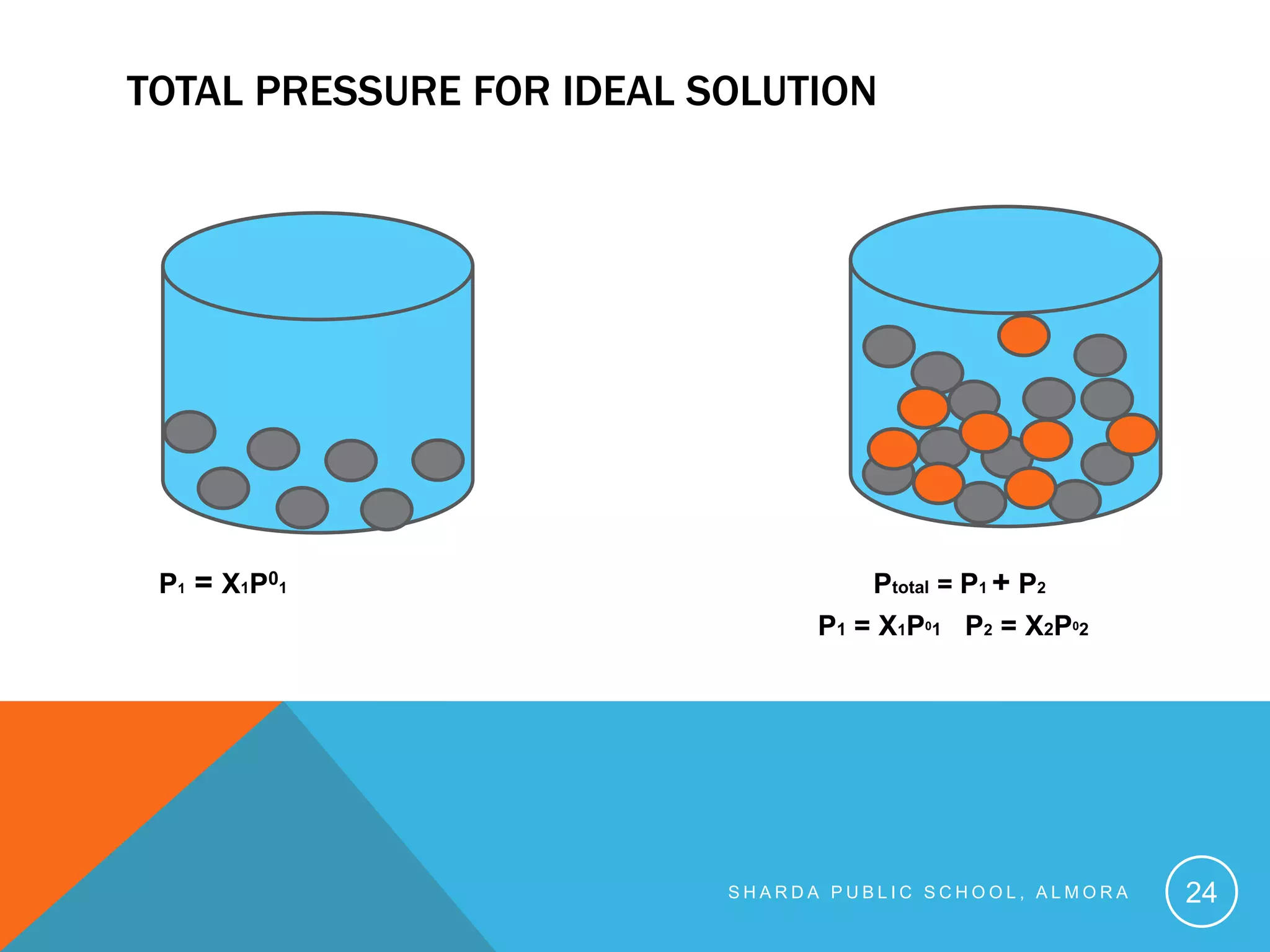
The document discusses solutions and solution chemistry. It begins by defining solutions as homogeneous mixtures of two or more pure substances, with the solute dispersed uniformly throughout the solvent. It then discusses how solutions form and the energy changes that occur, including the enthalpy changes of separating solute and solvent particles and forming new interactions between them. The rest of the document discusses various factors that affect solubility, such as the nature of the solute and solvent, intermolecular forces, temperature, and pressure. It also covers colligative properties of solutions like vapor pressure lowering, boiling point elevation, freezing point depression, and osmotic pressure.