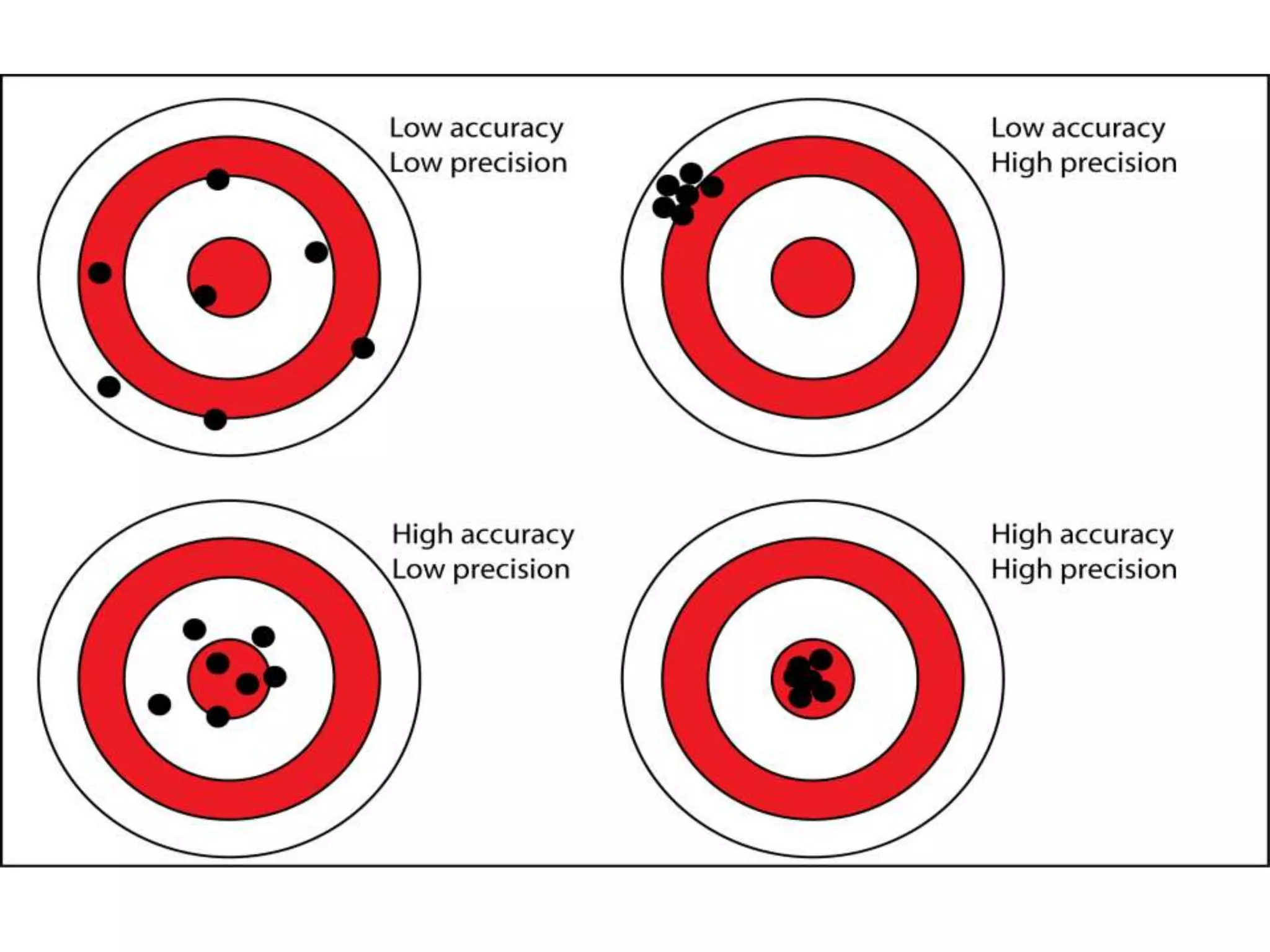
This document outlines guidelines for analytical method validation as described in ICH Q2. It defines validation as establishing evidence that a method is suitable for its intended purpose. Key parameters that must be validated include accuracy, precision, specificity, detection limit, quantitation limit, linearity, range, and robustness. The guidelines provide details on how to validate these parameters and determine method suitability.