Green Nanotechnology
- 1. GREEN NANO TECHNOLOGY Dr. Tanuja Nautiyal Associate Professor Department of Chemistry
- 2. INTRODUCTION What is Green Nanotechnology? Whenever the term “Green” is used, it refers to the environment friendly objects and “Technology” means the application of knowledge for practical purposes. 2
- 3. INTRODUCTION • Green nanotechnology is the study of how nanotechnology can benefit the environment, such as by using less energy during the manufacturing process, the ability to recycle products after use, and using eco-friendly materials. 3
- 4. PURPOSE • Nanotechnology, or tiny tech, uses science and engineering to manipulate and create materials out of atoms and molecules at the ultra-small scale of less than 100 nanometers. It is one hundred- thousandth the width of a human hair. • At the nanoscale level, conventional materials have unconventional and unexpected properties. 4
- 5. PURPOSE • Nanoparticles could also be used to remove industrial pollutants in contaminated air, soil, and groundwater, and nanofilters might be used to purify water and to desalinate water at an affordable cost. 5
- 6. PURPOSE • The technology could also be used to turn garbage into breakfast by mimicking how nature turns wastes into plant nutrients, thus following the nutrient cycling principle of sustainability. 6
- 7. Principles of Green Chemistry 7 • Prevention. Preventing waste is better than treating or cleaning up waste after it is created. • Atom economy. Synthetic methods should try to maximize the incorporation of all materials used in the process into the final product.
- 8. 8 • Less hazardous chemical syntheses. Synthetic methods should avoid using or generating substances toxic to humans and/or the environment. • Designing safer chemicals. Chemical products should be designed to achieve their desired function while being as non-toxic as possible. Principles of Green Chemistry
- 9. 9 • Safer solvents and auxiliaries. Auxiliary substances should be avoided wherever possible. • Design for energy efficiency. Energy requirements should be minimized. Principles of Green Chemistry
- 10. 10 Principles of Green Chemistry • Use of renewable feedstocks. Whenever it is practical to do so, renewable feedstocks or raw materials are preferable to non-renewable ones. • Reduce derivatives. Unnecessary generation of derivatives—such as the use of protecting groups—should be minimized or avoided if possible.
- 11. 11 • Catalysis. Catalytic reagents that can be used in small quantities to repeat a reaction are superior to reagents (ones that are consumed in a reaction). • Design for degradation. Chemical products should be designed so that they do not pollute the environment. Principles of Green Chemistry
- 12. 12 Principles of Green Chemistry • Real-time analysis for pollution prevention. Analytical methodologies need to be further developed to permit real-time monitoring and control before hazardous substances form. • Inherently safer chemistry for accident prevention. Whenever possible, the substances in a process, and the forms of those substances, should be chosen to minimize risks such as explosions and accidental releases.
- 13. 13 • Atom economy is a way to measure the atoms wasted when making a chemical. The higher the atom economy, the 'greener' the process. • 100 per cent atom economy means that all the atoms in the reactants have been converted to the desired product. ATOM ECONOMY
- 14. ATOM ECONOMY 14
- 15. TOOLS OF GREEN TECHNOLOGY • Green Starting Materials • Green Reagents • Green Reactions • Green Chemical Products • Green Methodologies 15
- 16. Green Technology Achievements Development of new insecticides. New synthesis of Ibuprofen. Development of CO2 as a solvent for dry cleaning. Barry Trost’s concept of Atom Economy for expressing the efficiency of a reaction. Use of waste CO2 as a blowing agent instead of CFC’s for foam polystyrene synthesis. 16
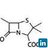
- 17. Synthesis of Ibuprofen: A Green Reaction Atom economy by this process is only 40.1%. 17
- 18. GREEN SYNTHESIS OF IBUPROFEN 18 Atom economy by this process is 77.4%.
- 19. Zero Waste Technology Zero Waste is a philosophy that encourages the redesign of resource life cycles so that all products are reused. The goal is for no trash to be sent to landfills or incinerators. The process recommended is one similar to the way that resources are reused in nature. 19
- 20. 20
- 21. TIPS FOR SUSTAINANBLE LAB • Run experiments on the micro scale to reduce waste. • Switch to green solvents: Use 2-methyl tetrahydrofuran in place of methylene chloride, and use cyclopentylmethyl ether in place of tetrahydrofuran, 1, 4-dioxane and ether. 21
- 22. TIPS FOR SUSTAINANBLE LAB • Neutralize basic phosphate-buffered HPLC waste or acidic HCl waste to pH 7 and pour down the drain. • Recycle electronics, ice packs, packaging materials, toner cartridges, pipette tip boxes, and water purification cartridges. 22
- 23. BIBLIOGRAPHY • G.T. Miller, Environmental Science, Thomas Learning, 2012 • Textbook of Environmental Studies, Dr. Sushmita Mohapatra, 2016 • https://en.wikipedia.org/wiki/Green_nanotechnology • https://www.nanowerk.com/nanotechnology-in-green- industries.php • https://en.wikipedia.org/wiki/Green_chemistry • https://en.wikipedia.org/wiki/Zero_waste 23
- 24. 24 GO GREEN