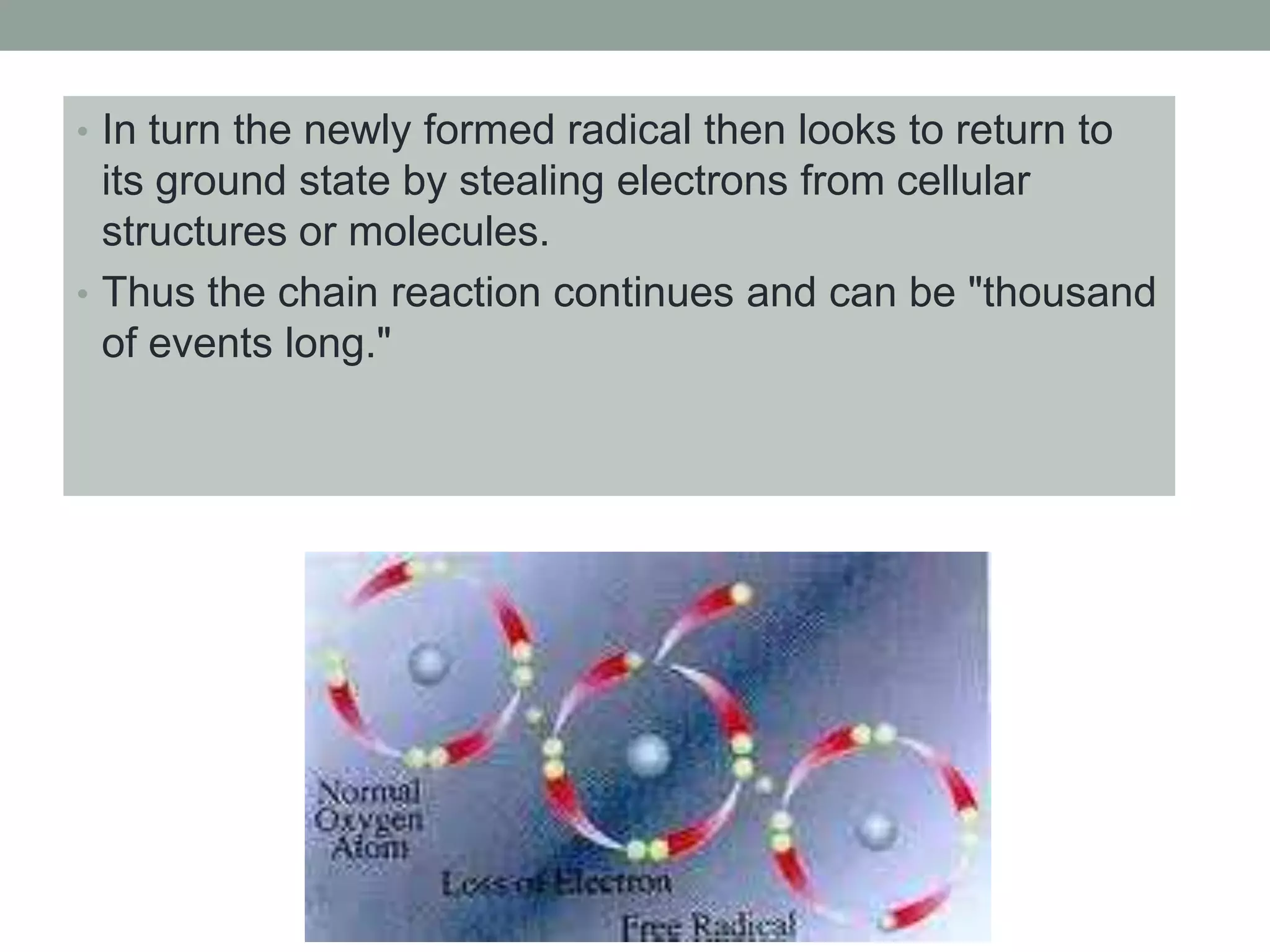
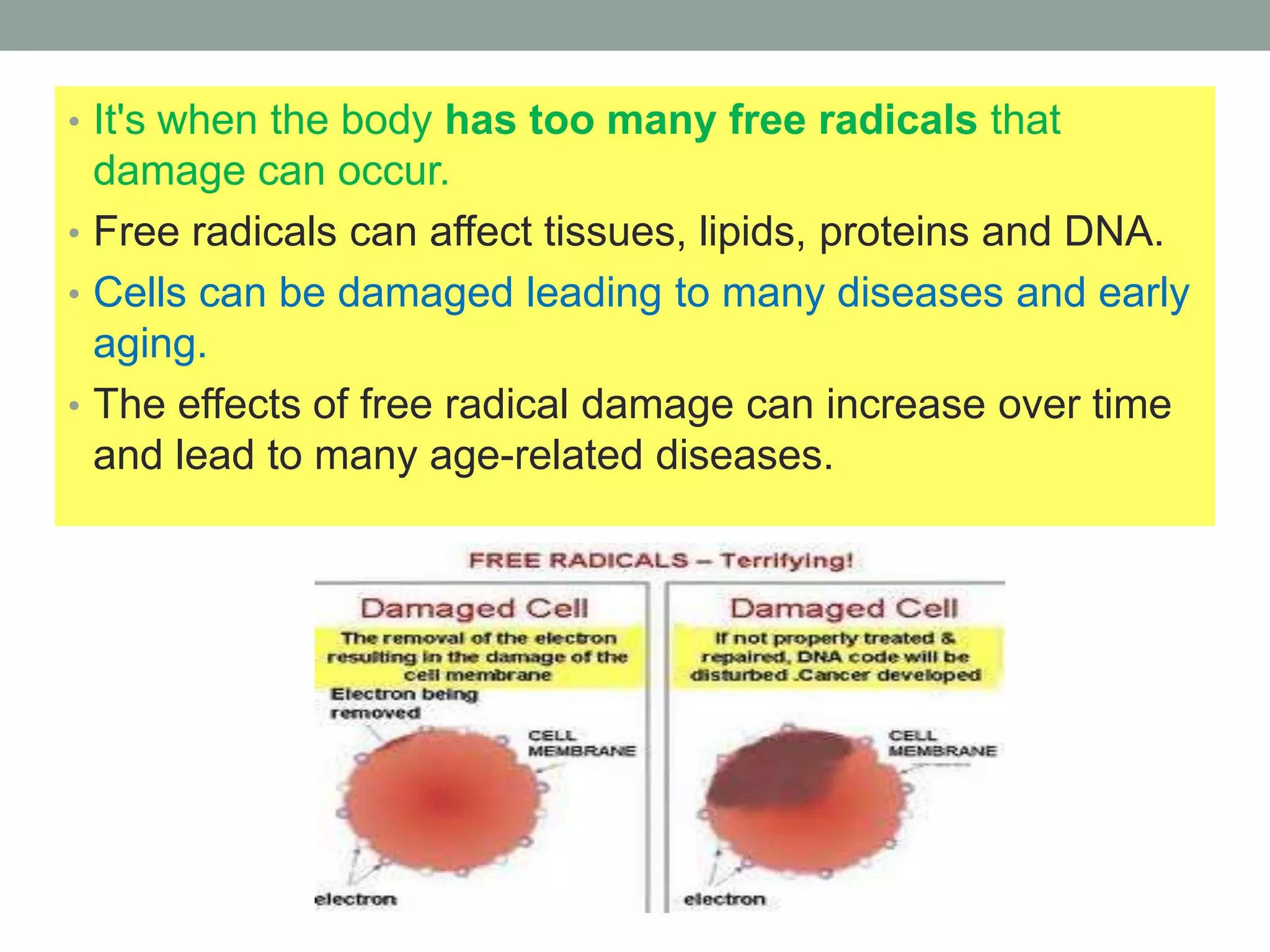
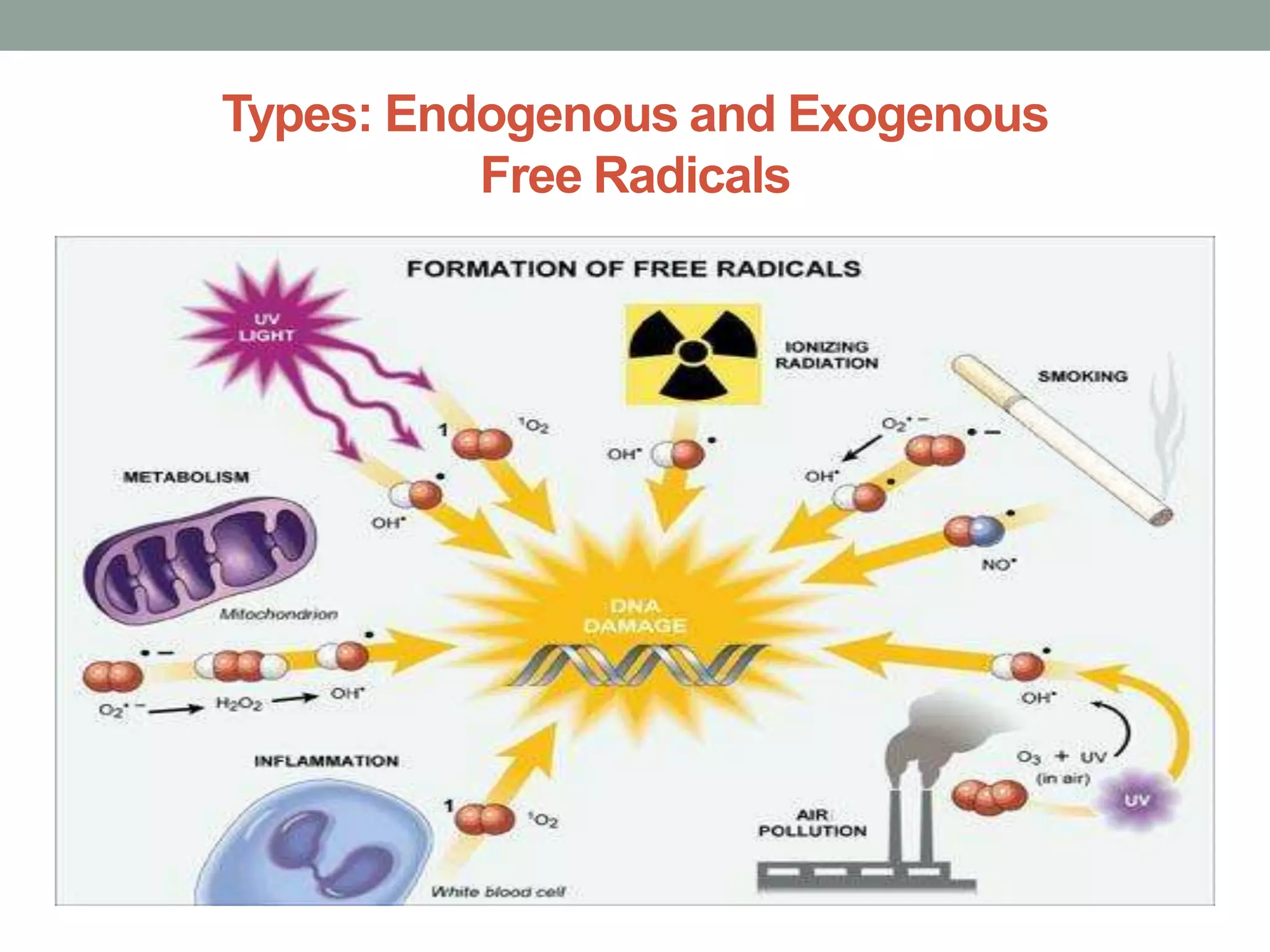
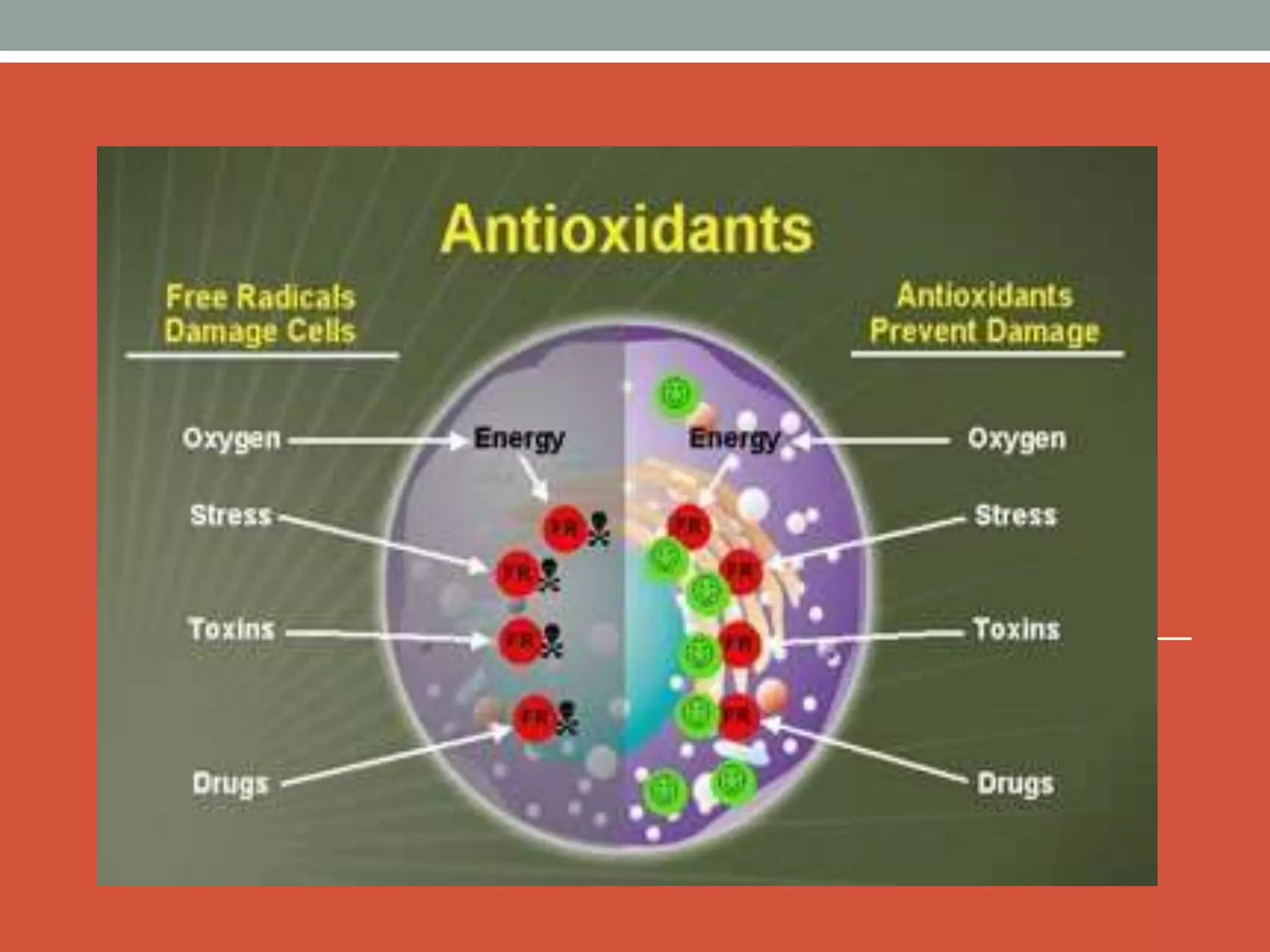
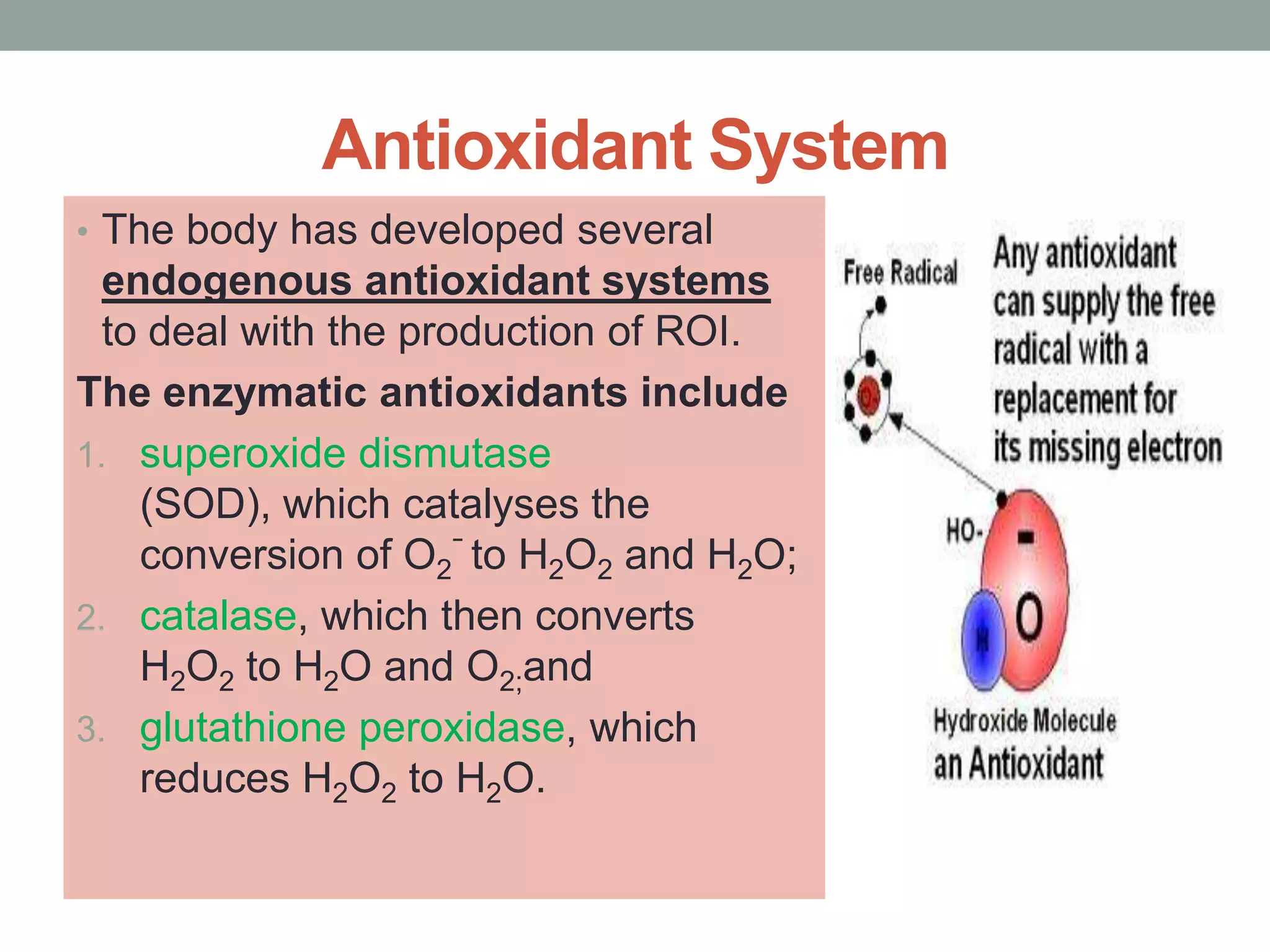
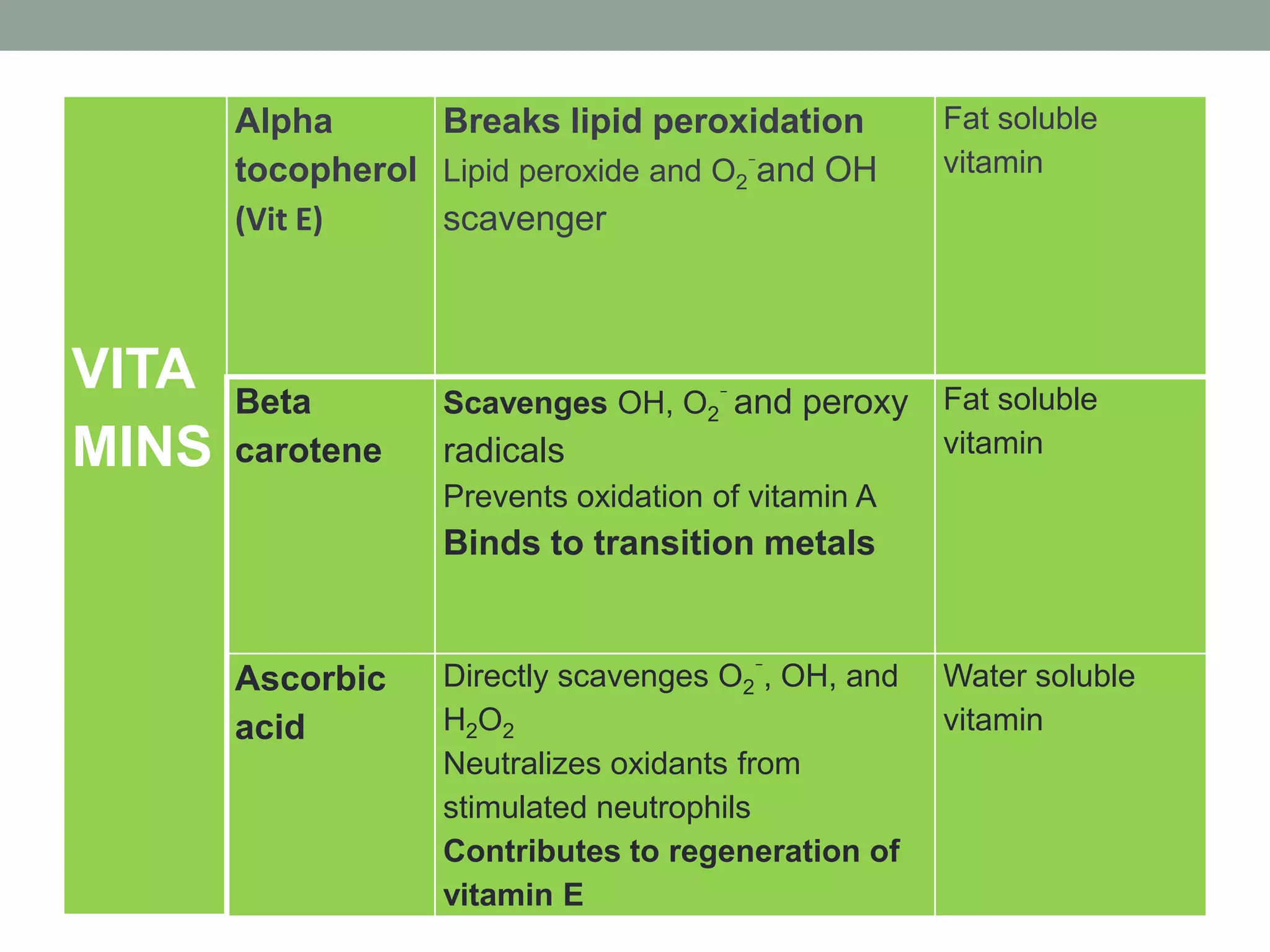
Free radicals are unstable molecules that can damage cells. They are formed through normal metabolic processes but also due to environmental toxins and radiation. The body has antioxidant defenses against free radicals but an excess can lead to oxidative stress and disease. Endogenous free radicals include reactive oxygen species like superoxide, hydrogen peroxide, and hydroxyl radicals produced during metabolism. Exogenous sources include tobacco smoke, drugs, radiation, and air pollution. Free radical damage accumulates with age and is linked to many age-related diseases.