This document discusses modifications made to allow a stationary diesel engine commonly used in developing countries to run on straight plant oils as a fuel substitute. The modification kit includes a preheating system and adjustments to the injector pressure and timing to improve atomization given plant oils' unique properties compared to diesel. Testing showed that with preheating of the high pressure fuel line and changes to injection parameters, the engine could efficiently utilize plant oils with performance similar to diesel, providing a potentially lower cost and sustainable fuel for remote rural areas.
![Compression Ignition Engine Modications for Straight Plant Oil
Fueling in Remote Contexts: Modication Design and Short-run
Testing
a,∗ b c a c,∗∗
M. Basinger , T. Reding , C. Williams , K. S. Lackner , V. Modi
a Earth and Environmental Engineering, Columbia University, New York City, USA
b Mechanical Engineering, Manhattan College, New York City, USA
c Mechanical Engineering, Columbia University, New York City, USA
Abstract
Though many plant oils have a similar energy density to fossil diesel fuel, several properties of plant
oils are considerably dierent from those of diesel. Engine modications can overcome some of these
dierences. An engine modication kit has been designed and tested for a slow speed, stationary,
indirect-injection diesel engine - the Lister-type CS 6/1, common throughout the developing world.
The kit allows waste vegetable oil fueling with similar performance to that of diesel fueling. The
kit's simple yet robust design is targeted for use as a development mechanism, allowing remote
farmers to use locally grown plant oils as a diesel substitute.
The modication kit includes a preheating system and the tuning of the injector pressure and
timing to better atomize given the unique properties of straight plant oils. The design methodology
for the modications is detailed and a suite of performance test results are described including fuel
consumption, eciency, pre-combustion chamber pressure, and various emissions. The results of
the study show how a combination of preheating the high pressure fuel line, advancing the injector
timing and increasing the injector valve opening pressure allows this engine to eciently utilize
plant oils as a diesel fuel substitute, potentially aiding remote rural farmers with a lower cost,
sustainable fuel source enabling important agro-processing mechanization in parts of the world
that needs it most.
Keywords: plant oils, straight vegetable oil, diesel engine, emissions, performance, CI engine
PACS: 88.20.ft
1. Introduction
The idea of fueling compression ignition engines on plant oil is as old as the diesel engine itself.
In Rudolph Diesel's preface to his 1912 patent he wrote that the use of vegetable oil for engine
fuel may seem insignicant today but such oil may become in the course of time, as important as
petroleum [1]. It seems that over the decades since this patent, whenever petroleum prices suddenly
increase a renewed interest in plant oil combustion occurs. This has resulted in a signicant body of
∗ Corresponding author
∗∗ Principal corresponding author
Email address: mlb2133@columbia.edu (M. Basinger)
Preprint submitted to Fuel October 15, 2009](https://image.slidesharecdn.com/basinger-fuel-article-1-120501102512-phpapp02/75/Compression-Ignition-Engine-Modifications-for-Straight-Vegetable-Oil-Fuel-1-2048.jpg)
![2
literature describing engine performance and resulting emissions of dierent oils in dierent engines.
Some studies have investigated modications to the engine that allow for straight fueling with plant
oil, others blend plant oils with diesel, while others carry out a combination of these approaches.
More recently, biodiesel production (transesterication) has become a popular endeavor. Even
so, biodiesel captures only a small fraction of the total diesel fuel market share in 2007 the U.S.'s
production of biodiesel was 1% the amount of fossil diesel sold [2]. Even if the entire world's
production of 115 billion liters of vegetable oil had been used for fuel in 2007, neglecting conversion
losses as well as the debate on the use of food materials for fuel, this would only satisfy about 3/4
of the U.S. diesel fuel demand [3].
However, some niche contexts do oer immediate opportunity. The technology development and
experimental results discussed in this paper are focused on the application of straight plant oils in
diesel engines in the developing country context. Many developing countries lack adequate energy
infrastructure. Modern fuels and generation systems are often inaccessible due to complex issues of
nancing, transportation, education/training, etc. The United Nations Development Programme
(UNDP) reported that worldwide in 2005, nearly 2.4 billion people used traditional biomass fuels
for cooking and nearly 1.6 billion people did not have access to electricity [4]. Mechanical power for
agriculture processing from small, stationary diesel engines is a core development mechanism for
rural populations. However, the cost and availability of fuel in these remote locations can prohibit
the use of this important energy resource.
The use of locally grown, non-edible, plant oils to fuel slow-speed diesel engines has potential
to provide a low cost, sustainable solution. The ruggedness of engines like the Listeroid CS and its
widespread availability lend itself to this application.
2. SVO Fueling Methodology
The combustion of plant oils in diesel engines is inuenced by qualities of both the oil and the
engine. Dierent studies have found dierent results and reached dierent conclusions depending
on the type of oil and engine that were used. Few experimental studies reach across engine type
and oil type to elucidate these trends [5, 6]. What follows is a brief review of the impact of the oil
and the impact of the engine on plant oil combustion.
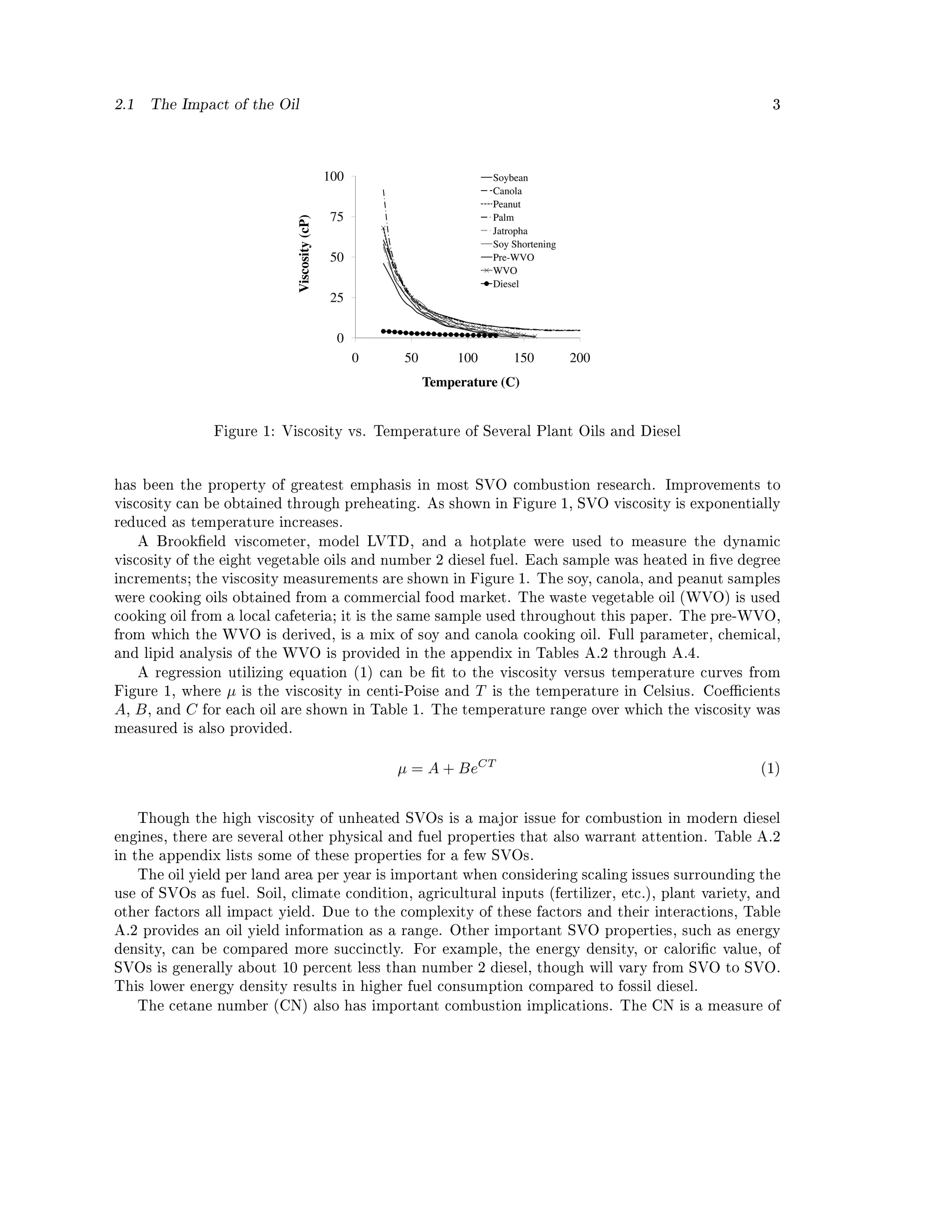
2.1. The Impact of the Oil
The processing of plant oils is complex. An entire industry surrounds the procedures and
practices involved in taking oil-crops from the eld to the food-stand, or fuel tank. Extremely
large, multivolume works, such as Baily's Industrial Oil Fats Products, detail every aspect of this
industry [7]. Some vegetable oil combustion studies have found that the degree or type of processing
of the oil has little impact on engine performance or emissions while others recommend at least
degumming in order to remove phosphatides [5, 6]. A detailed treatment of all the combustion
implications from the dierent degrees of processing of plant oils is beyond the scope of this paper.
The term straight vegetable oil (SVO) will hence forth be used to simply dierentiate plant
oil from biodiesel. Specic properties of dierent SVOs are discussed, in order to highlight broad
dierences from one type of plant oil to the next as they generally relate to combustion.
The two primary motivations behind transesterication are to remove the glycerin head of the
vegetable oil and to reduce the viscosity. The glycerin in SVOs has been shown to lead to engine
deposits in endurance testing [8, 9]. High viscosity can impede ow in the fuel lines and lter but is
of most concern with regard to its impact on atomization. For this reason the high viscosity of SVO](https://image.slidesharecdn.com/basinger-fuel-article-1-120501102512-phpapp02/75/Compression-Ignition-Engine-Modifications-for-Straight-Vegetable-Oil-Fuel-2-2048.jpg)
![2.1 The Impact of the Oil 4
Table 1: Viscosity-Temperature Coecients
A B C Correlation Coecient Temperature Range (C)
Soybean 1.110 109.6 -0.036 0.999 25 to 160
Canola 5.590 141.9 -0.038 0.998 25 to 200
Peanut 5.768 165.9 -0.041 0.998 25 to 200
Palm 3.265 327.2 -0.055 0.993 25 to 145
Jatropha 1.607 134.1 -0.037 0.999 25 to 145
Soy Shortening -3.638 89.30 -0.022 0.996 50 to 145
Pre-WVO 1.040 146.7 -0.039 0.998 25 to 145
WVO 3.486 171.1 -0.040 0.997 25 to 160
Diesel 1.015 5.058 -0.019 0.996 25 to 125
a fuel's ignition delay quality; a higher CN corresponds to a shorter ignition delay. Long ignition
delay is undesirable due to the consequences of engine knock. A CN between 40 and 60 is preferable
[10]. Some negative aspects of an SVO's ignition quality can be lessened or avoided through tuning
the engine's injection timing for the particular fuel.
Vegetable oils are hydrocarbons, though much heavier, and less volatile than number two diesel
[11]. The chemical composition of SVOs is important when considering combustion implications.
Table A.3 in the appendix shows the C:H:O:S:N ratio for several SVOs. The occurrence of oxygen
in the SVO molecule is advantageous, enhancing mixing-limited combustion and even reducing
particulate emissions, though potentially increasing NOx [12, 13]. The existence of nitrogen and
oxygen in SVOs has valuable lubricity benets [14].
The ratio of an SVO's C:H:O:S:N is meaningful, but the bond conguration of these elements, the
lipid prole, is also important to consider. Dierent fatty acid chains occur in signicantly dierent
amounts for dierent oils. This lipid prole has implications for combustion. Many investigations
have shown high amounts of unsaturated fatty acid chains, especially linolenic and linoleic chains,
increase engine wear as a result of the polymerizing quality of the heavy polyunsaturated lipids
[15, 9, 6]. There is a trade-o however. The degree of saturation of an oil impacts cold ow
properties. Soybean oil may have more polymerizing qualities compared to palm oil, but at low
temperatures, such as those often found in temperate climates during winter months, the cloud
point and pour point of palm oil can prohibit its use as a non-blended, non-preheated fuel. The
lipid prole has also been shown to directly inuence ignition delay and in turn NOx and particulate
emissions [16].
Ryan and Bagby found that it is not only an SVO's physical properties (viscosity), but its
chemistry can also impact atomization characteristics. Polyunsaturated lipids such as linolenic and
linoleic chains were most aected during the injection process, resulting in unexpected injector
spray characteristics [17].
Structural indices such as the saponication value (SV) and iodine value (IV) are used to quickly
describe constituent lipid qualities. The SV is a measure of the average molecular weight, or chain
length, of the fatty acids present in an oil. The IV describes the unsaturated quality, the amount
of double bonds, of an oil. An IV is assigned to an SVO based on the amount of iodine that can
be absorbed by the double bonds. The IV neglects the dierentiation between polyunsaturated
acids and monounsaturated acids. Knothe describes other, less common indices that can be used
to overcome the limitations of the IV [18]. For the time being however, the widespread use of the](https://image.slidesharecdn.com/basinger-fuel-article-1-120501102512-phpapp02/75/Compression-Ignition-Engine-Modifications-for-Straight-Vegetable-Oil-Fuel-4-2048.jpg)
![2.2 The Impact of the Engine 5
IV means that it is often readily available across studies, making it a useful index. Table A.4 in
the appendix provides the lipid proles and iodine values for several SVOs.
2.2. The Impact of the Engine
Several SVO engine study reviews highlight the role of the engine type and conguration in
inuencing combustion; depending on the engine and modication used in a study, results can dier
signicantly [8, 9, 10, 11, 19, 20, 21]. Engine speed and loading has been found to be important.
Vojtisek-Lom, Blazek, et al. investigated rapeseed SVO fueling and found at higher engine loads
CO levels improved while NOx worsened compared to diesel, while at low engine loads HC, CO,
and PM worsened [22]. In addition to load, the design of specic components also impacts SVO
combustion. Piston conguration and injector design are inuential, but most often an emphasis is
placed on the type of injection system, direct or indirect.
Modern diesel engines tend to have direct injection (DI) systems due to the improved eciency
and emissions it can oer. Older designs, sometimes relics from days with less stringent fuel quality
standards, utilized indirect injection (IDI) systems because of its ability to robustly burn lower
quality fuel. The general trend in SVO combustion studies seems to conrm this value of IDI
systems. Hemerlein, Korte et al. showed that DI engines fueled on rapeseed SVO tended to have
poorer emissions and were not suitable for direct fueling, while IDI engines with big cylinders were
suitable [5].
Ryan, Dodge, et al. tested multiple SVOs in a DI and IDI engine and found increased nozzle
coking and lubrication contamination compared to diesel in the DI engine but not in the IDI engine,
though the specic energy consumption of the DI engine was generally better then the IDI engine
[6]. However, Engler, Johnson, et al. found that though short run IDI engine tests were favorable
for various degummed SVOs, longer 40 hour tests revealed rapid fouling of the lubrication oil [23].
Suda showed that in some DI engines unburned fuel impingement on the cylinder wall results in
deposits, ring and cylinder wear, and lubrication oil contamination [24]. Suda also tested soybean
SVO in an IDI with a pre-combustion chamber and designed a special heat plug to allow the engine
to robustly burn the SVO.
Redesigning major engine components is often less desirable than more minor modications. The
most common minor engine modication is to preheat the SVO. Dierent studies have made dierent
conclusions with regard to just how much to preheat. Bari, Yu, et al. found heating between 55 and
70 degrees Celsius was adequate for reducing lter clogging and improving engine performance and
emissions characteristics [25, 26]. Pugazhvadivu and Jeyachandran found preheating to 135 degrees
Celsius to be preferable [27]. Nwafor investigated the impact of preheating rapeseed SVO to 70
degrees Celsius and found at low speed and partial loading it was benecial, but at higher speeds
and loads it had less impact [28]. Analysis of properties of various SVOs has shown temperatures
between 200-300°C to result in thermal decomposition, while higher temperatures approach the
ash point [29]. Even lower temperatures have been argued to result in overheating the SVO. Bari,
Lim, et al. noted that at 100 degrees Celsius vapor bubbles occurred in the fuel line, resulting in
non-ideal combustion [30]. Suda found that at 90 degrees Celsius oxidation can occur, resulting in
gum formation [24].
Another common minor modication is to increase the injector valve opening pressure (IVOP).
Increasing a diesel engine's IVOP has been shown to decrease fuel spray droplet diameter, and
increase velocity and penetration distance resulting in a host of engine performance and emissions
improvements [31, 32, 33, 34, 35]. Initial average fuel spray droplet diameter has been shown to be
inversely related to its velocity squared [36]. This means that as velocity increases (from increased](https://image.slidesharecdn.com/basinger-fuel-article-1-120501102512-phpapp02/75/Compression-Ignition-Engine-Modifications-for-Straight-Vegetable-Oil-Fuel-5-2048.jpg)
![6
IVOP), droplet size rapidly decreases. Droplet evaporation can be described by the D2 law, which
relates evaporation rate to the droplet diameter squared [37]. Decreasing the droplet diameter can
then signicantly increase evaporation rates, thus enhancing combustion. This dual exponential
relationship between droplet evaporation time and diameter, and between diameter and velocity
means that even relatively small increases to the IVOP can have signicant combustion advantages.
It has already been discussed how SVOs are more viscous and heavier then number 2 diesel fuel;
this has been shown to result in considerably larger droplet diameters and lower injection velocities,
as described by lower Weber numbers [38, 39]. For these reasons SVOs in particular can benet
from increased IVOP. Enoki, Hayashi, et al. noted an improvement in brake thermal eciency,
ignition, and combustion stability with increased IVOP in an IDI engine fueled on various SVOs
[33]. Puhan, Jegan, et al. also found improved engine performance and emissions from increased
IVOP in a DI engine fueled on linseed methyle esters [40].
There is a point where increasing the IVOP becomes counterproductive. This is due to increased
spray penetration resulting in wall impingement [33]. For this reason it can be valuable to tune
a particular engine's IVOP for the specic SVO.
Injection timing is another minor modication that has been employed to help the performance
and emissions of SVO fueled diesel engines. Haldar, Gosh, et al. observed enhanced engine perfor-
mance with advanced timing, they attributed this to the lower cetane number of SVOs [41]. Bari,
Yu, et al. investigated timing eects on a WVO in a DI engine. They found advanced timing im-
proved eciency and reduced CO emissions, though it elevated NOx emissions [26]. Nwafor, Rice,
et al. also found benets from advancing the timing of a rapeseed SVO fueled IDI engine. The
engine ran smoother and both CO and CO2 emissions improved [42]. This was attributed to the
longer ignition delay and slower burning rates of plant oils. However, delay period was also found
to be inuenced by engine load, speed and temperature. Similar to IVOP ndings, advancing the
timing too far can have negative consequences, resulting in erratic engine behavior [42].
3. Experimental Setup
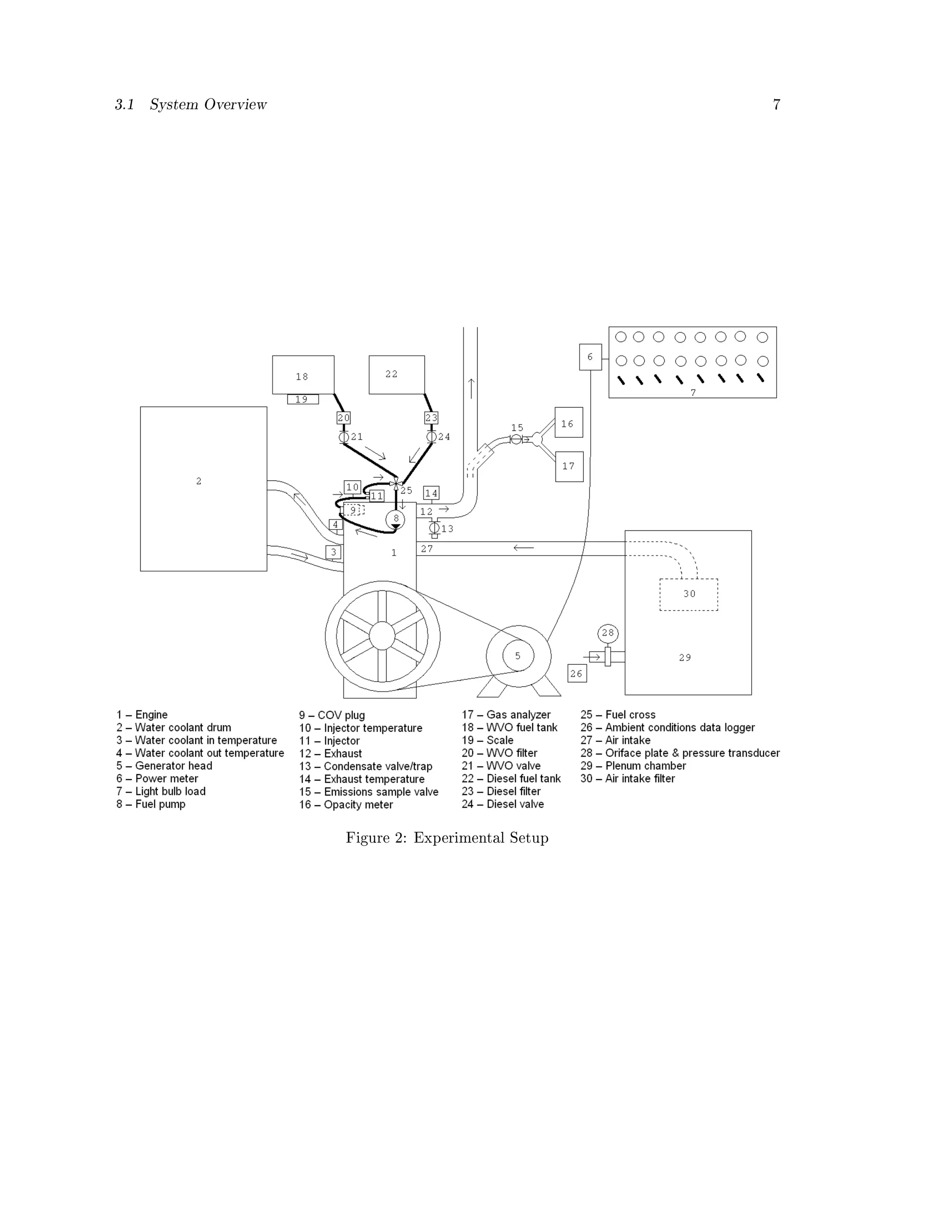
As shown in Figure 2 a slow speed stationary engine common to remote rural developing country
settings was fueled with waste vegetable oil (WVO). A modication kit was developed and tested.
3.1. System Overview
Listeroid engines are used throughout developing countries for agro-processing. These engines
are typically 6 to 16 horsepower, vertical, stationary, water cooled with large ywheels. These
engines weigh more than 300 kilograms. For this study a Listeroid CS (cold start) 6/1, 650 RPM,
4-stroke, 114.3 mm x 139.7 mm bore/stroke, water cooled, IDI diesel engine was used to drive
an ST-5 5kW generator head which was loaded by a bank of light bulbs. The ST-5 was chosen to
provide the load to the engine because it is a generator commonly paired with Listeroids throughout
the developing world. Engine load was measured from a power meter which logged volts, amps,
frequency, and power factor. Engine speed was measured with a Hall Eect sensor and a magnet
on the ywheel.
The engine coolant system was comprised of a 55 gallon drum lled with water, circulating via
a passive thermal siphon cycle. Two type J thermocouples were used to measure water coolant
level entering and exiting the engine. A thermostat was used in the engine coolant exit to speed up
the rate at which the engine reached steady state. Steady state was dened as the point where the](https://image.slidesharecdn.com/basinger-fuel-article-1-120501102512-phpapp02/75/Compression-Ignition-Engine-Modifications-for-Straight-Vegetable-Oil-Fuel-6-2048.jpg)
![8
water coolant temperature leaving the engine was stable and consistent; this occurred after about
90 degrees Celsius.
The air intake ow rate was measured via an orice plate pressure transducer. A plenum
chamber was utilized to attenuate the air ow pulses, sized per SAE standards [43]. Ambient
temperature, pressure, and relative humidity were measured near the plenum chamber entrance.
A dual fuel tank approach was utilized the engine was started on diesel to allow the preheater
to come up to temperature and shutdown on diesel so that the high pressure fuel lines and pump
were purged of the WVO. Each fuel tank had its own lter. The WVO was pre-ltered to 1-micron
before being added to the fuel tank.
The WVO's on-engine lter (post fuel tank) was 80 mesh, to reduce a pressure drop that could
result in starving the engine of fuel. To minimize mixing between the two fuels, the injector's fuel
return line was not routed back to either fuel tank, but instead directly to the juncture of the two
fuel lines (SVO and diesel) situated just before the fuel pump. This point is labeled as the fuel
cross in Figure 2.
The opacity of the exhaust was measured using an AutoLogic model # 310-0332 opacity meter
to take readings at 16 Hz that were then averaged across a 10 minute sampling window. To measure
gaseous emissions, an Enerac 700 integrated emissions system was used to measure O2, CO, CO2,
unburned hydrocarbons (UHCs), NO, NO2, and SO2. Data was collected once per second as parts
per million (ppm) or vol%, depending on the concentration. Readings were averaged across a ten
minute steady state window (steady state referring to both engine stabilization and the stabilization
of the gas analyzer measurements).
4. Preheater Design
In order to lower the viscosity of the WVO to a level comparable to diesel, a preheater was
designed to take advantage of the Listeroid CS's special characteristics. The original design of the
engine included a Change Over Valve (COV) for adjusting the compression ratio for easier start-
up in cold climates. This original design is shown in Figure 3. As this engine is now manufactured
and used in primarily warm climates, the COV has been replaced by a COV plug. This COV plug's
direct access to a large amount of waste heat through the cooling jacket, nearby location to the
injector, and easy removal for use in a modication kit, made it an appealing candidate for use as
a preheater.
Various methods of using the COV plug as a preheater were explored. A priority was placed
on a design that could be easily manufactured in a basic machine shop making it appropriate for
deployment and servicing in a developing country context. The nal design consisted of machining
a V-shaped passageway to route the fuel through as shown in Figure 4. The inlet and outlet
were tapped to allow compression ttings to be attached so that the high pressure line could be
connected as shown in Figure 2.
More complex geometries could have been utilized to further aid heat transfer, but the V-shape
channel was chosen as it was believed to possess an optimal ease and cost of local manufacturability
for the target context. The passageway diameter was chosen to be 0.635 cm (0.25 in). It was
assumed that any larger diameter would potentially adversely aect the injection timing or risked
overexerting the fuel pump (due to the increased volume of fuel and its compressibility under high
pressure). The length of the passageway was chosen to be 11.43 cm (4.5 in); a longer length would
have been within 1.3 cm (0.5 in) of the pre-combustion chamber leaving a thickness of material that
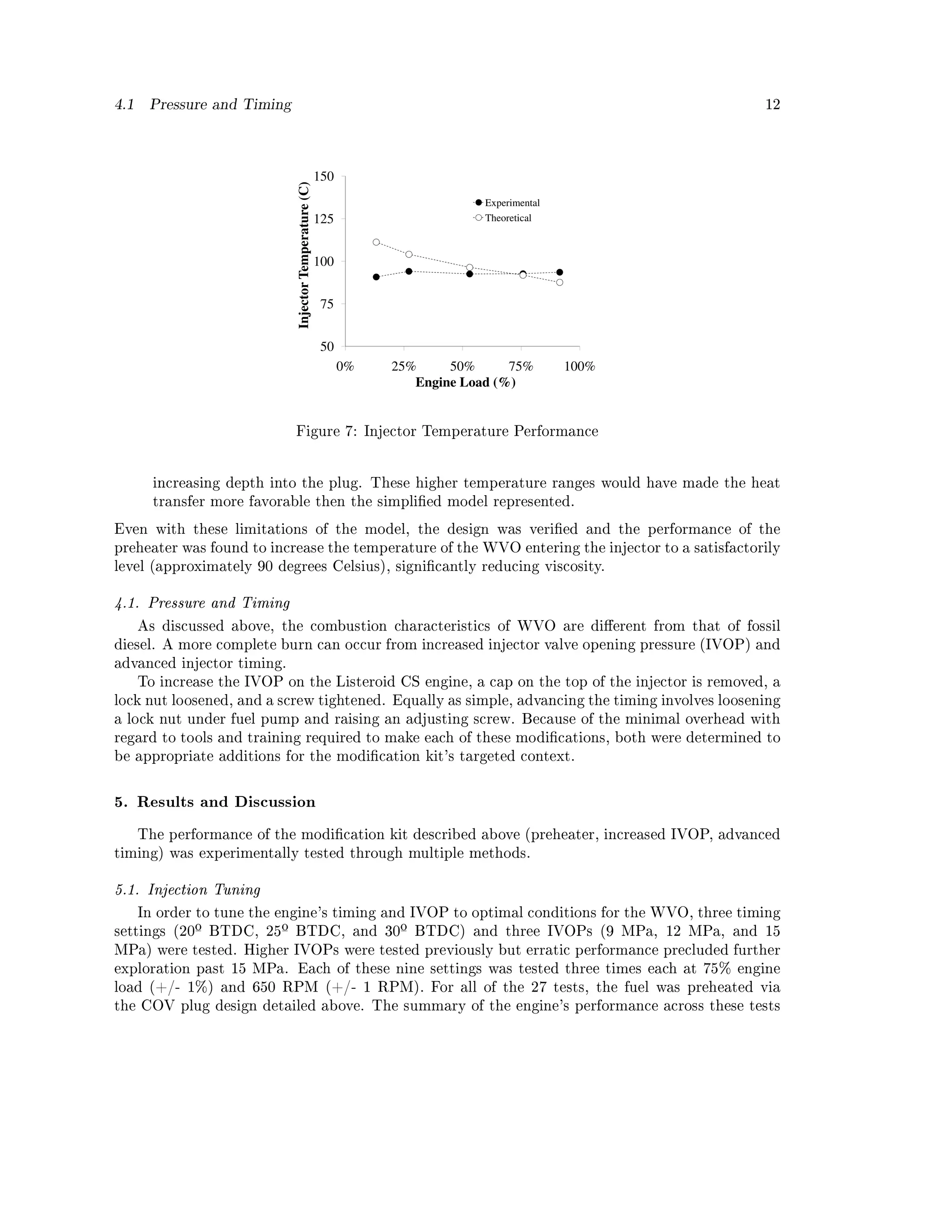
might be susceptible to failure. A simplied heat transfer model aided the design of the geometry,](https://image.slidesharecdn.com/basinger-fuel-article-1-120501102512-phpapp02/75/Compression-Ignition-Engine-Modifications-for-Straight-Vegetable-Oil-Fuel-8-2048.jpg)
![9
Figure 3: Original Lister CS Head and COV[44]
Figure 4: COV Plug Modication](https://image.slidesharecdn.com/basinger-fuel-article-1-120501102512-phpapp02/75/Compression-Ignition-Engine-Modifications-for-Straight-Vegetable-Oil-Fuel-9-2048.jpg)
![10
Figure 5: Heat Transfer Model
showing that the fuel would not be over or under heated. The performance was then conrmed
experimentally.
Before building the preheater, to estimate the performance of the V-shaped passageway was
modeled as a 1-D heat transfer problem. The modeled passageway was a straight tube with diameter
D and length L with the same values as the V-shaped diameter and length. The mass ow rate
of the fuel ow through the passage way was designated m,
˙ with a bulk temperature T b, and a wall
temperature T w, as shown in Figure 5. This simplication allowed the utilization of well known
empirical correlations for determining the heat transfer coecients.
The bulk temperature of the fuel (T b ) was dened by equation (2):
Tf + Ti
Tb = (2)
2
Where Ti was the initial temperature of the fuel entering the passageway and Tf was the nal
temperature leaving the passageway. In a convection dominated system, the overall power utilized
to raise the temperature of the fuel can be calculated by equation (3):
q = mcp (Tf − Ti ) = hA (Tw − Tb )
˙ ˙ (3)
where cp is the specic heat of the fuel and is a function of T b. A is the surface area in the
tube. h is the convection coecient and is a function of the Nusselt number (N u). For laminar,
fully developed ow in a horizontal tube, an empirical relation has been developed and is expressed
in equation (4) [45, 46]
hD 1/5 1.8
Nu = = 0.61 (ReRa) 1+ 1/5
(4)
k (ReRa)
Where Re and Ra were the Reynolds Number and Raleigh Number, respectively, and dened
in equations (5) and (6).
UD
Re = (5)
ν
gβ (Tw − Tb ) D3
Ra = (6)
να
where U was the average velocity of the fuel, ν was the kinematic viscosity of the fuel, g was the
gravitational acceleration, β was the volumetric thermal expansion coecient of the fuel (assumed
to be constant), and α was the thermal diusivity of the fuel. ν and α were functions of T b . For a
given T w and T i , T f can be iteratively solved for through utilizing equations (2) through (6). In](https://image.slidesharecdn.com/basinger-fuel-article-1-120501102512-phpapp02/75/Compression-Ignition-Engine-Modifications-for-Straight-Vegetable-Oil-Fuel-10-2048.jpg)
![11
1.3cm depth
160
3.8cm depth
Temperature (C)
6.4cm depth
140
120
100
0% 25% 50% 75% 100%
Engine Load
Figure 6: COV Temperature Prole
order to nd appropriate values of Tw a thermal prole of the COV plug was measured through
the following experimental setup.
Five thermocouples were installed into the COV plug in an X conguration, rst at a depth
of 1.3 cm (0.5 in) into the plug from the exterior of the engine, then 3.8 cm (1.5 in), and nally 6.4
cm (2.5 in). These depths extended across 80% of the total COV plug length. The ve measured
temperatures were averaged at each depth for each load tested and this thermal prole of the plug
is shown in Figure 6. Error bars indicate standard deviation between the 5 thermocouples, averaged
across the ve loads at the specic depth.
For purposes of a simple theoretical calculation of Tf the wall temperature of the passageway
(Tw ) was assumed to be uniform across all depths, but vary depending on the load, so the average
temperature across all depths was used as the Tw for each load.
To justify the use of equations (2) and (3) the role of convection relative to conduction needed
to be determined so the Rayleigh Number (Ra) of the waste vegetable oil traveling through the
passageway was calculated using the measured Tw . The Rayleigh number exceeded 50,000 for all
engine loads (10%-90%), validating the assumption that convection dominated [45].
Tf was found through iteratively solving equations (2) through (6). This theoretical Tf was
compared to the experimentally measured temperature at the injector utilizing the actual geometry.
Figure 7 shows the agreement between the calculated theoretical and the experimentally measured.
The dierences between the calculated and measured results are likely due to the following.
1. The simplifying assumption of laminar ow is inaccurate. The fuel pump pushes fuel through
the preheater in pulses which likely enhance heat transfer.
2. The experimental measurement of the fuel temperature was not at the exit of the preheater
(Tf ), but instead approximately 15 cm (6 in) downstream at the inlet to the injector. Though
this 15 cm (6 in) of fuel line was insulated, some temperature loss would have occurred,
meaning the experimental values in Figure 7 are lower than the actual Tf .
3. The wall temperature of the passageway (Tw ) was assumed to be uniform and only vary
with load. But the measured COV plug thermal prole showed increasing temperature with](https://image.slidesharecdn.com/basinger-fuel-article-1-120501102512-phpapp02/75/Compression-Ignition-Engine-Modifications-for-Straight-Vegetable-Oil-Fuel-11-2048.jpg)
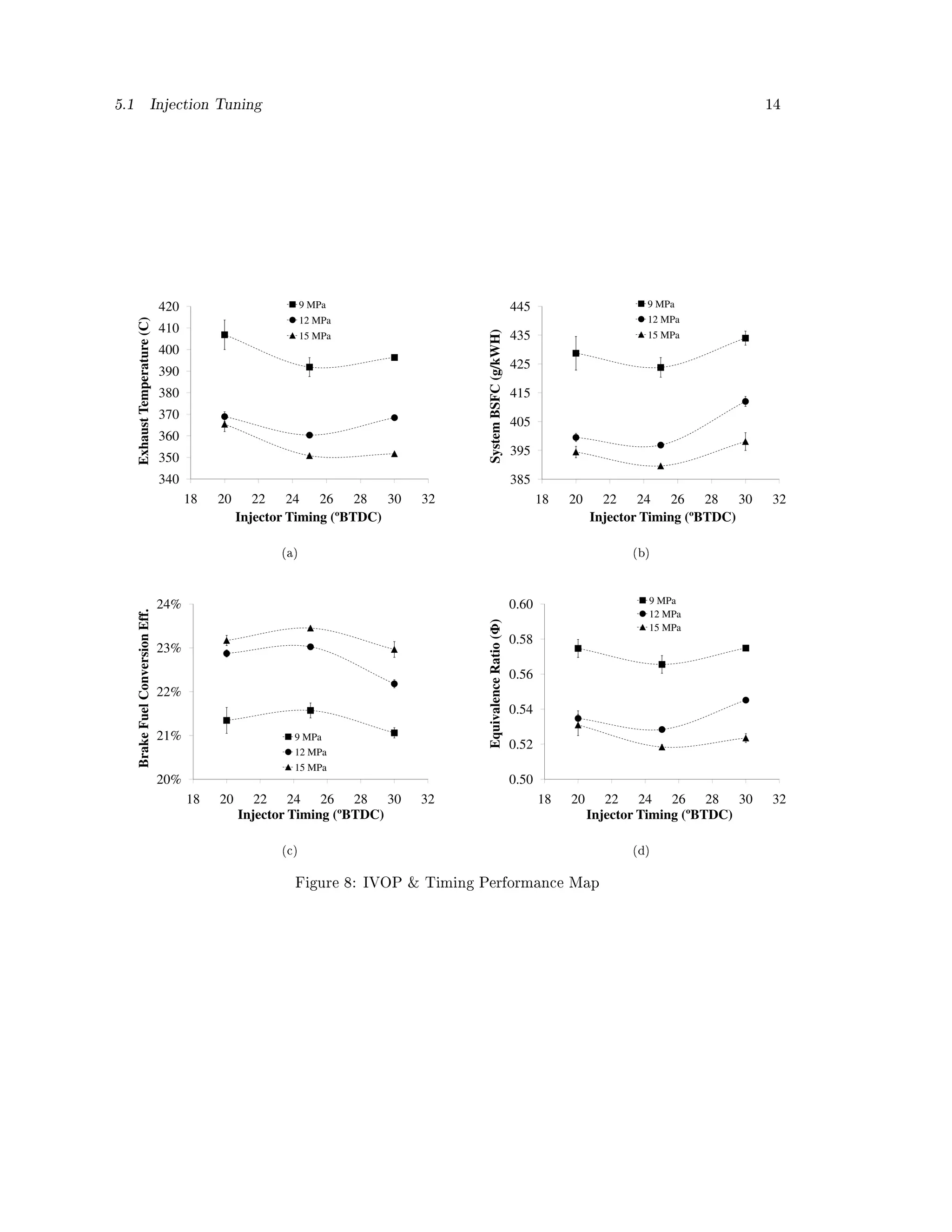
![5.1 Injection Tuning 13
is detailed below. Error bars signify standard error across the three repetitions, for each of the
9 dierent settings. Figure 8 shows various engine performance parameters. All four parameters
point toward an optimized tuning of the injector timing at 25º BTDC with an IVOP of 15 MPa.
The temperature of the middle of the exhaust stream was measured immediately o the engine
head with a type-J thermocouple. The results are shown in Figure 8 (a). A clear dierence
in temperature occurs primarily from increasing the IVOP. As the timing advances the exhaust
temperature is lowered to a point, and then increases again. The lowered temperature is likely
indicative of more complete combustion, whereas the hotter temperatures are likely due to less
complete combustion that continued late into the cycle [27]. These results follow the same trend
as the ndings from Bari et. al. where advanced timing of a DI engine fueled on WVO resulted in
lower exhaust temperature, due to combustion occurring earlier, thus allowing the burnt gas more
time to cool [26].
Figure 8 (b) shows the brake specic fuel consumption for the system (generator and electrical
losses were not subtracted). With a trend nearly identical to the exhaust temperature, the best
performance was found at the timing of 25º BTDC with an IVOP of 15 MPa. These results relate
well to the ndings of Nwafor et. al., where advanced timing of an IDI engine resulted in lower
BSFC at low engine speeds [42].
Given WVO's measured higher heating value of 39.4 MJ/kg, the brake fuel conversion eciency
was calculated (the generator and electrical system was assumed 82.5% ecient). Figure 8 (c) shows
how tuning the IVOP and timing can result in a gain of 2% eciency. Such a gain is signicant
when considering the modest eciency of the engine. This trend toward improved eciency at
advanced timing is similar to Haldar's ndings [41].
The equivalence ratio is the actual fuel to air ratio divided by the stoichiometric fuel to air ratio
and is a direct indicator of the quality of combustion. Figure 8 (d) shows the equivalence ratio
across the nine test points. In strong agreement with all other engine performance parameters, the
equivalence ratio map reinforces the hypothesis that when fueling the Listeroid CS 6/1 on WVO,
optimal tuning occurs at the timing of 25º BTDC with an IVOP of 15 MPa.
These engine performance benets that were gained by advancing the timing and increasing
the IVOP can likely be attributed to the specic atomization and ignition qualities of the WVO.
As discussed earlier, plant oils have poorer atomization qualities due to their specic physical and
chemical properties. The Cetane Number (CN) of the WVO used in the experiments, as shown
in Table A.2, is low compared to fossil diesel, meaning that ignition is delayed. Advancing the
injection timing then helps to overcome this high ignition delay. Similarly, increased injection valve
opening pressure has been shown to reduce the droplet size, decreasing the burn time and leading
to more complete combustion (as described above). This more complete combustion is also seen in
the emissions.
The measured opacity values are reported in Figure 9. The results show that at the most
advanced injector timing (30° BTDC), regardless of IVOP, opacity is worse than at stock timing
(20° BTDC). However, in agreement with the engine performance data from Figure 8, increasing
the IVOP improves the opacity. This vividly shows an engine tuning envelope eect - advanced
timing and increased IVOP improves combustion, but only to a point, where it then begins to
degrade combustion. When injection is advanced too far cylinder conditions are not optimal for
good atomization. Temperatures and pressures rise rapidly close to TDC. These conditions are
important for appropriate mixing and vaporization which lead to good combustion. Advancing the
injection too much introduces the fuel spray into the cylinder before these conditions are available.
The measured gaseous emissions are reported in Figure 10. For both the carbon monoxide and](https://image.slidesharecdn.com/basinger-fuel-article-1-120501102512-phpapp02/75/Compression-Ignition-Engine-Modifications-for-Straight-Vegetable-Oil-Fuel-13-2048.jpg)
![5.1 Injection Tuning 15
12
9 MPa
12 MPa
10
15 MPa
8
Opacity (%)
6
4
2
0
18 20 22 24 26 28 30 32
Injector Timing (ºBTDC)
Figure 9: IVOP Timing Exhaust Opacity Map
the oxides of nitrogen, the 12 MPa IVOP results so closely resembled the 15 MPa IVOP results
that they have not been shown in Figure 10 (a) and (b). Instead, the low and high IVOP setting
are shown to illustrate the envelope.
At the lower IVOP more CO is generated, but less NOx. (NOx is reported as the sum of
measured NO + NO2.) This common CO - NOx trade o (also described as a PM-NOx trade o )
points again to the quality of the combustion. As the IVOP is increased, atomization improves. In
diesel engine combustion, carbon monoxide production tends to be low under fuel lean conditions
(equivalence ratio less than 1). During the combustion process carbon monoxide is produced, but
with adequate oxidant, mixing, and at necessary temperatures much of it is oxidized to carbon
dioxide. At the highest IVOP (15 MPa), as timing is advanced CO rapidly increases. This may
be explained by the poorer mixing and oxidation conditions that occur at the more advanced
timing the fuel spray is encountering lower pressures and temperatures and because the IVOP is
higher, more of the spray encounters these conditions (the increased IVOP results in faster injection:
reduced injection period).
The NOx values between the dierent IVOPs are not dramatically dierent (the error bars
in Figure 10 (b) between the two IVOPs overlap at low and mid timing). Increasing the IVOP
from 9 to 15 MPa does not then drastically increase NOx. Timing on the other hand does seem
to noticeably aect NOx. The envelope is not as sharp, NOx somewhat levels o as timing is
increased past 25° BTDC. NOx formation is commonly attributed to the Zeldovich mechanism (a
thermal mechanism). With regard to advanced injection timing, Patterson and Henien describe
NO formation increasing in two ways [47]. As timing is advanced ignition delay increases but less
so than the actual advancement (in terms of crank angle), resulting in earlier autoignition. Higher
NO formation is then related to the longer ignition delay as it allows for more fuel evaporation and
mixing in the lean ame region of the spray. But in other spray regions NO may also increase
due to higher temperatures. This description seems applicable to what is observed in Figure 10 (b).
Bari et. al. also found NOx to increase with advanced timing when fueled on WVO [26].
Figure 10 (c) and (d) show the measured UHC and SO2 concentrations, respectively. Both
cases support the same trend observed throughout all of the other IVOP / timing maps. Unburned
hydrocarbon emissions appear to stay relatively level at advanced timing likely due to the better](https://image.slidesharecdn.com/basinger-fuel-article-1-120501102512-phpapp02/75/Compression-Ignition-Engine-Modifications-for-Straight-Vegetable-Oil-Fuel-15-2048.jpg)
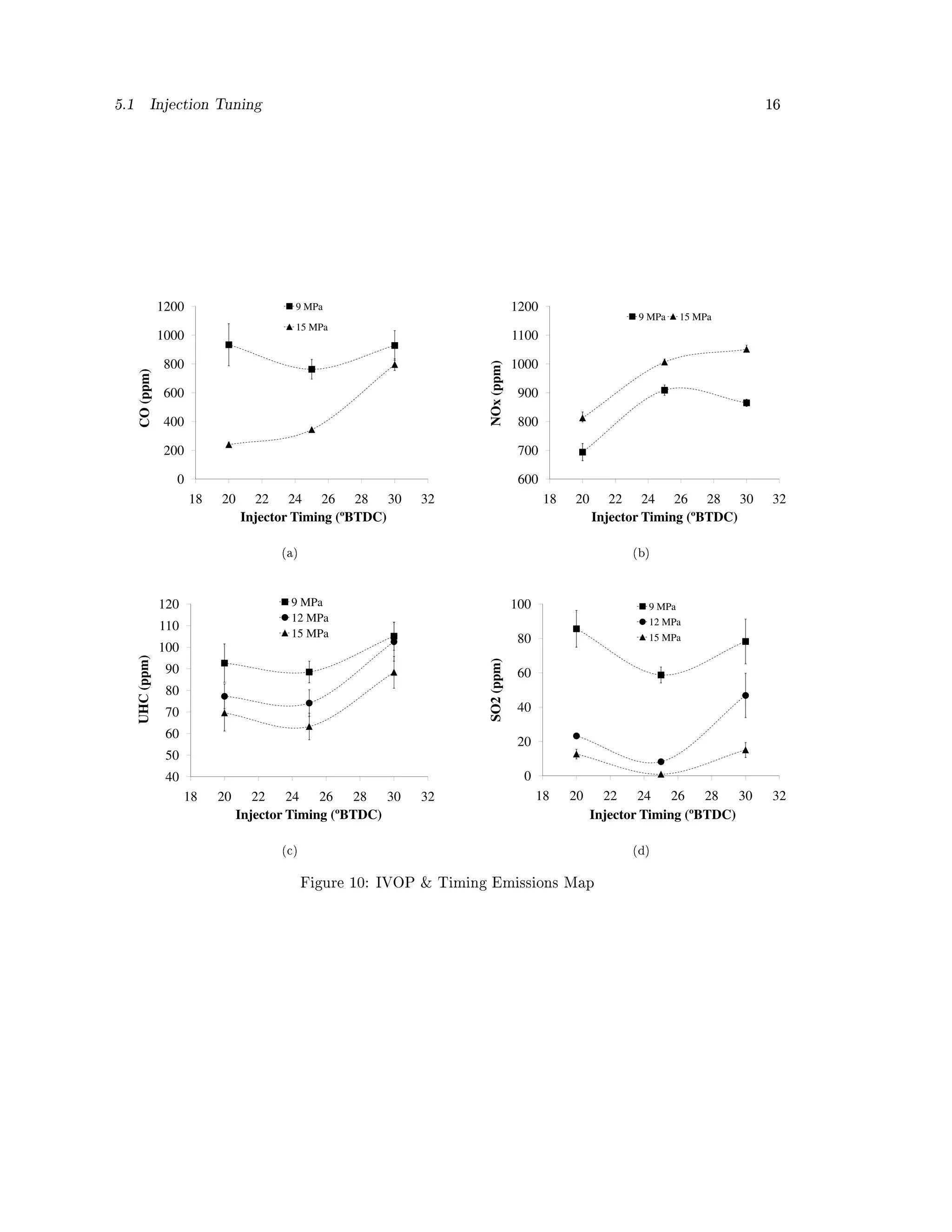
![5.2 Pre-combustion Pressure 17
matching of the ignition delay to timing, but then UHC more rapidly increase as timing is further
advanced past 25° BTDC. This may be because as timing continues to be advanced the spray is
introduced into lower pressure and temperature conditions which results in less vaporization and
larger droplets which may not burn as completely or may potentially impinge on the walls.
In the case of SO2, emissions became undetectable at the optimized tuning of 25º BTDC with
an IVOP of 15 MPa. Sulfur dioxide is produced from the sulfur in the fuel or lubrication oil and is
formed more vigorously in fuel-rich conditions (equivalence ratios greater than 1). But even in fuel
lean conditions, the air to fuel ratio impacts SO2 formation. Figure 10 (d) mirrors Figure 8 (d); SO2
tracks closely with equivalence ratio. The conversion of SO2 to SO3 and eventually H2SO4 (which
is strongly hydrophilic and mixes with water and particulate matter in the exhaust, reducing the
amount of SO2) has been the object of other studies, especially with regard to catalysis design, and
it has been shown that the equivalence ratio and exhaust temperature signicantly inuence these
conversions [48]. Given this tendency of SO2, the lower equivalence ratio and exhaust temperatures
are likely the cause of the lower SO2 emissions at the injection timing of 25º BTDC and IVOP of
15 MPa.
The error bars in Figure 8, Figure 9, and Figure 10 are the standard error across the samples
taken per specic IVOP and timing position. Across the gures these error bars tend to decrease
as timing is advanced to 25° BTDC and IVOP is increased to 15 MPa. This means that not only
does engine performance and emissions tend to improve at this engine tuning position, but the
performance and emissions are more consistent.
One limitation of this study's injection tuning exercise is that it was only carried out at one
engine loading point, 75% load. Studies such as Bari et. al. have found engine performance and
emissions results to vary across loads as timing is changed. This study tuned the timing and IVOP
at 75% engine loading as this was assumed to be the most common engine loading for this specic
engine in its specic context (agro-processing in developing countries).
5.2. Pre-combustion Pressure
A pressure transducer provided by Kulite Semiconductor Products Inc. was used to map the
pressure in the pre-combustion chamber versus the crank angle. Three cases were tested:
ˆ diesel at standard (unmodied) conditions (room temperature fuel, IVOP of 9 MPa, and
injector timing of 20º BTDC)
ˆ WVO under unmodied conditions (room temperature fuel, IVOP of 9 MPa, and injector
timing of 20º BTDC)
ˆ WVO under modied conditions (fuel heated to 100°C, IVOP of 15 MPa, and injector timing
of 25º BTDC).
The engine was loaded to 75% and run at 650 RPM. The pressure transducer was mounted into the
COV plug preventing its use as a preheater; instead, the high pressure line was heated electrically.
The pressure traces are shown in Figure 11 (a). Ignition is usually identied by heat release,
specically the initial spike on the rate of heat release (RoHR) curve. To accurately calculate the
heat release of an IDI engine both the prechamber and main chamber pressures must be measured.
Though heat release can be calculated with only one or the other pressure, the calculation can
have as large as a 25% error, especially during early combustion [36]. Due to the availability of
only the prechamber pressure, heat release was not calculated. Instead, ignition was approximately](https://image.slidesharecdn.com/basinger-fuel-article-1-120501102512-phpapp02/75/Compression-Ignition-Engine-Modifications-for-Straight-Vegetable-Oil-Fuel-17-2048.jpg)
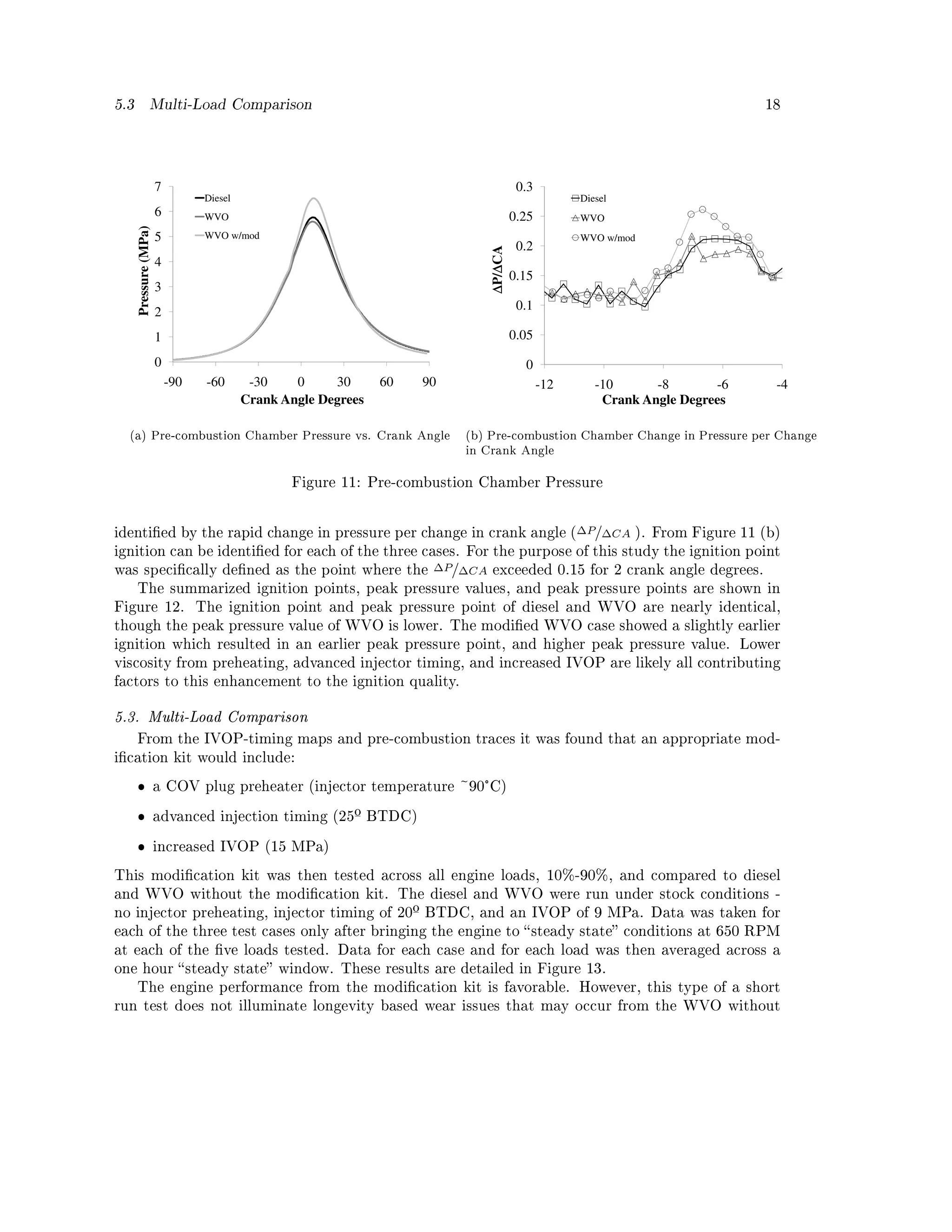
![5.3 Multi-Load Comparison 19
10 Diesel
WVO
ºBTDC, ºTTDC, MPa
8 WVO w/mod
6
4
2
0
Ignition point Peak Pressure Point Peak Pressure
(ºBTDC) (ºATDC) (MPa)
Figure 12: Pre-combustion Pressure Test Summary
modication case. In the short term, the modication kit performed similarly to, though slightly
better than the unmodied case, and diesel performed best of all.
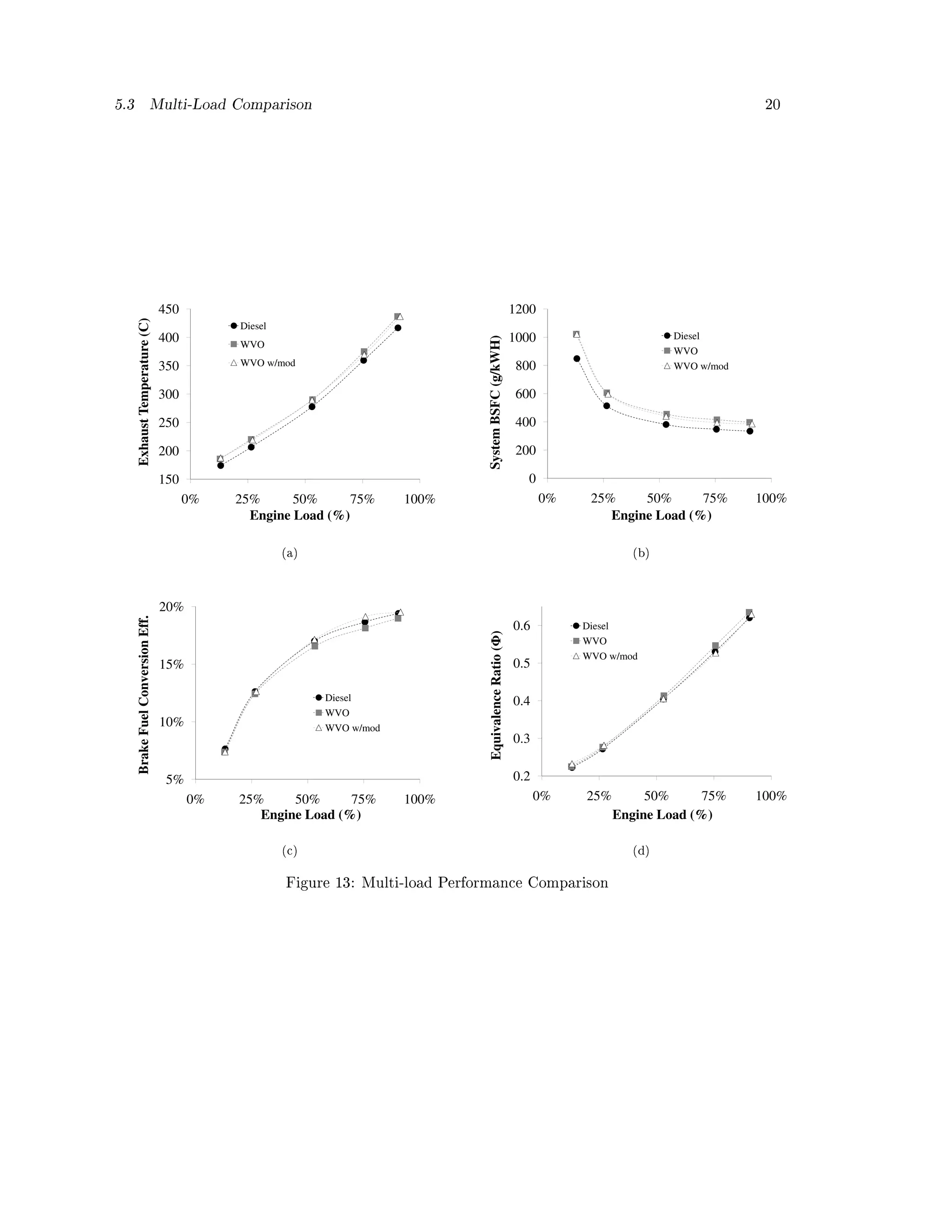
Figure 13 (a) shows the measured exhaust temperature. Across all loads: the WVO case was the
highest, the WVO case with modication kit was slightly lower, and the diesel case was lower still.
As mentioned in the above injection tuning section, the exhaust temperature is likely indicative
of the completeness of combustion. The WVO at stock timing, IVOP, and temperature is not
combusting as completely as the WVO with the modication kit. Diesel likely had the lowest
temperature because it is a lighter fuel than WVO. The heavier (less evaporate) WVO molecules
continued to burn late into the cycle, resulting in a higher temperature than the diesel.
Figure 13 (b) shows a similar trend in the System BSFC. The diesel case performed noticeably
better than either WVO case. It's important to note that at a caloric value of 45.8 MJ/kg versus
WVO's 39.4 MJ/kg, diesel has a strong energy density advantage. The eciency measurement
shown in Figure 13 (c) accounts for this dierence in heating values. At higher engine loading the
modied WVO case has nearly a 1% gain in eciency compared to the unmodied WVO case.
This same type of trend in BSFC and eciency was also found by Nwafor et. al. and Bari et al.
[42, 26].
The equivalence ratios for each of the three cases in Figure 13 (d) are nearly indistinguishable
from one another, though at high loads the unmodied WVO case does have slightly poorer values.
Bari et. al. also found the air to fuel ratio to decrease signicantly between diesel and WVO, and
to decreases slightly more when timing was advanced for the WVO case [26]. The same type of
trend was found in this study between the two WVO cases, especially at higher loads. Though the
equivalence ratio was not as low.
For each of the three cases the opacity was measured and is detailed in Figure 14. The low
levels measured do not present a strong contrast between the three cases, though an interesting
phenomenon occurred as engine loading increased: at lower engine loads the diesel exhaust was
less opaque but at higher loads the modied WVO performed best. Across all engine loads the
unmodied WVO had the most opaque exhaust. This higher opacity level of the WVO without
modication seems to follow from its relatively poorer equivalence ratio. The likely explanation of
the lower opacity of the exhaust from WVO in the modied engine operation at high load when](https://image.slidesharecdn.com/basinger-fuel-article-1-120501102512-phpapp02/75/Compression-Ignition-Engine-Modifications-for-Straight-Vegetable-Oil-Fuel-19-2048.jpg)
![5.3 Multi-Load Comparison 21
12
Diesel
10 WVO
WVO w/mod
Opacity (%)
8
6
4
2
0
0% 25% 50% 75% 100%
Engine Load (%)
Figure 14: Multi-Load Opacity Comparison
compared to a diesel operated engine is that at low loads the lighter molecular weight of the diesel
results in a spray that atomizes to smaller droplet sizes, which burn more completely. But as
loading increases, the role of the increased IVOP becomes more important. The likely reduction in
droplet size and the improved mixing from the increased IVOP are keeping the equivalence ratio
lower then the diesel case and thus the opacity is slightly lower. There is more air to oxidize the
fuel, resulting in less soot formation.
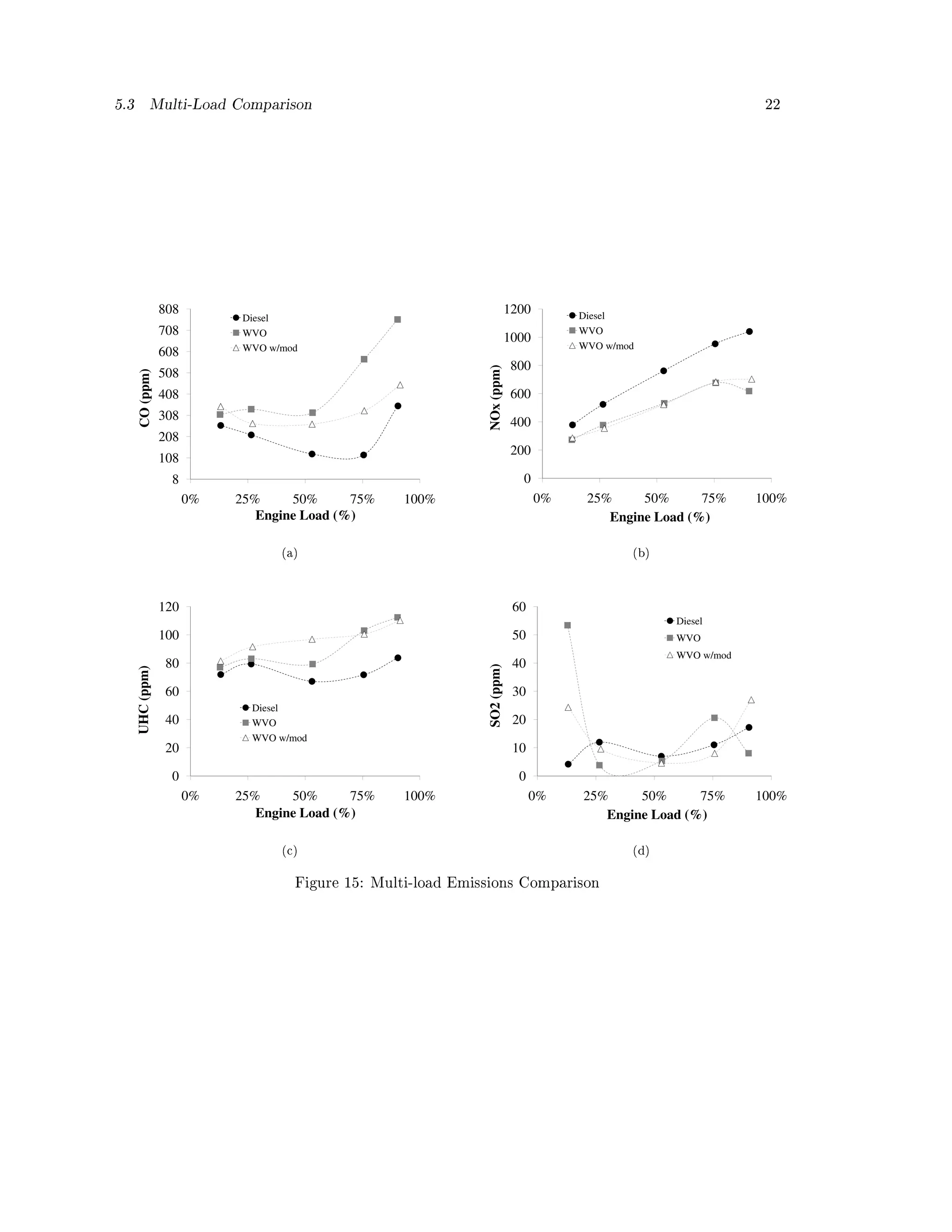
The measured emissions for each case are compared in Figure 15. As indicated by the lower CO,
UHC, SO2, and the higher NOx levels, the diesel seems to have the best combustion characteristics.
Unmodied WVO generally emits more CO and SO2, though the modied WVO case did tend to
have slightly higher UHC and lower NOx.
The CO results for the unmodied WVO and diesel cases shown in Figure 15 (a) resemble the
trend found by Bari et. al. [26]. At all loads diesel CO is the lowest. At low loads unmodied
WVO is relatively at, then at mid load the CO begins to increase. The modied WVO case is in
between the diesel and unmodied case. This is likely because the modication improves mixing
and evaporation, but the heavier WVO molecules with their slower evaporation rate still do not
burn as completely as the diesel.
When comparing the opacity values to the CO values of the unmodied WVO case and diesel
case at high loads, it appears that the increased availability of oxidant from improved mixing and
the lower droplet size (due to the increased IVOP) is enough to reduce the soot, but not enough to
also lower the CO below the diesel level. The increased IVOP is denitely impactful, but the WVO
is still molecularly heavier than diesel.
The NOx values in Figure 15 (b) show the diesel to have the highest values across all loads, while
both WVO cases are nearly identical. Like the CO, this points to a more complete combustion in
the diesel case, which in turn provides hotter temperatures for greater thermal NOx generation.
The UHC values in Figure 15 (c) also show the diesel to have the best emissions, though at low
loads the three cases are very similar. At medium loads the modied WVO case is higher than the
unmodied case. This might be due to increased wall impingement from the higher IVOP in the
modied case. At high loads the two WVO cases are indistinguishable.](https://image.slidesharecdn.com/basinger-fuel-article-1-120501102512-phpapp02/75/Compression-Ignition-Engine-Modifications-for-Straight-Vegetable-Oil-Fuel-21-2048.jpg)

![24
AppendixA. Selected Properties of Several Plant Oils and Diesel
Table A.2: Physical and Fuel Properties of Several Plant Oils and Diesel
Diesel Soybean Rapeseed Peanut Palm Jatropha WVO
Yield
450-480 590-1200 850-1100 2800-6000 740-1590
(L/Ha-Yr)
Viscosity
a 2.6-3.6 33 37-42 40 39 34-37 36
(cSt)
Density
820-845 914-924 912-920 888-902 860-910 860-933 910-940
(kg/m3)
Caloric Value
43-46 36.9-39.6 36.8-39.7 39.5-39.8 36.5-40.1 37.8-42.1 39.2-39.6
(MJ/kg)
Cetane
45-56 36-38 38-41 35-42 42-49 38-45 36-37
Number
[49, 11, [11, 54, [11, 54, [11, 30, [60, 61,
[49, 50, original
Reference 54, 55, 56, 55, 58, 5, 55, 58, 59, 50, 55, 52, 11, 12, 55,
51, 52, 53] data
57, 53, 58] 56, 57, 53] 56, 57, 53] 56, 57] 56, 62, 57]
a Viscosity of Diesel, Palm, Jatropha, and WVO measured at 40C; all others measured at 38C
Table A.3: Chemical Composition of Several Plant Oils and Diesel
Element Diesel Soybean Rapeseed Peanut Palm Jatropha WVO
C 80.33-86 76.2-77.1 77.9-78 70-76.55 50.27 76.11-76.56 76.50-77.78
H 12.36-14.8 11.6-12.9 10-13.2 11.97 7.07 10.52-13.19 11.55-12.07
O 1.19 10-10.4 8.9-12 11.48 36.28 11.06 11.1-11.57
S 0.1-0.25 0.01 0.0012-0.01 0.01 0.4-0.63 0.02-0.03
N 1.76 1.9 0.42 0.34 0.02-0.03
C Residue 0.1-0.14 0.24-0.27 0.3-0.31 0.22-0.24 0.22-0.24 0.7-0.9
Ash 0.01 0.006-0.01 0.01-0.54 0.005-0.02 5.33 0.03-0.036
[61, 5, 52, [54, 58, 63, [54, 58, 64, original
Reference [54, 58, 64] [52, 65] [60, 61, 12]
53] 53] 5] data](https://image.slidesharecdn.com/basinger-fuel-article-1-120501102512-phpapp02/75/Compression-Ignition-Engine-Modifications-for-Straight-Vegetable-Oil-Fuel-24-2048.jpg)
![25
Table A.4: Lipid Prole of Several Plant Oils
Plant Oil:
Lipids Soybean Rapeseed Peanut Palm Jatropha WVO
Lauric
0-1.15 5.9 0.02-0.04
(C12:0)
Myristic
0.1 0.5-2.74 0.1-2.7 0.14-0.25
(C14:0)
Palmitic
11.3-13.9 3.49-3.5 8-11.34 26.18-47.5 14.1-15.3 6.74-12.4
(C16:0)
Palmitoleic
0.1-0.3 0.1-1.66 1.3 0.47-1.0
(C16:1)
Stearic
2.1-3.6 0.85-1.6 1.8-2.4 3.5-11.97 3.7-9.8 2.79-4.47
(C18:0)
Oleic
23.2-24.9 33-64.4 48.28-53.3 35.49-46.1 21.8-45.8 31.50-58.2
(C18:1)
Linoleic
53-56.2 20.4-22.3 28.4-32 6.5-12.76 29-47.4 21.2-42.20
(C18:2)
Linolenic
4.3-6.31 7.9-8.23 0.3-0.93 0-2.25 0.3 5.85-7.4
(C18:3)
Arachidic
0.3 0.9-1.32 0.4-1.74 0.3 0.39-0.62
(C20:0)
Gadoleic
0.3 9.3 2.4 0.2-2.56
(C20:1)
Behenic
2.52-3 0.2 0.34-0.35
(C22:0)
Erucic
0.3 23 0-1.49 0.06-0.08
(C22:1)
Lignoceric
0.1 1.23-1.8 0.1 0.07-0.09
(C24:0)
Iodine
69.82-152 81-120 80-119.55 44-65.5 92-112 107-115
Value
[54, 58, 18, [54, 58, 18, [67, 54, 58, [67, 18, 9, original
Reference [60, 68, 69]
9, 66] 9, 66] 18, 66] 66] data
References
[1] P. Vasudevan, S. Sharma, A. Kumar, Liquid fuel from biomass: an overview, Journal of Sci-
entic Industrial Research 64 (11) (2005) 822831.
[2] United States Department of Energy, Annual energy report (2008).
[3] D. Pimentel, A. Marklein, M. A. Toth, M. N. Karpo, G. S. Paul, R. Mccormack, J. Kyriazis,](https://image.slidesharecdn.com/basinger-fuel-article-1-120501102512-phpapp02/75/Compression-Ignition-Engine-Modifications-for-Straight-Vegetable-Oil-Fuel-25-2048.jpg)
![26
T. Krueger, Food versus biofuels: Environmental and economic costs, Human Ecology 37 (1)
(2009) 112.
[4] V. Modi, Energy services for the poor, Tech. rep., Millennium Project Task Force 1 (2004).
[5] N. Hemmerlein, V. Korte, H. Richter, G. Schroder, Performance, exhaust emissions and dura-
bility of modern diesel engines running on rapeseed oil, SAE Paper 910848 (1991).
[6] T. W. Ryan, L. G. Dodge, T. J. Callahan, The eects of vegetable oil properties on injection
and combustion in two dierent diesel engines, Journal of the American Oil Chemists 61 (10)
(1984) 16101619.
[7] Y. Hui (Ed.), Bailey's Industrial Oil Fat Products, 5th Edition, Wiley, New York, 1996.
[8] M. S. Graboski, R. L. McCormick, Combustion of fat and vegetable oil derived fuels in diesel
engines, Progress in Energy and Combustion Science 24 (2) (1998) 125164.
[9] S. L. Nettles-Anders, D. B. Olsen, Survey of straight vegetable oil composition impact on
combustion properties, SAE Paper 2009-01-0487 (2009).
[10] A. S. Ramadhas, S. Jayaraj, C. Muraleedharan, Use of vegetable oils as i.c. engine fuelsa
review, Renewable Energy 29 (5) (2004) 727742.
[11] A. Babu, G. Devaradjane, Vegetable oils and their derivatives as fuels for ci engines: An
overview, SAE Paper 2003-01-0767 (2003).
[12] H. K. Gangwar, A. K. Agarwal, Emission and combustion characteristics of vegetable oil
(jatropha curcus) blends in an indirect ignition transportation engine, SAE Paper 2008-28-
0034 (2008).
[13] C. Rakopoulos, K. Antonopoulos, D. Rakopoulos, D. Hountalas, E. Giakoumis, Comparative
performance and emissions study of a direct injection diesel engine using blends of diesel fuel
with vegetable oils or bio-diesels of various origins, Energy Conversion and Management 47 (18-
19) (2006) 32723287.
[14] L. Schumacher, Biodiesel lubricity, Tech. rep., University of Idaho (2003).
[15] R. Korus, J. Jo, C. Peterson, A rapid engine test to measure injector fouling in diesel engines
using vegetable oil fuels, Journal of the American Oil Chemists' Society 62 (11) (1985) 1563
1564.
[16] A. Schonborn, N. Ladommatos, R. Allan, J. Williams, J. Rogerson, Eect of the molecular
structure of individual fatty acid alcohol esters (biodiesel) on the formation of nox and partic-
ulate matter in the diesel combustion process, SAE Paper 2008-01-1578 (2008).
[17] T. W. I. Ryan, M. O. Bagby, Identication of chemical changes occuring during the transient
injection of selected vegetable oils, SAE Paper 930933 (1993).
[18] G. Knothe, Structure indices in fa chemistry. how relevant is the iodine value?, Journal of the
American Oil Chemists' Society 79 (9) (2002) 847854.](https://image.slidesharecdn.com/basinger-fuel-article-1-120501102512-phpapp02/75/Compression-Ignition-Engine-Modifications-for-Straight-Vegetable-Oil-Fuel-26-2048.jpg)
![27
[19] P. Mondal, M. Basu, N. Balasubramanian, Direct use of vegetable oil and animal fat as al-
ternative fuel in internal combustion engine, Biofuels Bioproducts Biorening-Biofpr 2 (2)
(2008) 155174.
[20] C. Peterson, S. Jones, Using unmodied vegetable oils as a diesel fuel extender a literature
review, Tech. rep., University of Idaho (2002).
[21] P. A. D. A. K. Hossain, Plant oils as fuels for compression ignition engines: A technical review
and life-cycle analysis, Renewable Energy 35 (2010) 113.
[22] M. Vojtisek-Lom, J. Blazek, M. Dufek, M. Fenkl, Investigation of combustion rates and in-
jection and ignition onset of heated rapeseed oil in direct-injection turbodiesel engines, Tech.
Rep. 2009-01-1914, SAE (2009 2009).
[23] C. Engler, L. Johnson, W. Lepori, C. Yarbrough, Eects of processing and chemical char-
acteristics of plant oils on performance of an indirect-injection diesel engine 60 (8) (1983)
15921596.
[24] K. J. Suda, Vegetable oil or diesel fuel-a exible option, Tech. Rep. 840004, SAE International,
engine Study (1984 1984).
[25] S. Bari, C. Yu, T. Lim, Filter clogging and power loss issues while running a diesel engine with
waste cooking oil, Proceedings of the Institution of Mechanical Engineers. Part D, Journal of
automobile engineering 216 (12) (2002) 9931001.
[26] S. Bari, C. W. Yu, T. H. Lim, Eect of fuel injection timing with waste cooking oil as a fuel
in a direct injection diesel engine, Proceedings of the Institution of Mechanical Engineers Part
D-Journal of Automobile Engineering 218 (D1) (2004) 93104.
[27] M. Pugazhvadivu, K. Jeyachandran, Investigations on the performance and exhaust emissions
of a diesel engine using preheated waste frying oil as fuel, Renewable Energy 30 (14) (2005)
21892202.
[28] O. M. I. Nwafor, The eect of elevated fuel inlet temperature on performance of diesel engine
running on neat vegetable oil at constant speed conditions, Renewable Energy 28 (2) (2003)
171181.
[29] J. Dweck, C. Sampaio, Analysis of the thermal decomposition of commercial vegetable oils
in air by simultaneous tg/dta, Journal of Thermal Analysis and Calorimetry 75 (2) (2004)
385391.
[30] S. Bari, T. H. Lim, C. W. Yu, Eects of preheating of crude palm oil (cpo) on injection system,
performance and emission of a diesel engine, Renewable Energy 27 (3) (2002) 339351.
[31] I. Celikten, An experimental investigation of the eect of the injection pressure on engine
performance and exhaust emission in indirect injection diesel engines, Applied Thermal Engi-
neering 23 (16) (2003) 20512060.
[32] Z.-M. Cao, K. Nishino, S. Mizuno, K. Torii, Piv measurement of internal structure of diesel
fuel spray, Experiments in uids 29 (7) (2000) S211S219.](https://image.slidesharecdn.com/basinger-fuel-article-1-120501102512-phpapp02/75/Compression-Ignition-Engine-Modifications-for-Straight-Vegetable-Oil-Fuel-27-2048.jpg)
![28
[33] K. Enoki, S. Hayashi, N. Sawa, Eective utilization of blended plant fuels for pre-combustion
chamber compression ignition engines, SAE Paper 940330 (1994).
[34] T. Su, C. Chang, R. Reitz, P. Farrell, A. Pierpont, T. Tow, Eects of injection pressure and
nozzle geometry on spray smd and d.i. emissions, SAE Paper 952360 (1995).
[35] K. Varde, D. Popa, L. Varde, A study of diesel spray characteristics at high injection, Tech.
rep., U.S. Dept. of Transportation, Oce of the Secretary of Transportation (1985).
[36] J. B. Heywood, Internal Combustion Engine Fundamentals, McGraw-Hill Book Company, New
York, 1988.
[37] K. Openshaw, A review of jatropha curcas: an oil plant of unfullled promise, Biomass and
Bioenergy 19 (1) (2000) 115.
[38] C. S. Lee, S. W. Park, S. I. Kwon, An experimental study on the atomization and combustion
characteristics of biodiesel-blended fuels, Energy Fuels 19 (5) (2005) 22012208.
[39] T. Murayama, Y.-t. Oh, N. Miyamoto, T. Chikahisa, N. Takagi, K. Itow, Low carbon ower
buildup, low smoke, and ecient diesel operation with vegetable oils by converstion to mono-
esters and blending with diesel oil or alcohols, SAE Paper 841161 (1984).
[40] S. Puhan, R. Jegan, K. Balasubbramanian, G. Nagarajan, Eect of injection pressure on
performance, emission and combustion characteristics of high linolenic linseed oil methyl ester
in a di diesel engine, Renewable Energy 34 (5) (2009) 12271233.
[41] S. Haldar, B. Ghosh, A. Nag, Studies on the comparison of performance and emission char-
acteristics of a diesel engine using three degummed non-edible vegetable oils, Biomass and
Bioenergy 33 (8) (2009) 10131018.
[42] O. M. I. Nwafor, G. Rice, A. I. Ogbonna, Eect of advanced injection timing on the performance
of rapeseed oil in diesel engines, Renewable Energy 21 (3-4) (2000) 433444.
[43] SAE International, Measurement of intake air or exhaust gas ow of diesel engines, SAE
International Standard J244 (1992).
[44] R.A. Lister Co., Ltd., Dursley, Gloucestershire, England, Instruction Book and Spare Parts
List.
[45] B. Gebhart, Y. Jaluria, R. J. Mahajan, B. Sammakia (Eds.), Buoyancy-Induced Flows and
Transport, Hemisphere Publishing Company, New York, 1988.
[46] W. M. Rohsenow, J. P. Hartnett, E. N. Ganic (Eds.), Handbook of Heat Transfer Fundamen-
tals, McGraw-Hill Book Company, New York, 1985.
[47] D. J. Patterson, N. A. Henein, Emissions from Combustion Engines and Their Control, Ann
Arbor Science Publishers Inc., An Arbor, 1972.
[48] G. Corro, Sulfur impact on diesel emission control - a review, Reaction Kinetics and Catalysis
Letters 75 (1) (2002) 89106.](https://image.slidesharecdn.com/basinger-fuel-article-1-120501102512-phpapp02/75/Compression-Ignition-Engine-Modifications-for-Straight-Vegetable-Oil-Fuel-28-2048.jpg)
![29
[49] E. Alptekin, M. Canakci, Characterization of the key fuel properties of methyl ester-diesel fuel
blends, Fuel 88 (1) (2009) 7580.
[50] S. C. A. de Almeida, C. R. Belchior, M. V. G. Nascimento, L. d. S. R. Vieira, G. Fleury,
Performance of a diesel generator fuelled with palm oil, Fuel 81 (16) (2002) 20972102.
[51] M. D. Clevenger, M. O. Bagby, C. E. Goering, A. W. Schwab, L. D. Savage, Developing an
accelerated test of coking tendencies of alternative fuels, Transactions of the ASAE 31 (4)
(1988) 10541058.
[52] M. A. Kalam, H. H. Masjuki, Emissions and deposit characteristics of a small diesel engine
when operated on preheated crude palm oil, Biomass and Bioenergy 27 (3) (2004) 289297.
[53] H. Masjuki, A rapid test to measure performance, emission and wear of a diesel engine fueled
with palm oil diesel, Journal of the American Oil Chemists' Society 70 (1993) 10211025.
[54] A. Demirbas, Chemical and fuel properties of seventeen vegetable oils, Energy Sources 25 (7)
(2003) 721728.
[55] P. Girard, A. Fallot, Review of existing and emerging technologies for the production of biofuels
in developing countries, Energy for Sustainable Development 10 (2) (2006) 92108, develop-
ment.
[56] W. Korbitz, Worldwide trends in production and marketing of biodiesel, Austrian Biofuels
Institute (May 2000).
[57] V. Modi, S. McDade, D. Lallement, J. Saghir, Energy services for the millenium development
goals, Tech. rep., UNDP (2005).
[58] C. Goering, A. Schwab, M. Daugherty, E. Pryde, A. Keakin, Fuel properties of eleven vegetable
oils, American Society of Agricultural Engineers CONF-811221 (1982).
[59] R. Altin, S. Cetinkaya, H. S. Yucesu, The potential of using vegetable oil fuels as fuel for diesel
engines, Energy Conversion and Management 42 (5) (2001) 529538.
[60] W. Achten, L. Verchot, Y. Franken, E. Mathijs, V. Singh, R. Aerts, B. Muys, Jatropha bio-
diesel production and use, Biomass and Bioenergy 32 (12) (2008) 10631084, jatropha.
[61] D. Agarwal, A. K. Agarwal, Performance and emissions characteristics of jatropha oil (pre-
heated and blends) in a direct injection compression ignition engine, Applied Thermal Engi-
neering 27 (13) (2007) 23142323.
[62] E. L. M. Rabe, Jatropha oil in compression ignition engines, Ph.D. thesis, Eindhoven University
of Technology (2007).
[63] T.-A. Ngo, J. Kim, S. K. Kim, S.-S. Kim, Pyrolysis of soybean oil with h-zsm5 (proton-exchange
of zeolite socony mobil??5) and mcm41 (mobil composition of matter no. 41) catalysts in a
xed-bed reactor, Energy In Press, Corrected Proof.
[64] C. Kaya, C. Hamamci, A. Baysal, O. Akba, S. Erdogan, A. Saydut, Methyl ester of peanut
(arachis hypogea l.) seed oil as a potential feedstock for biodiesel production, Renewable Energy
34 (5) (2009) 12571260.](https://image.slidesharecdn.com/basinger-fuel-article-1-120501102512-phpapp02/75/Compression-Ignition-Engine-Modifications-for-Straight-Vegetable-Oil-Fuel-29-2048.jpg)
![30
[65] M. O. Bagby, Vegetable oils for diesel fuel: Opportunities for development, Tech. Rep. 87-1588,
American Society of Agricultural Engineers (1987).
[66] T. Ogawa, M. Okada, Inuence of properties and composition of diesel fuels on particle emis-
sions, Tech. rep., RD Review of Toyota CRDL (November 2003).
[67] A. Abollé, L. Kouakou, H. Planche, The viscosity of diesel oil and mixtures with straight
vegetable oils: Palm, cabbage palm, cotton, groundnut, copra and sunower, Biomass and
Bioenergy In Press, Corrected Proof.
[68] J. S. de Oliveira, P. M. Leite, L. B. de Souza, V. M. Mello, E. C. Silva, J. C. Rubim, S. M.
Meneghetti, P. A. Suarez, Characteristics and composition of jatropha gossypiifoliaand jat-
ropha curcas l. oils and application for biodiesel production, Biomass and Bioenergy 33 (3)
(2009) 449453.
[69] M. Senthil Kumar, A. Ramesh, B. Nagalingam, An experimental comparison of methods to
use methanol and jatropha oil in a compression ignition engine, Biomass and Bioenergy 25 (3)
(2003) 309318.](https://image.slidesharecdn.com/basinger-fuel-article-1-120501102512-phpapp02/75/Compression-Ignition-Engine-Modifications-for-Straight-Vegetable-Oil-Fuel-30-2048.jpg)