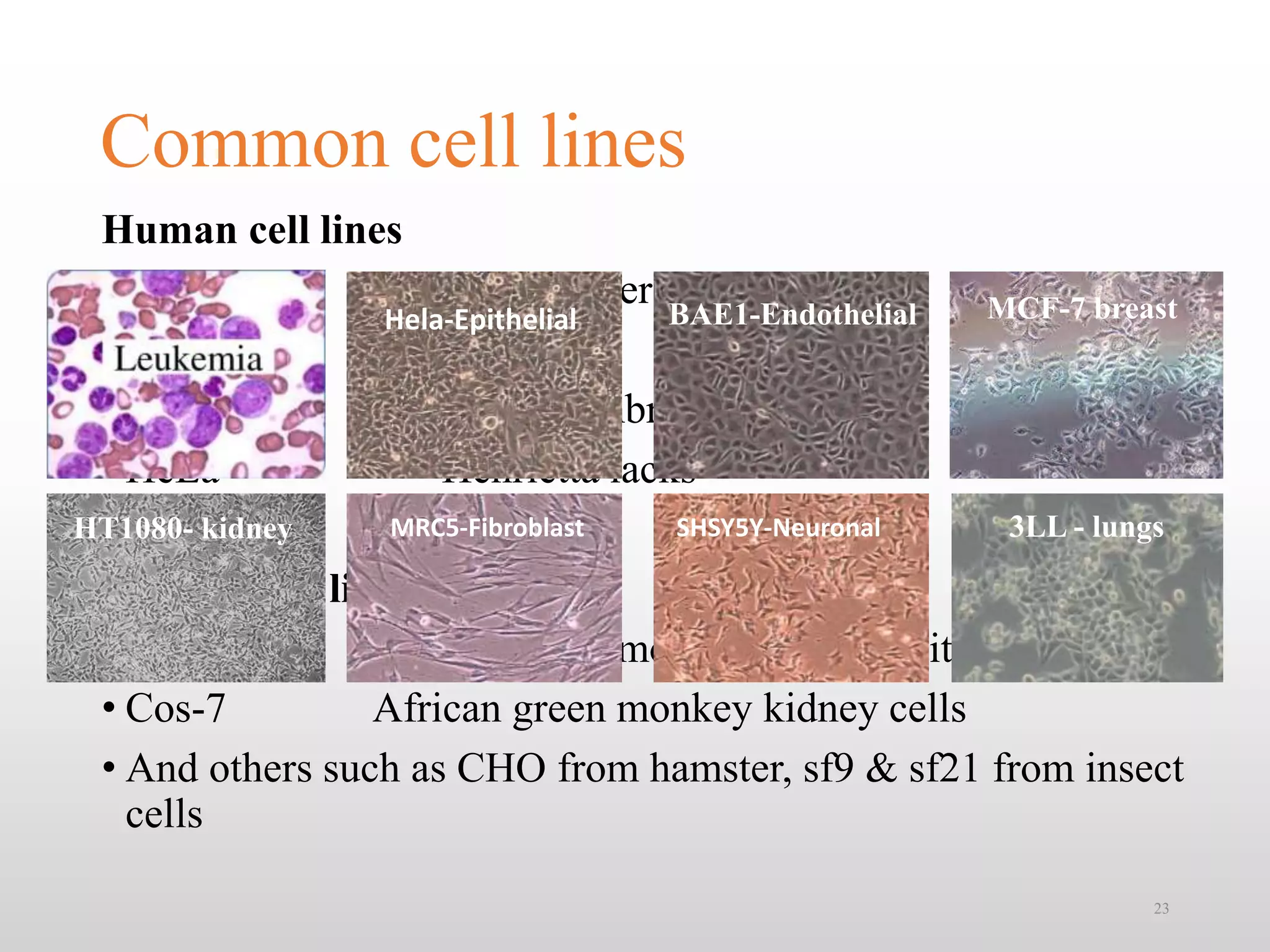
This document discusses animal cell culture techniques, including their historical development and applications in biotechnology. Key topics include the establishment of cell lines, the use of different culture media, and the implications for research and pharmaceutical production. Additionally, it highlights the advantages, limitations, and requirements for successful cell culture practices.