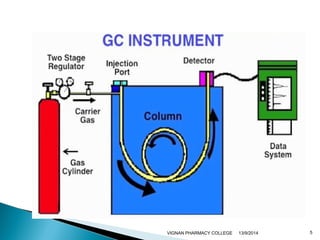
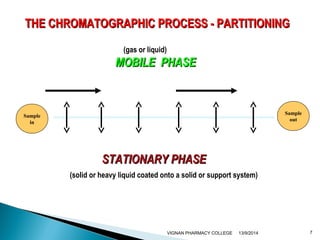
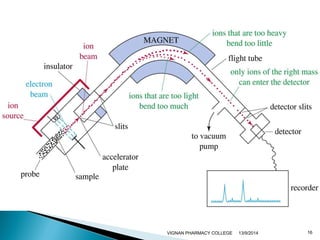
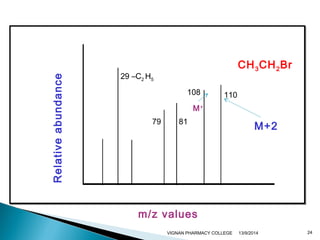
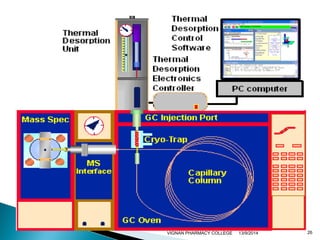
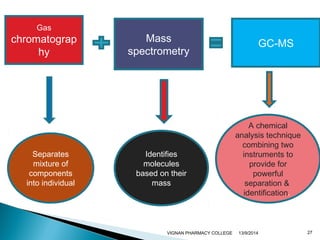
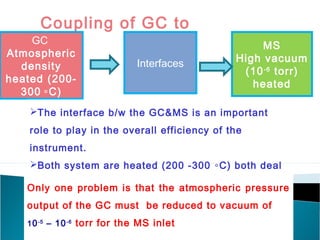
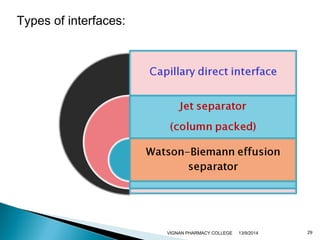
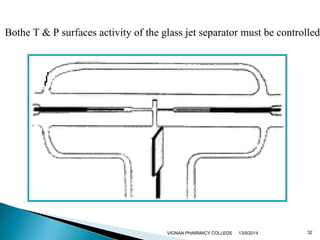
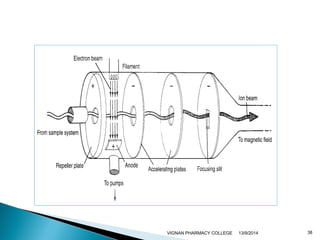
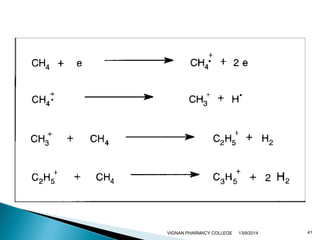
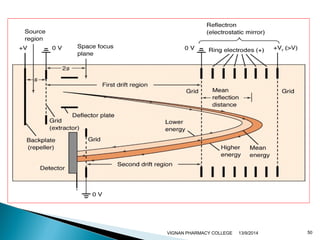
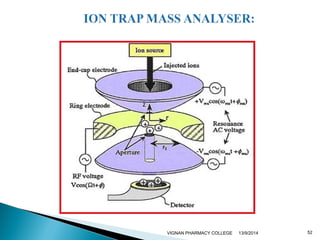
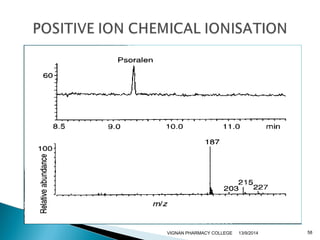
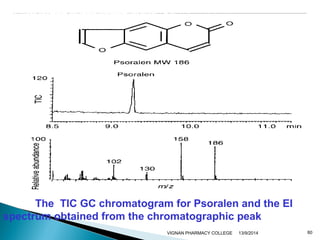
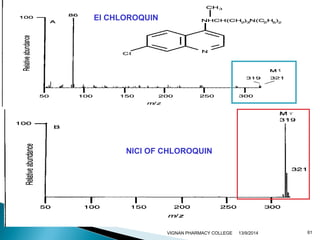
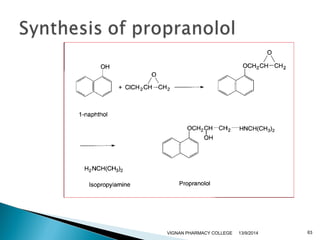
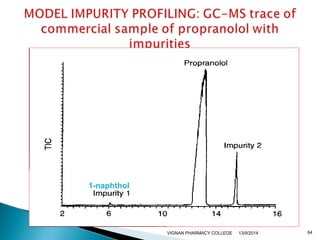
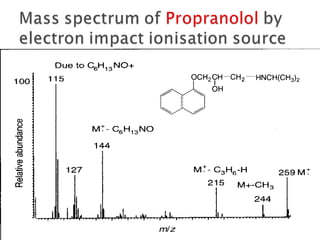
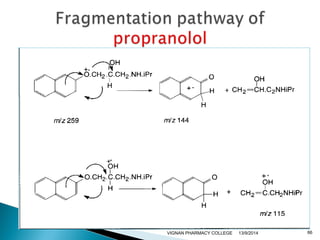
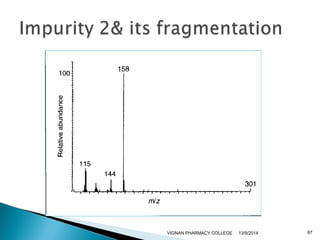
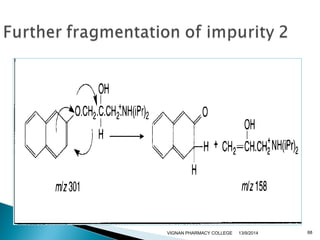
The document discusses gas chromatography (GC) and mass spectrometry (MS) as analytical techniques used for the separation and identification of chemical compounds. It explains the principles of GC, including the roles of mobile and stationary phases, and highlights the process of mass spectrometry in determining molecular weights and structures through ionization methods. Additionally, it covers the coupling of GC with MS for advanced analysis and various applications in fields like pharmaceuticals, environmental monitoring, and toxicology.