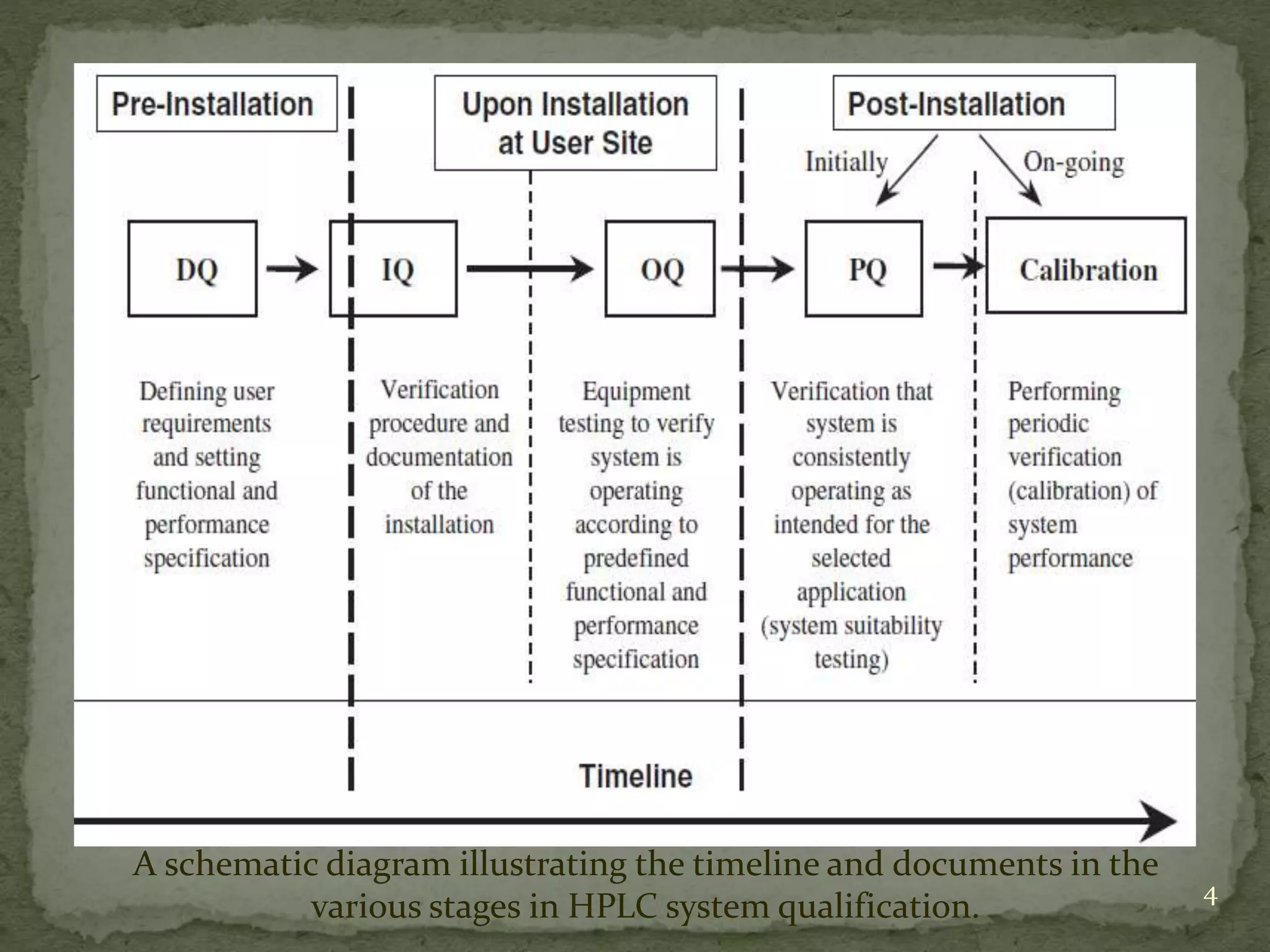
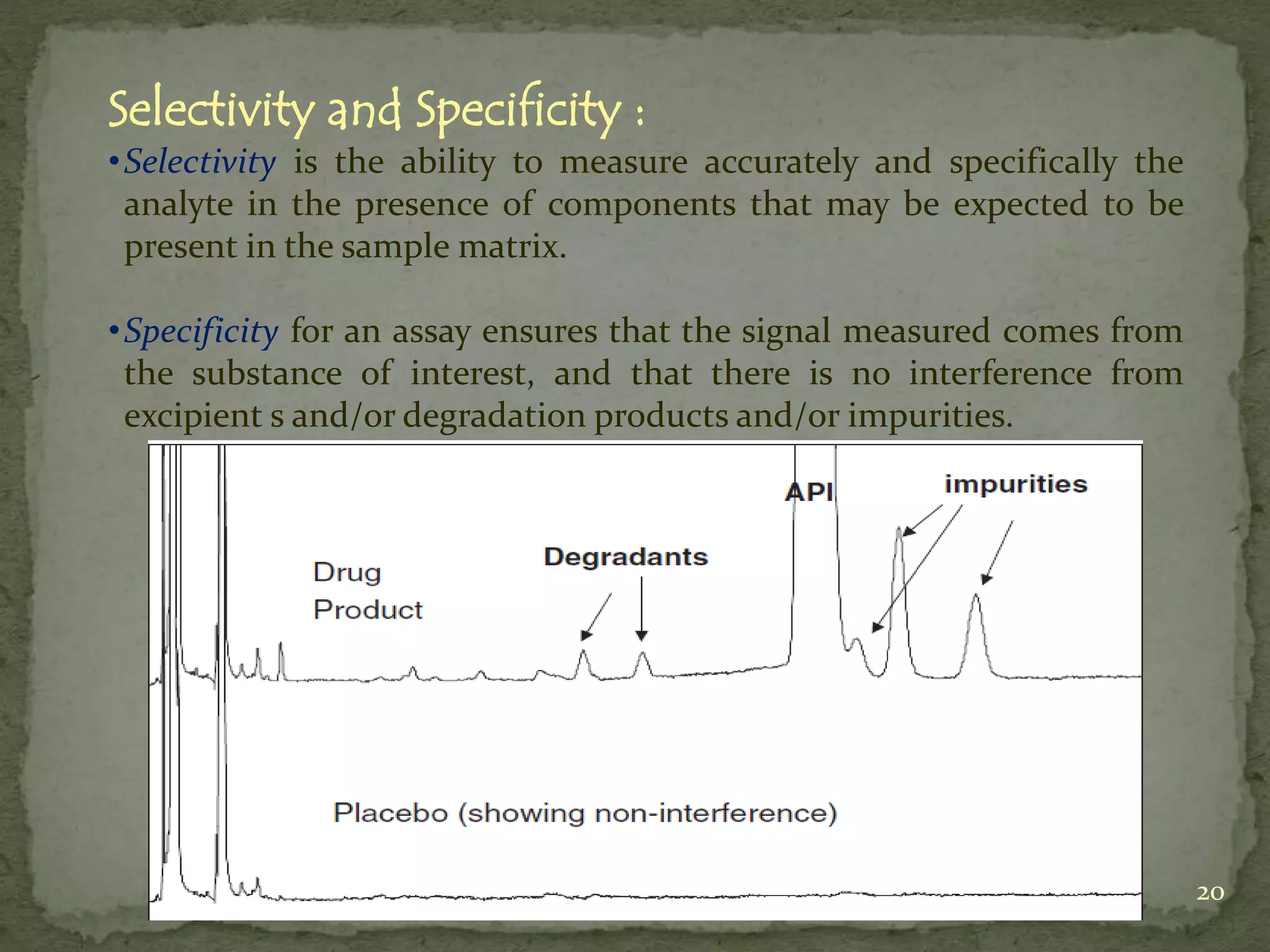
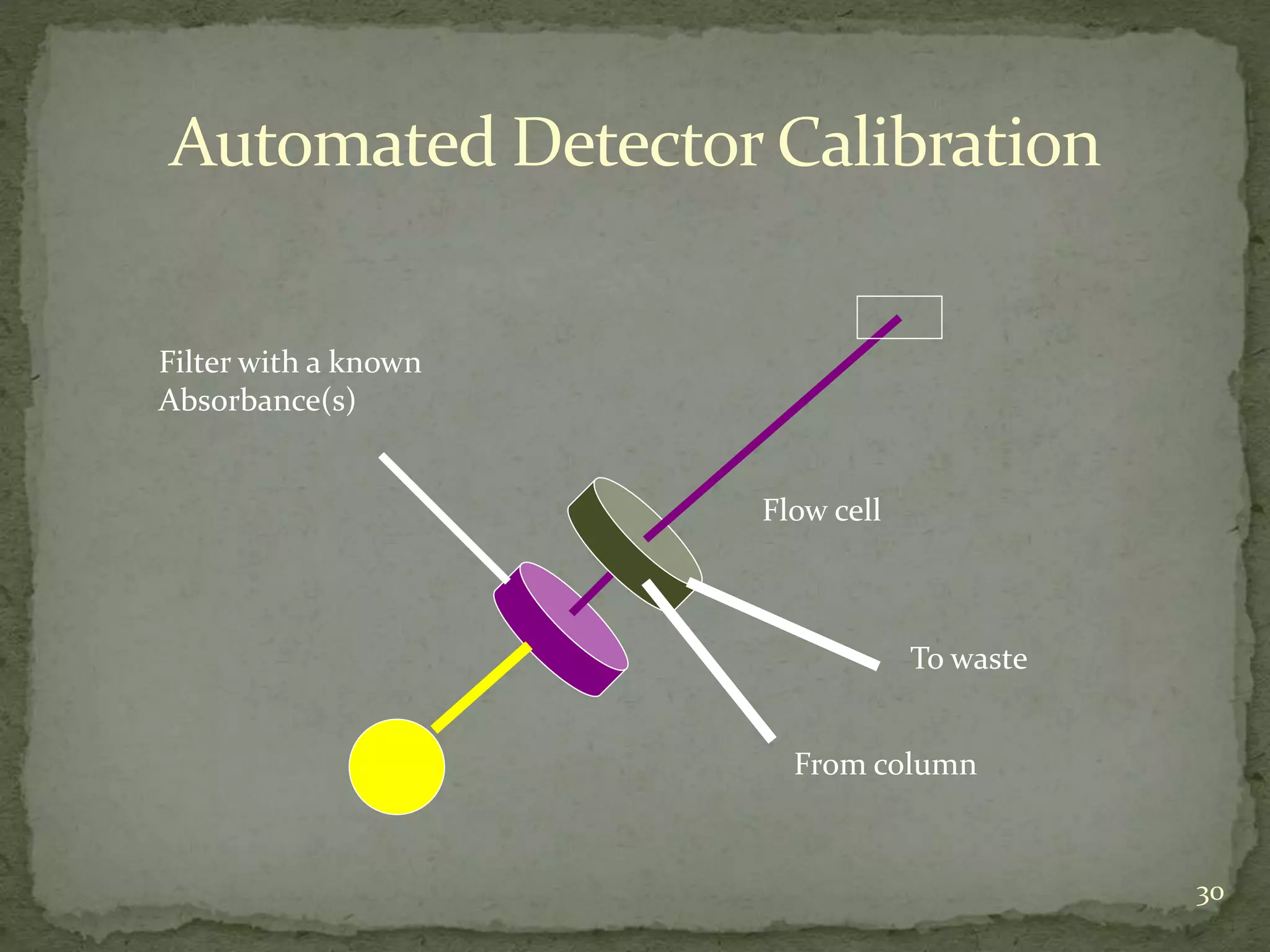
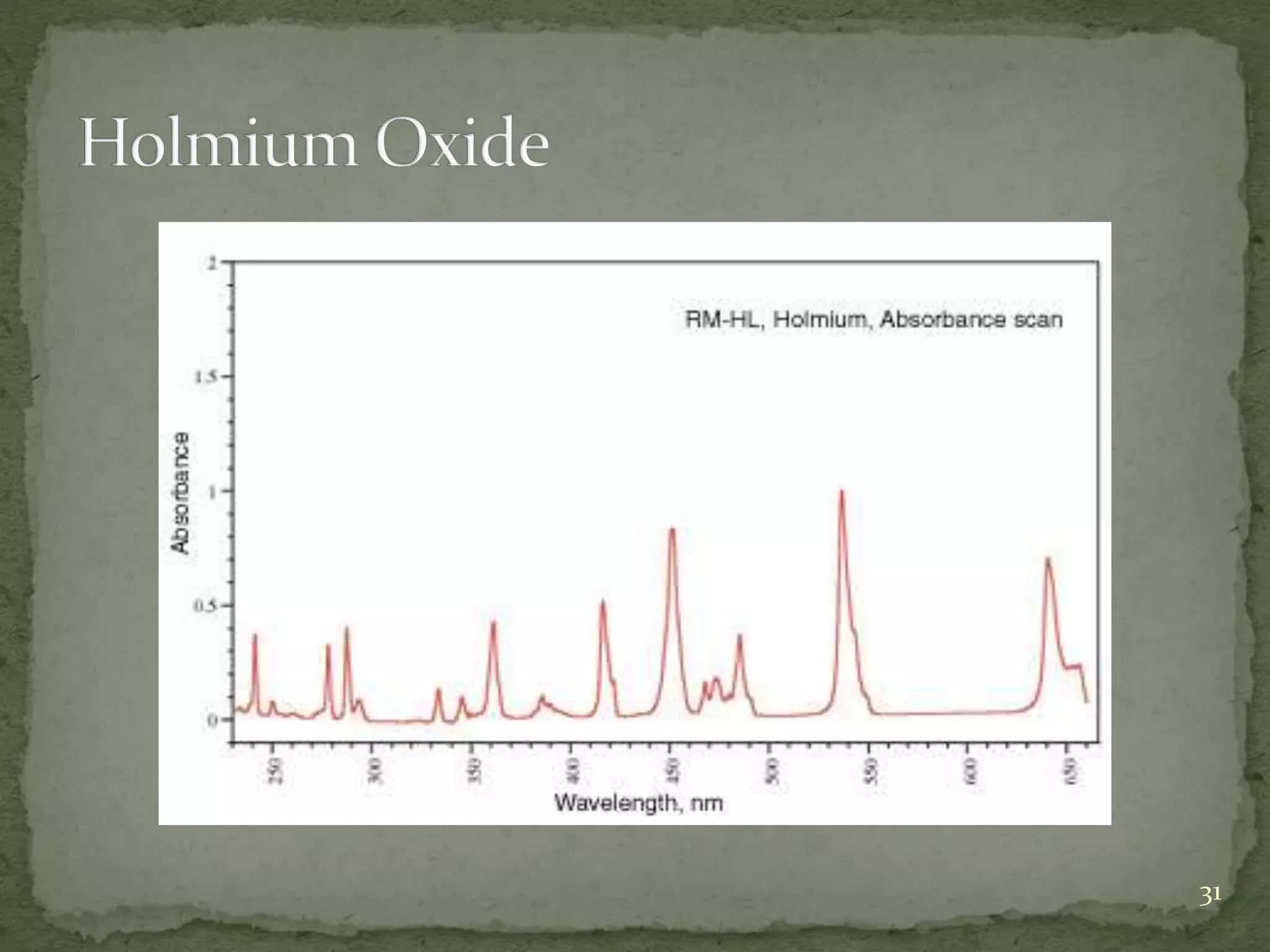
This document discusses validation and calibration of HPLC systems. It defines validation as establishing that an analytical procedure meets requirements for its intended use through laboratory studies. A validation protocol outlines how validation will be conducted. Equipment validation demonstrates that equipment is suitable for use and comparable to routine equipment. Calibration involves demonstrating that an instrument produces results within specified limits compared to a reference standard. The document outlines parameters to validate like accuracy, precision, specificity, range, robustness and more. It provides details on testing these parameters and accepting calibration of modules like the pump, injector, detector and column heating.