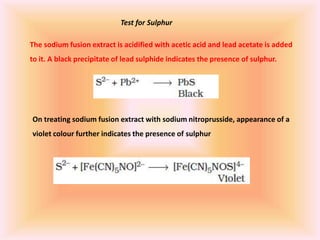
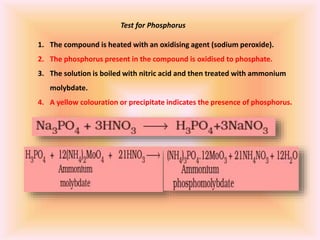
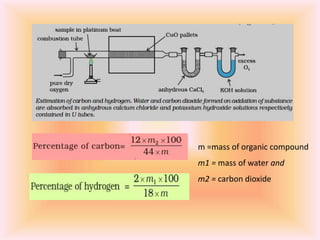
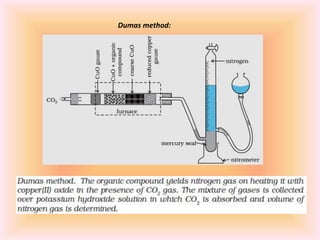
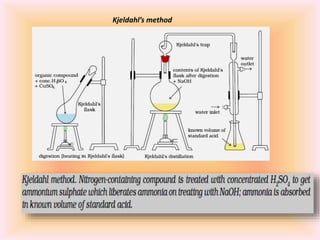
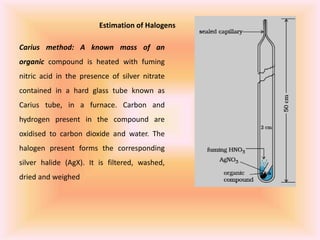
The document outlines methods for the qualitative and quantitative analysis of organic compounds, focusing on the detection of elements such as carbon, hydrogen, nitrogen, sulfur, halogens, phosphorus, and oxygen. Techniques include heating with copper(II) oxide, Lassaigne's test, Dumas and Kjeldahl methods for nitrogen estimation, and carius method for halogen analysis. Additionally, it describes how to calculate percentages of these elements based on experimental data and reactions.