2 chemical bonding learning outcomes
•Download as DOCX, PDF•
0 likes•6,900 views
Report
Share
Report
Share
Recommended
All chapters of engineering chemistry

IT the all chapter in eng chemistry for btech 1st years under calicut university
CBSE Class 11 Chemistry Chapter 2 (The Structure of Atom)

CBSE Class 11 Chemistry Chapter 2 (The structure of atom) | Homi-Institute
Nano - Chemistry

A presentation of the topic - "Nano-chemistry". This presentation is useful for all engineering students. I have done this presentation as for a chemistry project. This presentation can also be used for presenting at any college for any contest.
*If uploading in any other website, please provide the credits.*
Recommended
All chapters of engineering chemistry

IT the all chapter in eng chemistry for btech 1st years under calicut university
CBSE Class 11 Chemistry Chapter 2 (The Structure of Atom)

CBSE Class 11 Chemistry Chapter 2 (The structure of atom) | Homi-Institute
Nano - Chemistry

A presentation of the topic - "Nano-chemistry". This presentation is useful for all engineering students. I have done this presentation as for a chemistry project. This presentation can also be used for presenting at any college for any contest.
*If uploading in any other website, please provide the credits.*
SEMICONDUCTORS,BAND THEORY OF SOLIDS,FERMI-DIRAC PROBABILITY,DISTRIBUTION FUN...

This PPT contains valence band,conduction band& forbidden energy gap,Free carrier charge density,intrinsic and extrinsic semiconductors,Conductivity in semiconductors
Semester - I B) Reactive Intermediates by Dr Pramod R Padole

B) Reactive intermediates : Carbocations, Carbanions and Free Readicals: their generation, stability & reactions.
Introduction to coordination chemistry

This is a introductory presentation for the beginners who wish to understand the basics of coordination chemistry.
Electrochemistry

lecture slide on:
Gibbs free energy and Nernst Equation, Faradaic Processes and Factors Affecting Rates of Electrode Reactions, Potentials and Thermodynamics of Cells, Kinetics of Electrode Reactions, Kinetic controlled reactions,Essentials of Electrode Reactions,BUTLER-VOLMER MODEL FOR THE ONE-STEP, ONE-ELECTRON PROCESS,Current-overpotential curves for the system, Mass Transfer by Migration And Diffusion,MASS-TRANSFER-CONTROLLED REACTIONS,
Photochemical reactions, 8(2)

Photoelectric Effect and Photochemical Reactions. Photons of Light and Chemical Reactions. photodissociation of O2. It was found that Stopping voltage is proportional to the frequency of the incident light but independent of the light intensity
Ionic solids vs Covalent solids

Solids can be classified as Ionic solids, covalent network solids, metallic solids and molecular solids.
Term symbols

Reference,
https://en.wikipedia.org/wiki/Term_symbol
James E. Huheey, Ellen A. Keiter, Richard L.Keiter and Okhil K. Medhi, Inorganic Chemistry, Principles of Structure and Reactivity. 4th Edn. Pearsons
Organic reaction mechanism full

Organic reaction mechanism and intermediates their stabilities fully discussed.
Transition metal complexes unit 1

this ppt looks at different complexes as well as ligand exchange and its explanation.
Green chemistry, Its Applications and Benefits

Introduction to the concept of Green Chemistry and why is it a viable option to switch into.
More Related Content
What's hot
SEMICONDUCTORS,BAND THEORY OF SOLIDS,FERMI-DIRAC PROBABILITY,DISTRIBUTION FUN...

This PPT contains valence band,conduction band& forbidden energy gap,Free carrier charge density,intrinsic and extrinsic semiconductors,Conductivity in semiconductors
Semester - I B) Reactive Intermediates by Dr Pramod R Padole

B) Reactive intermediates : Carbocations, Carbanions and Free Readicals: their generation, stability & reactions.
Introduction to coordination chemistry

This is a introductory presentation for the beginners who wish to understand the basics of coordination chemistry.
Electrochemistry

lecture slide on:
Gibbs free energy and Nernst Equation, Faradaic Processes and Factors Affecting Rates of Electrode Reactions, Potentials and Thermodynamics of Cells, Kinetics of Electrode Reactions, Kinetic controlled reactions,Essentials of Electrode Reactions,BUTLER-VOLMER MODEL FOR THE ONE-STEP, ONE-ELECTRON PROCESS,Current-overpotential curves for the system, Mass Transfer by Migration And Diffusion,MASS-TRANSFER-CONTROLLED REACTIONS,
Photochemical reactions, 8(2)

Photoelectric Effect and Photochemical Reactions. Photons of Light and Chemical Reactions. photodissociation of O2. It was found that Stopping voltage is proportional to the frequency of the incident light but independent of the light intensity
Ionic solids vs Covalent solids

Solids can be classified as Ionic solids, covalent network solids, metallic solids and molecular solids.
Term symbols

Reference,
https://en.wikipedia.org/wiki/Term_symbol
James E. Huheey, Ellen A. Keiter, Richard L.Keiter and Okhil K. Medhi, Inorganic Chemistry, Principles of Structure and Reactivity. 4th Edn. Pearsons
Organic reaction mechanism full

Organic reaction mechanism and intermediates their stabilities fully discussed.
Transition metal complexes unit 1

this ppt looks at different complexes as well as ligand exchange and its explanation.
Green chemistry, Its Applications and Benefits

Introduction to the concept of Green Chemistry and why is it a viable option to switch into.
What's hot (20)
SEMICONDUCTORS,BAND THEORY OF SOLIDS,FERMI-DIRAC PROBABILITY,DISTRIBUTION FUN...

SEMICONDUCTORS,BAND THEORY OF SOLIDS,FERMI-DIRAC PROBABILITY,DISTRIBUTION FUN...
Semester - I B) Reactive Intermediates by Dr Pramod R Padole

Semester - I B) Reactive Intermediates by Dr Pramod R Padole
Viewers also liked
Viewers also liked (20)
3 stoichiometry, formulas and equations learning outcomes

3 stoichiometry, formulas and equations learning outcomes
Environmentatl chemistry water (questions and answers)

Environmentatl chemistry water (questions and answers)
Similar to 2 chemical bonding learning outcomes
Most Essential Learning Competencies (MELC) in Senior High School (STEM) Gene...

Most Essential Learning Competencies (MELC) in Senior High School (STEM) Gene...EngineerPH EducatorPH
https://www.deped.gov.ph/wp-content/uploads/2019/01/General-Chemistry-1-and-2.pdf
General Chemistry
GenChem
STEM
Science, Technology, Engineering, and Mathematics
K to 12 Senior High School STEM Specialized Subject – General Chemistry 1 and 2
Quarter 1 – General Chemistry 1
Matter and Its Properties
Measurements
Atoms, Molecules and Ions
Stoichiometry
Percent Composition and Chemical Formulas
Chemical reactions and chemical equations
Mass Relationships in Chemical Reactions
Gases
Dalton’s Law of partial pressures
Gas stoichiometry
Kinetic molecular theory of gases
Quarter 2 – General Chemistry 1
Electronic Structure of Atoms
Electronic Structure and Periodicity
Chemical Bonding
Organic compounds
Quarter 3 – General Chemistry 2
Intermolecular Forces and Liquids and Solids
Physical Properties of Solutions
Thermochemistry
Chemical Kinetics
Quarter 4 – General Chemistry 2
Chemical Thermodynamics
Chemical Equilibrium
Acid-Base Equilibria and Salt Equilibria
Electrochemistry13 808 PM docs.google.com Covalent Bonding and lonic Bonding study.pdf

10. Identify and discuss the goods oriented remedies of the seller and the buyer 11. ldentify and
discuss the moncy oriented damages of the seller and the buyer 12 identify and discuss the
\"specific performance\" remedies of the seller and buyer.
Solution
Q. 10) Goods oriented remedies of a buyer are as follows
1) When seller became insolvent and buyer wants to recover the goods purchased
In this case , When seller has paid first installment of proice so now he has booked special
property interest in the identified good and seller became insolvent within 10 days following first
receipt of price then to get the good from seller , buyer needs to pay remaining portion of price
and get the product.
2) To sue the buyer.
In this case , when seller is unlawfully not ready to give the product to buyer as buyer has paid in
full price of goods then buyer can sue him.
3) Buyer can enforce security interest in goods
in this case , remedy shall be when buyer fully rejects but he has the product with himself then
buyer has security interest to the extent of any expenses incurred by buyer .
Buyer shall sell the goods and pay the proceeds to seller above the security interest.
Goods oriented remedies of seller are as follows:
1) When buyer breaches
In this case , seller can withhold the delivery of remaining goods
2) When seller comes to know that buyer has become insolvent
In this case , seller will stop the delivery of goods till the time goods are in transit.
3) When buyer breaches and goods are in process
in this case , seller shall stop the production and unfinished goods shall be sold as scrap so the
damages of seller can be recovered
Q 11) Money oriented remedies of buyer are as follows
1) When seller breaches regarding payment made
in this case , buyer should recover as much price he has already paid and then return the goods.
2) when seller breaches then buyer should cover
in this case , buyer can buy substitute while acting in good faith and then try to cover the
expenses incurred. If buyer doesnt cover then he will not be able to cover his damages
3) When seller breaches then buyer needs to cover damages for non delivery
in this case , damages = (contract price -market price ) + expenses incurred.
Money oriented remedies of seller are as follows
1) When buyer breaches then
in this case , seller shall resell the goods to recover damages in good faith and evn seller may
give notice to buyer for breach of contract
2) When buyer breaches regarding non acceptance of goods
in this case , seller shall recover damages as the difference between the market price and place of
sell plus any expenses incurred.
3) When buyer breaches regarding price to be paid
in this case , seller shall recover the price by conforming goods are lost and damaged after risk of
loss is transfered to buyer
Specific performance remedies of the buyer are as follows
1) When seller breaches then buyer may cancel
in this case , buyer can cancel by repudiating the contract.
2) when sel.
Similar to 2 chemical bonding learning outcomes (20)
1 periodic table and atomic structure learning outcomes

1 periodic table and atomic structure learning outcomes
1periodictableandatomicstructurelearningoutcomes 140308200450-phpapp02

1periodictableandatomicstructurelearningoutcomes 140308200450-phpapp02
Most Essential Learning Competencies (MELC) in Senior High School (STEM) Gene...

Most Essential Learning Competencies (MELC) in Senior High School (STEM) Gene...
13 808 PM docs.google.com Covalent Bonding and lonic Bonding study.pdf

13 808 PM docs.google.com Covalent Bonding and lonic Bonding study.pdf
More from Martin Brown
Jcsp library project Reading promotion - 10 great opening lines

Jcsp library project Reading promotion - 10 great opening lines
Jcsp library project Reading promotion - 10 great halloween reads - poster

Jcsp library project Reading promotion - 10 great halloween reads - poster
More from Martin Brown (20)
Jcsp library project Reading promotion - 10 great opening lines

Jcsp library project Reading promotion - 10 great opening lines
Jcsp library project Reading promotion - 10 great halloween reads - poster

Jcsp library project Reading promotion - 10 great halloween reads - poster
Recently uploaded
The Art Pastor's Guide to Sabbath | Steve Thomason

What is the purpose of the Sabbath Law in the Torah. It is interesting to compare how the context of the law shifts from Exodus to Deuteronomy. Who gets to rest, and why?
The approach at University of Liverpool.pptx

How libraries can support authors with open access requirements for UKRI funded books
Wednesday 22 May 2024, 14:00-15:00.
1.4 modern child centered education - mahatma gandhi-2.pptx

Child centred education is an educational approach that priorities the interest, needs and abilities of the child in the learning process.
CLASS 11 CBSE B.St Project AIDS TO TRADE - INSURANCE

Class 11 CBSE Business Studies Project ( AIDS TO TRADE - INSURANCE)
How to Create Map Views in the Odoo 17 ERP

The map views are useful for providing a geographical representation of data. They allow users to visualize and analyze the data in a more intuitive manner.
The geography of Taylor Swift - some ideas

Geographical themes connected with Taylor Swift's ERAS tour - coming to the UK in June 2024
How libraries can support authors with open access requirements for UKRI fund...

How libraries can support authors with open access requirements for UKRI funded books
Wednesday 22 May 2024, 14:00-15:00.
ESC Beyond Borders _From EU to You_ InfoPack general.pdf

ESC Beyond Borders _From EU to You_ InfoPack general.pdfFundacja Rozwoju Społeczeństwa Przedsiębiorczego
Wolontariat grupowy2024.06.01 Introducing a competency framework for languag learning materials ...

http://sandymillin.wordpress.com/iateflwebinar2024
Published classroom materials form the basis of syllabuses, drive teacher professional development, and have a potentially huge influence on learners, teachers and education systems. All teachers also create their own materials, whether a few sentences on a blackboard, a highly-structured fully-realised online course, or anything in between. Despite this, the knowledge and skills needed to create effective language learning materials are rarely part of teacher training, and are mostly learnt by trial and error.
Knowledge and skills frameworks, generally called competency frameworks, for ELT teachers, trainers and managers have existed for a few years now. However, until I created one for my MA dissertation, there wasn’t one drawing together what we need to know and do to be able to effectively produce language learning materials.
This webinar will introduce you to my framework, highlighting the key competencies I identified from my research. It will also show how anybody involved in language teaching (any language, not just English!), teacher training, managing schools or developing language learning materials can benefit from using the framework.
Polish students' mobility in the Czech Republic

Polish students mobility to the Czech Republic within eTwinning project "Medieval adventures with Marco Polo"
Instructions for Submissions thorugh G- Classroom.pptx

This presentation provides a briefing on how to upload submissions and documents in Google Classroom. It was prepared as part of an orientation for new Sainik School in-service teacher trainees. As a training officer, my goal is to ensure that you are comfortable and proficient with this essential tool for managing assignments and fostering student engagement.
Overview on Edible Vaccine: Pros & Cons with Mechanism

This ppt include the description of the edible vaccine i.e. a new concept over the traditional vaccine administered by injection.
Operation Blue Star - Saka Neela Tara

Operation “Blue Star” is the only event in the history of Independent India where the state went into war with its own people. Even after about 40 years it is not clear if it was culmination of states anger over people of the region, a political game of power or start of dictatorial chapter in the democratic setup.
The people of Punjab felt alienated from main stream due to denial of their just demands during a long democratic struggle since independence. As it happen all over the word, it led to militant struggle with great loss of lives of military, police and civilian personnel. Killing of Indira Gandhi and massacre of innocent Sikhs in Delhi and other India cities was also associated with this movement.
Home assignment II on Spectroscopy 2024 Answers.pdf

Answers to Home assignment on UV-Visible spectroscopy: Calculation of wavelength of UV-Visible absorption
Digital Tools and AI for Teaching Learning and Research

This Presentation in details discusses on Digital Tools and AI for Teaching Learning and Research
MARUTI SUZUKI- A Successful Joint Venture in India.pptx

Let us know about Maruti Suzuki, a successful Joint venture in India.
Students, digital devices and success - Andreas Schleicher - 27 May 2024..pptx

Andreas Schleicher presents at the OECD webinar ‘Digital devices in schools: detrimental distraction or secret to success?’ on 27 May 2024. The presentation was based on findings from PISA 2022 results and the webinar helped launch the PISA in Focus ‘Managing screen time: How to protect and equip students against distraction’ https://www.oecd-ilibrary.org/education/managing-screen-time_7c225af4-en and the OECD Education Policy Perspective ‘Students, digital devices and success’ can be found here - https://oe.cd/il/5yV
How to Make a Field invisible in Odoo 17

It is possible to hide or invisible some fields in odoo. Commonly using “invisible” attribute in the field definition to invisible the fields. This slide will show how to make a field invisible in odoo 17.
Recently uploaded (20)
The Art Pastor's Guide to Sabbath | Steve Thomason

The Art Pastor's Guide to Sabbath | Steve Thomason
1.4 modern child centered education - mahatma gandhi-2.pptx

1.4 modern child centered education - mahatma gandhi-2.pptx
CLASS 11 CBSE B.St Project AIDS TO TRADE - INSURANCE

CLASS 11 CBSE B.St Project AIDS TO TRADE - INSURANCE
How libraries can support authors with open access requirements for UKRI fund...

How libraries can support authors with open access requirements for UKRI fund...
ESC Beyond Borders _From EU to You_ InfoPack general.pdf

ESC Beyond Borders _From EU to You_ InfoPack general.pdf
2024.06.01 Introducing a competency framework for languag learning materials ...

2024.06.01 Introducing a competency framework for languag learning materials ...
Instructions for Submissions thorugh G- Classroom.pptx

Instructions for Submissions thorugh G- Classroom.pptx
Overview on Edible Vaccine: Pros & Cons with Mechanism

Overview on Edible Vaccine: Pros & Cons with Mechanism
Home assignment II on Spectroscopy 2024 Answers.pdf

Home assignment II on Spectroscopy 2024 Answers.pdf
Digital Tools and AI for Teaching Learning and Research

Digital Tools and AI for Teaching Learning and Research
MARUTI SUZUKI- A Successful Joint Venture in India.pptx

MARUTI SUZUKI- A Successful Joint Venture in India.pptx
Students, digital devices and success - Andreas Schleicher - 27 May 2024..pptx

Students, digital devices and success - Andreas Schleicher - 27 May 2024..pptx
2 chemical bonding learning outcomes
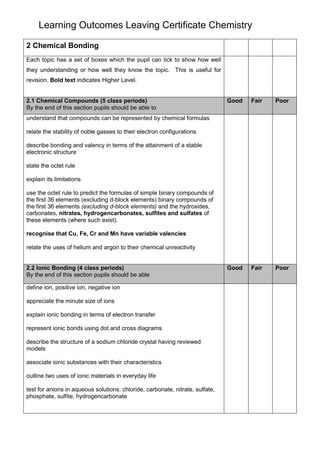
- 1. Learning Outcomes Leaving Certificate Chemistry 2 Chemical Bonding Each topic has a set of boxes which the pupil can tick to show how well they understanding or how well they know the topic. This is useful for revision. Bold text indicates Higher Level. 2.1 Chemical Compounds (5 class periods) By the end of this section pupils should be able to Good Fair Poor Good Fair Poor understand that compounds can be represented by chemical formulas relate the stability of noble gasses to their electron configurations describe bonding and valency in terms of the attainment of a stable electronic structure state the octet rule explain its limitations use the octet rule to predict the formulas of simple binary compounds of the first 36 elements (excluding d-block elements) binary compounds of the first 36 elements (excluding d-block elements) and the hydroxides, carbonates, nitrates, hydrogencarbonates, sulfites and sulfates of these elements (where such exist). recognise that Cu, Fe, Cr and Mn have variable valencies relate the uses of helium and argon to their chemical unreactivity 2.2 Ionic Bonding (4 class periods) By the end of this section pupils should be able define ion, positive ion, negative ion appreciate the minute size of ions explain ionic bonding in terms of electron transfer represent ionic bonds using dot and cross diagrams describe the structure of a sodium chloride crystal having reviewed models associate ionic substances with their characteristics outline two uses of ionic materials in everyday life test for anions in aqueous solutions: chloride, carbonate, nitrate, sulfate, phosphate, sulfite, hydrogencarbonate
- 2. Learning Outcomes Leaving Certificate Chemistry 2.3 Covalent Bonding (4 class periods) By the end of this section pupils should be able to Good Fair Poor Good Fair Poor Good Fair Poor define molecule appreciate the minute size of molecules explain covalent bonding in terms of the sharing of pairs of electrons (Single, double and triple covalent bonds) represent covalent bonds in molecules using dot and cross diagrams distinguish between sigma and pi bonding distinguish between polar and non-polar covalent bonding test a liquid for polarity using a charged plastic rod give examples of polar and non-polar materials in everyday life (two examples in each case) associate covalent substances with their characteristics test the solubility of ionic and covalent substances in different solvents 2.4 Electronegativity (2 class periods) By the end of this section pupils should be able define electronegativity recognise the trends in electronegativity values down a group and across a period explain the general trends in electronegativity values down a group across a period. relate differences in electronegativity to polarity of bonds predict bond type using electronegativity differences 2.5 Shapes of Molecules and Intermolecular Forces (5 class Periods) By the end of this section pupils should be able describe the shapes of simple molecules use appropriate modeling techniques to illustrate molecular shape explain the basis for electron pair repulsion theory use electron pair repulsion theory to explain the shapes of molecules of type ABn for up to four pairs of electrons around the central atom refer to bond angles (Shapes of molecules with pi bonds not to be considered)
- 3. Learning Outcomes Leaving Certificate Chemistry explain the relationship between symmetry and polarity in a molecule (dipole moments not required) describe and distinguish between intramolecular bonding and intermolecular forces (van der Waals’, dipole-dipole, hydrogen bonding) describe the effects of intermolecular forces on the boiling point of covalent substances relate the differences in boiling points of H2 and O2 , C2H2 and HCHO and of H2O and H2S to the effect of intermolecular forces 2.6 Oxidation Numbers (5 class periods) By the end of this section pupils should be able define oxidation number, oxidation state define oxidation and reduction in terms of change of oxidation numbers state the rules for oxidation numbers (exclude peroxides, except for hydrogen peroxide) calculate oxidation numbers of transition metals in their compounds and of other elements use oxidation numbers in nomenclature of transition metal compounds give an example of an oxidising and a reducing bleach Good Fair Poor