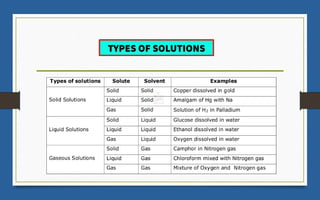
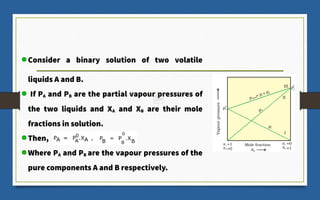
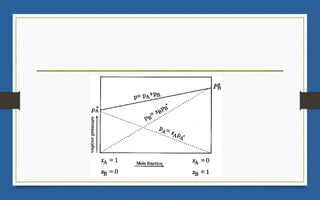
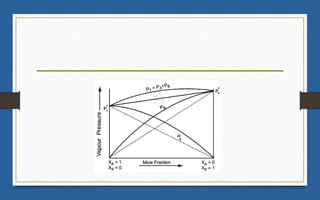
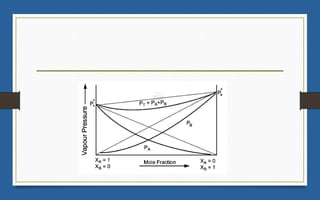
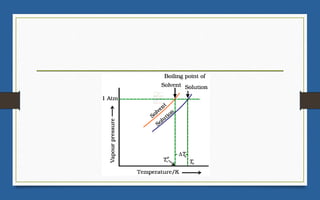
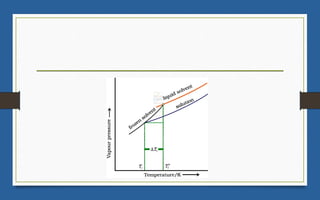
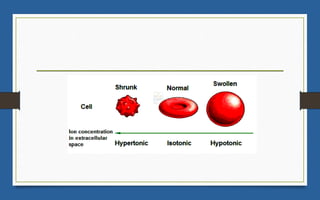
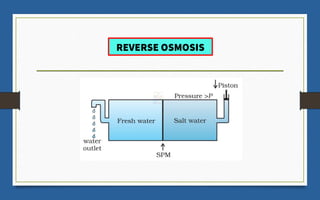
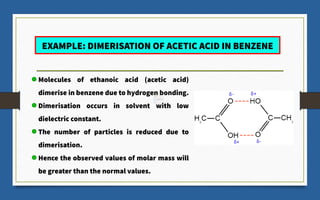
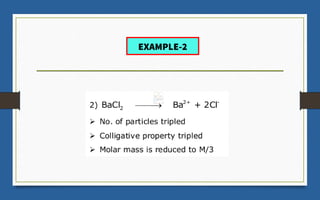
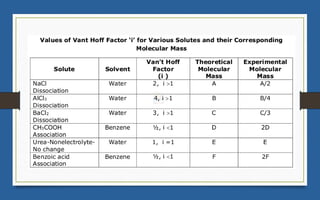
This document discusses various topics related to solutions and colligative properties. It begins by defining a solution as a homogeneous mixture of two or more pure substances whose composition can vary within limits. It then discusses different types of solutions, methods for expressing concentration, and factors that affect solubility. The document also covers Henry's law, Raoult's law, and their applications. Finally, it discusses colligative properties such as lowering of vapor pressure, elevation of boiling point, depression of freezing point, and osmotic pressure. Determination of molar mass using these properties is also described along with some examples.