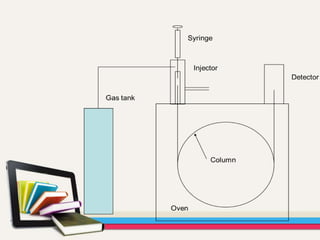
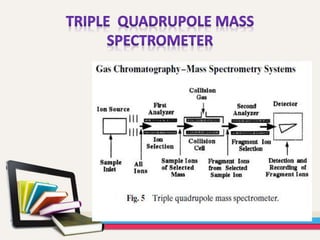
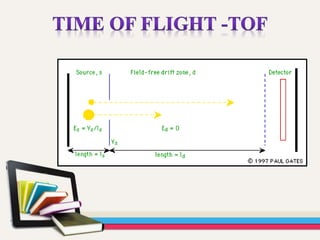
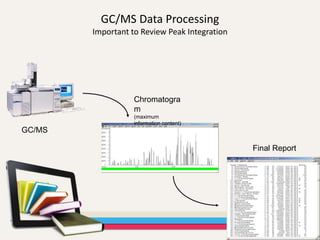
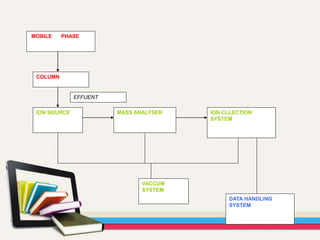
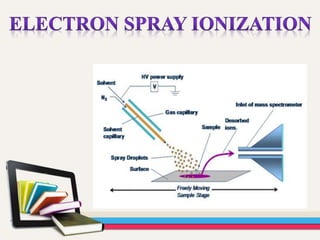
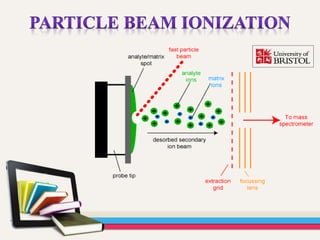
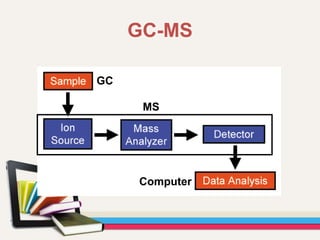
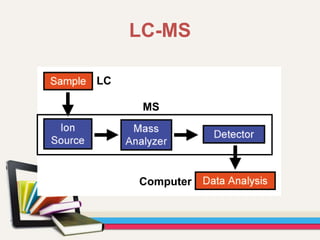
- Gas chromatography-mass spectrometry (GC-MS) is an analytical method that combines the features of gas-liquid chromatography and mass spectrometry to identify different substances within a test sample.
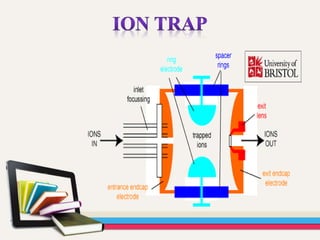
- GC is used to separate and analyze compounds that can be vaporized without decomposition, while MS is used to measure the mass-to-charge ratios of ions to determine the molecular mass and structure of molecules eluting from the GC.
- The combination of GC and MS provides a powerful tool for analyzing complex mixtures by separating compounds and identifying their chemical structures.







































































![1. Principles and Intrumentation of gas chromatography-mass
spectroscopy by W. M. A. Niessen, hyphen MassSpec
Counsultancy,Leiden,The Netherlands.
2. gas chromatography-mass spectroscopy from Wikipedia ,The free
encyclopedia.
3. Gas chromatography by U. A. Devkate Sir.
4. Encyclo pedia of Chromatography – Jack Cazes
5. Gas chromatography by Ian A[1]. Fowlis 2nd Edition
6. hollas_J.M._ modern _Spectroscopy
7. Handbook of Instrumental Technique for Analytical Chemistry by
Frank Settle.
8. Moderm Instrumentation Methods and Techniques by Francis Rouessac
and Annick Rouessac.
9. Instrumental methods of Analysis by Willard,Merritt,Dean,Settle,7th](https://image.slidesharecdn.com/chromatographygcmslcms-180812080122/85/Chromatography-gc-ms-amp-lc-ms-85-320.jpg)


