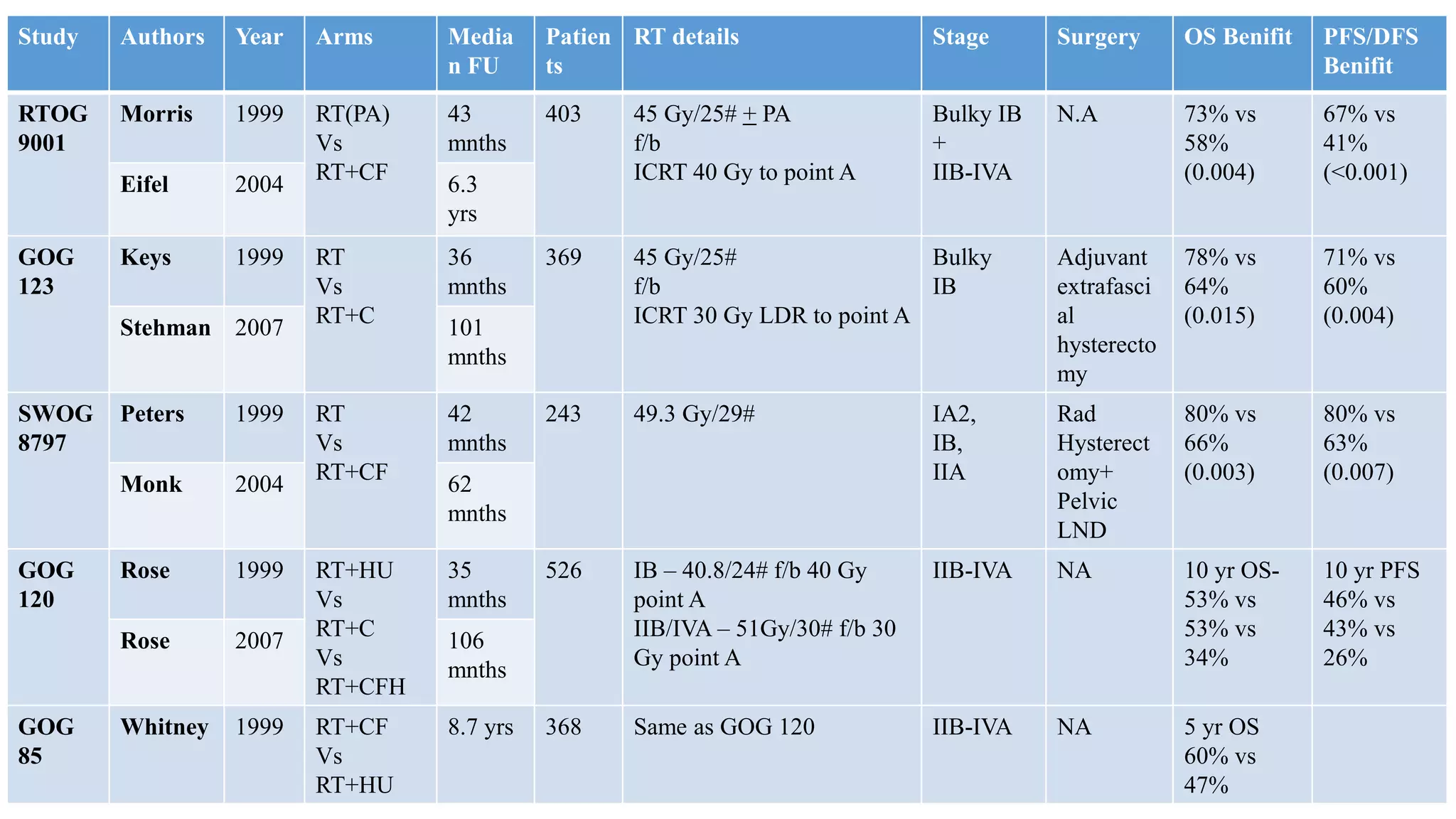
This document summarizes several key studies on the use of concurrent chemo-radiation therapy for carcinoma of the cervix. Five randomized controlled trials from the 1980s-1990s showed significantly improved progression-free and overall survival when cisplatin-based chemo-radiation was used compared to radiation alone. Subsequent larger trials like GOG 120 and RTOG 9001 reinforced these findings. Long term follow up data continued to show survival benefits with acceptable toxicity rates for concurrent chemo-radiation, which is now the standard of care for locally advanced cervical cancer.



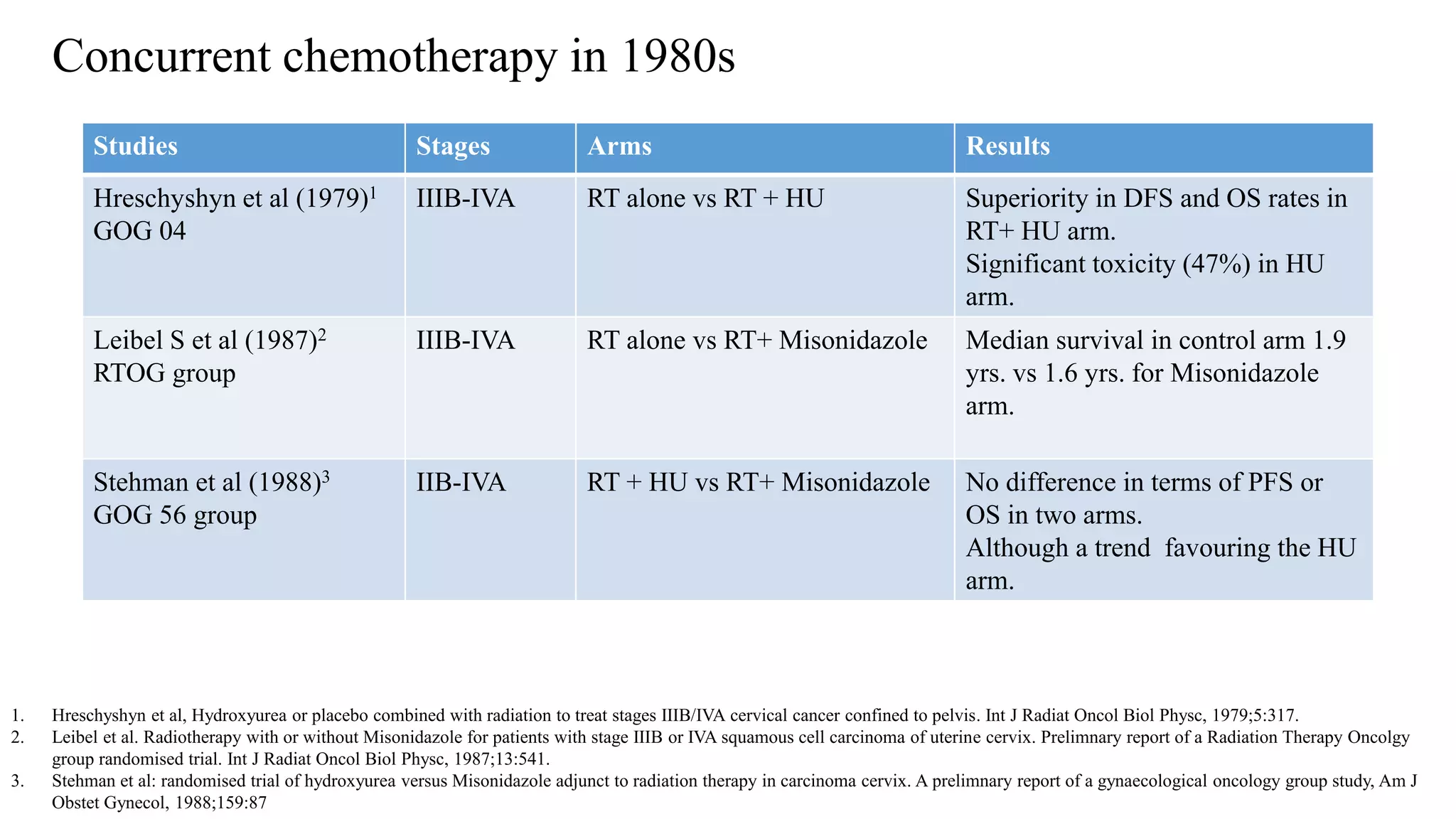
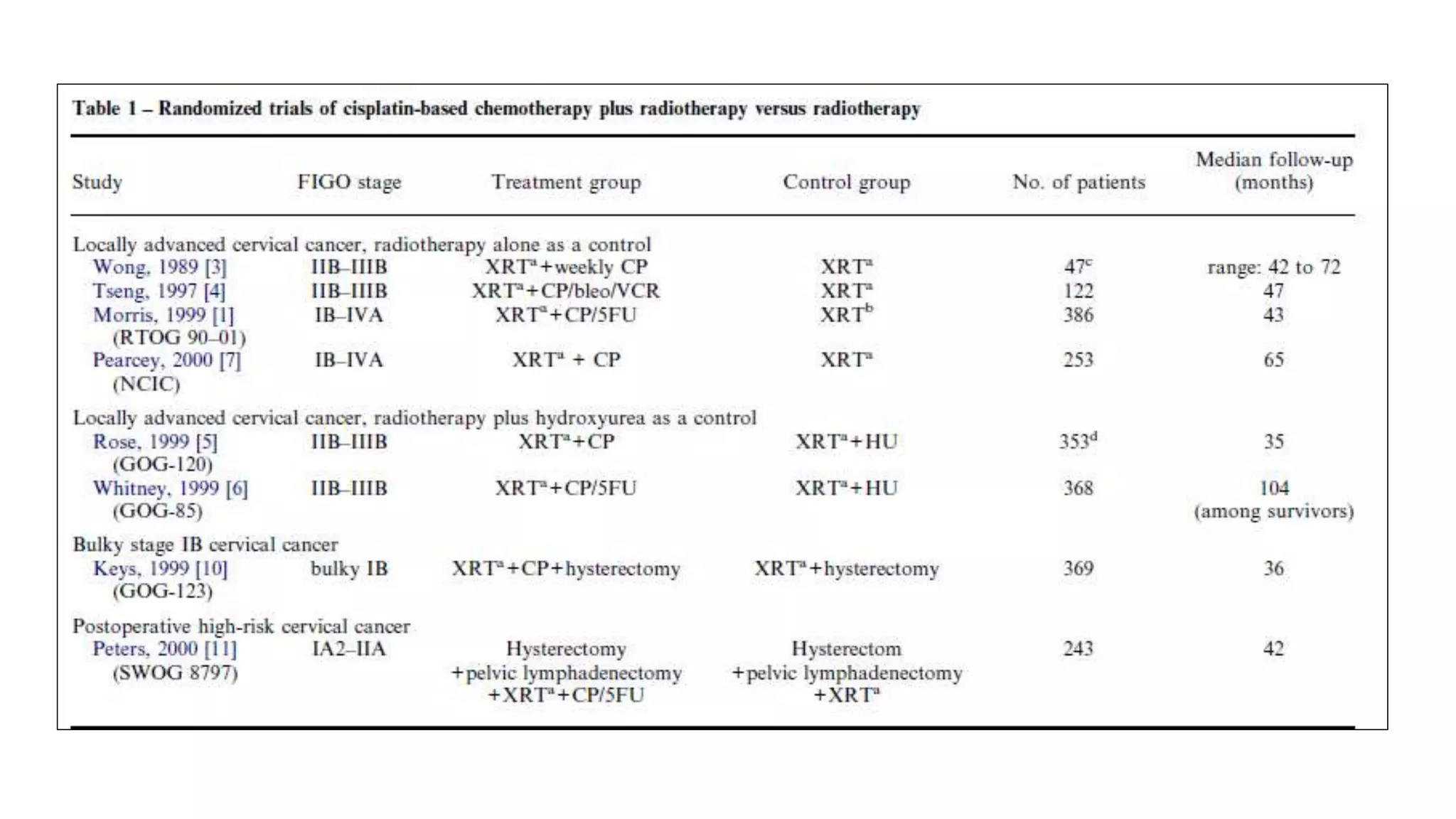
![• A series of five randomized trials in a variety of disease stages matured around then:
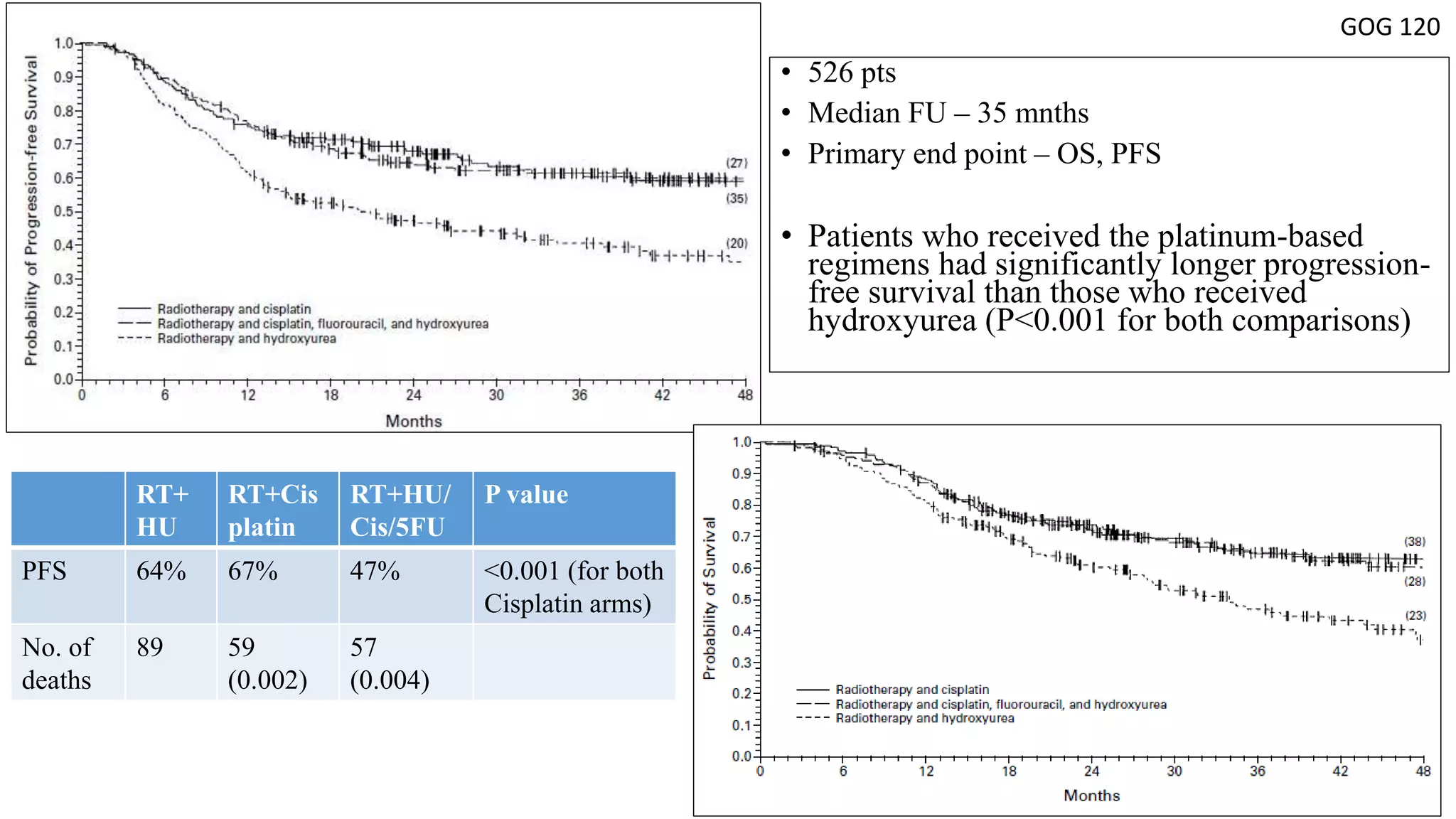
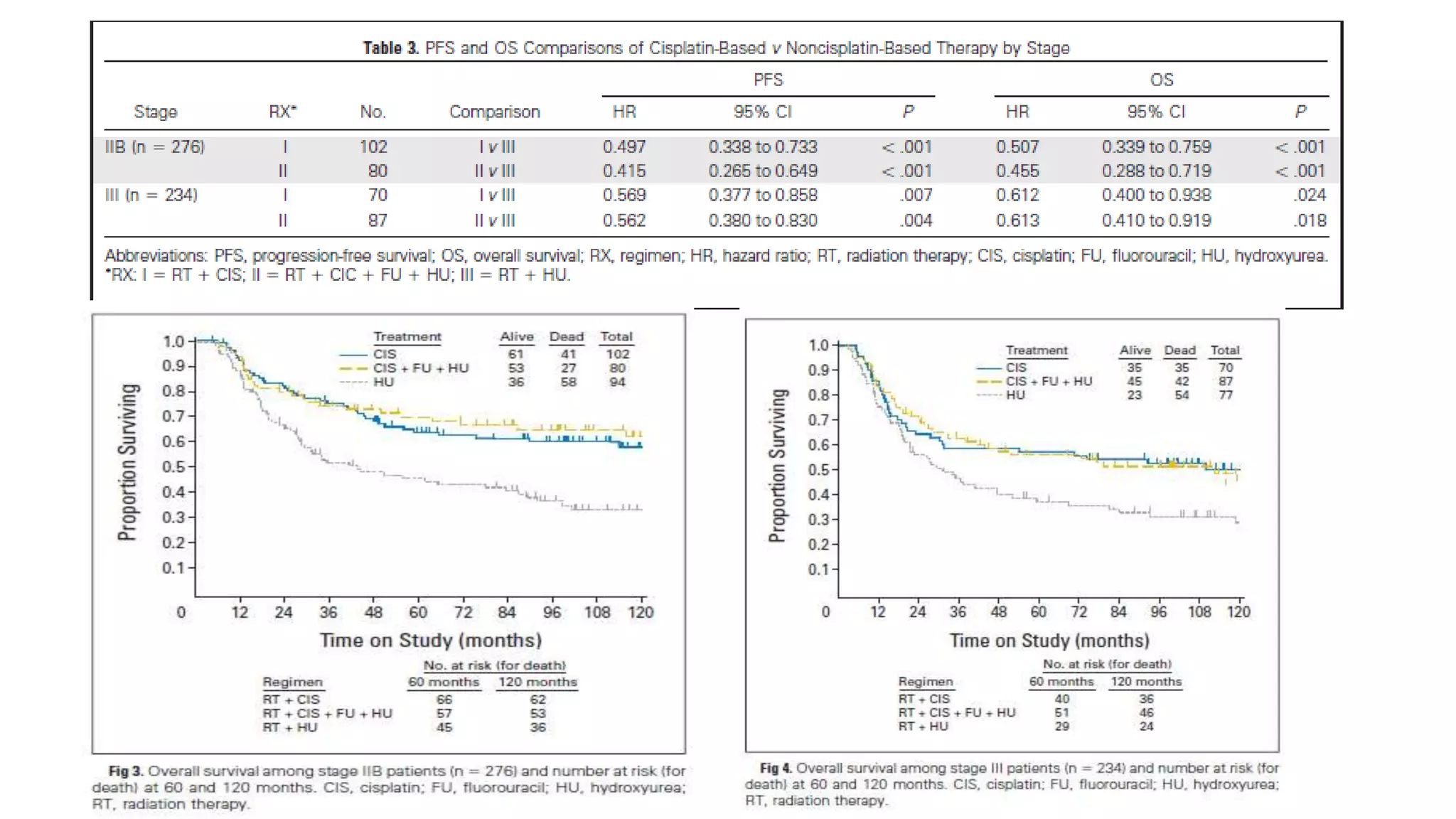
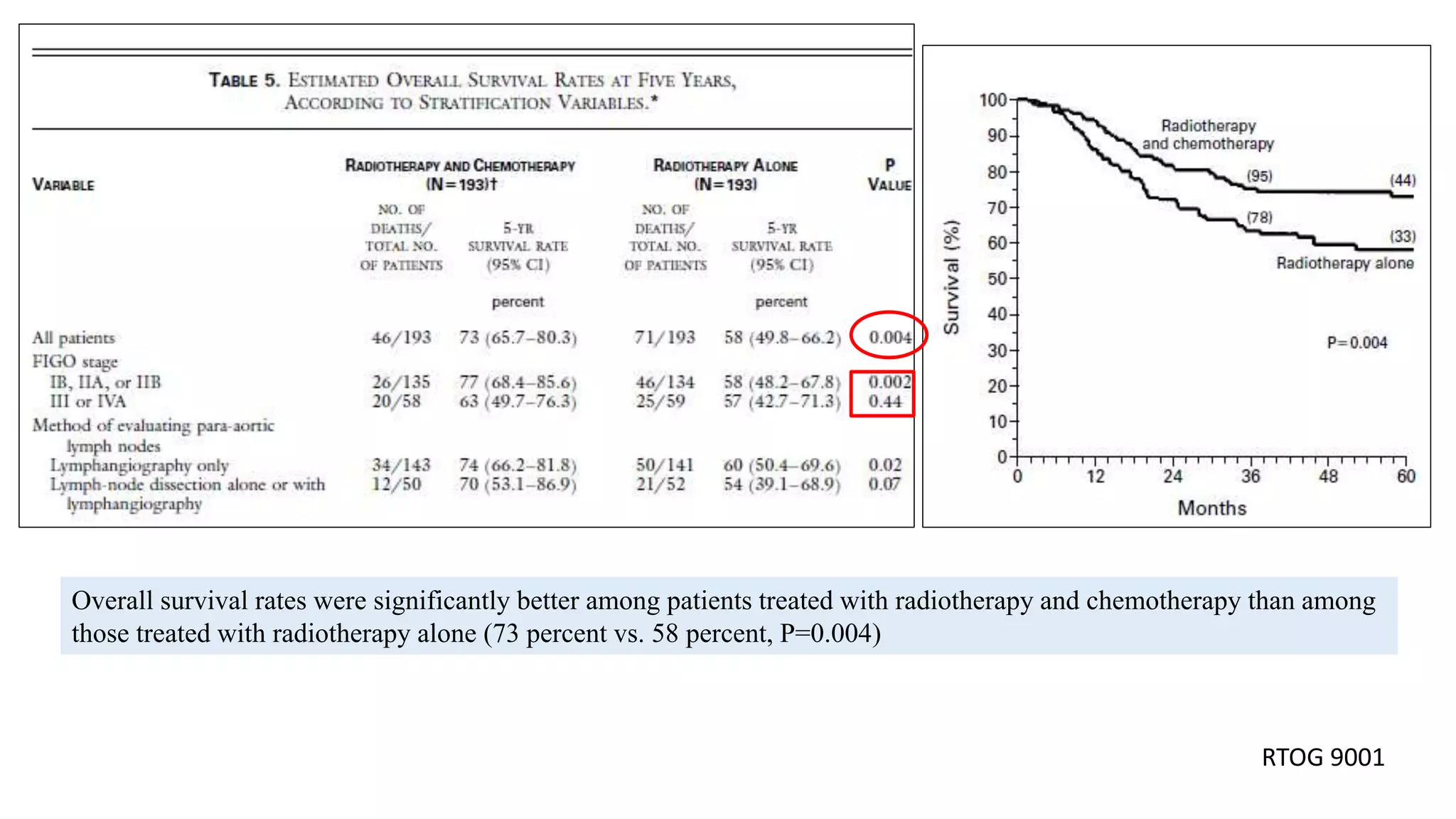
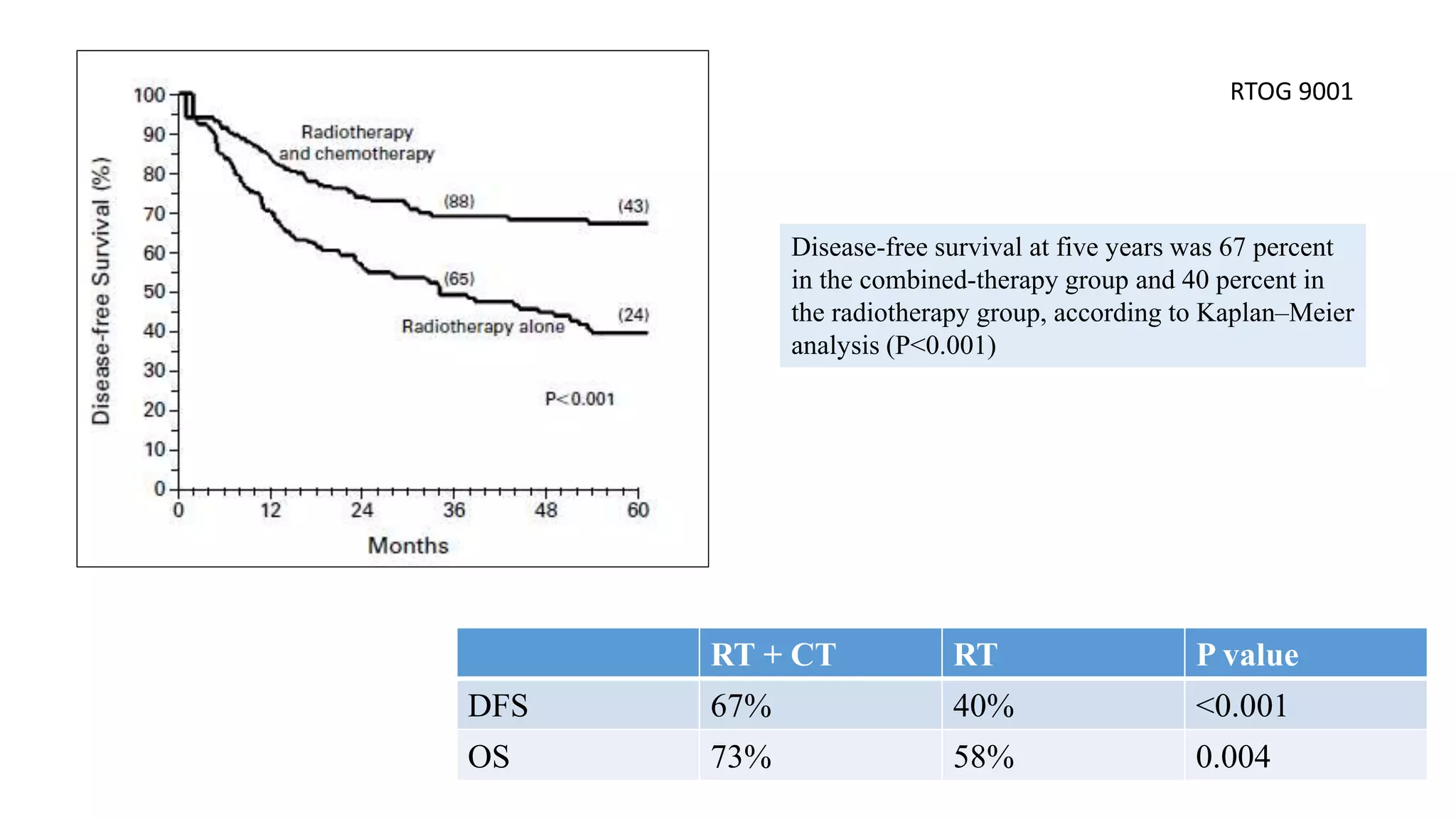
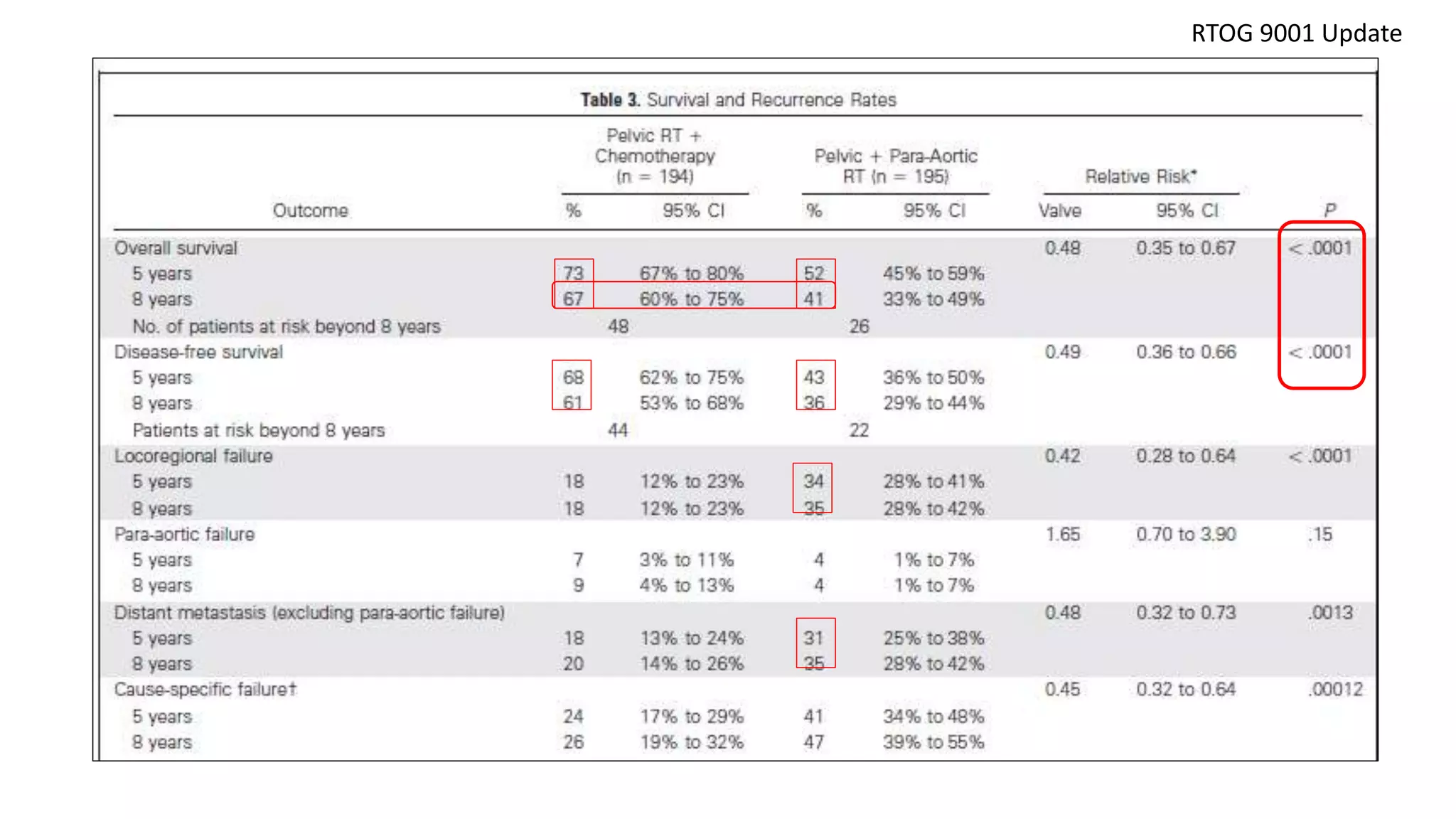
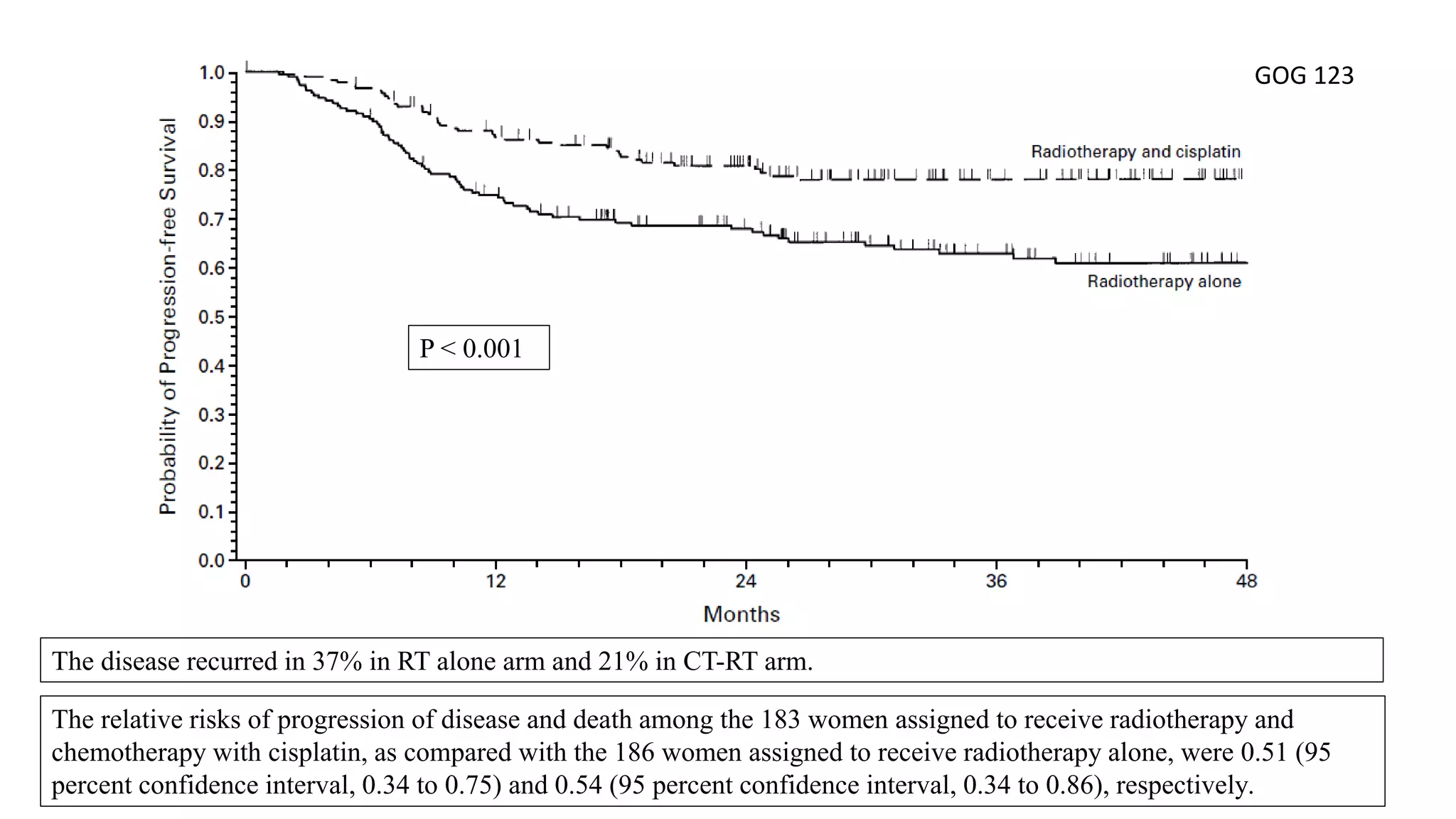
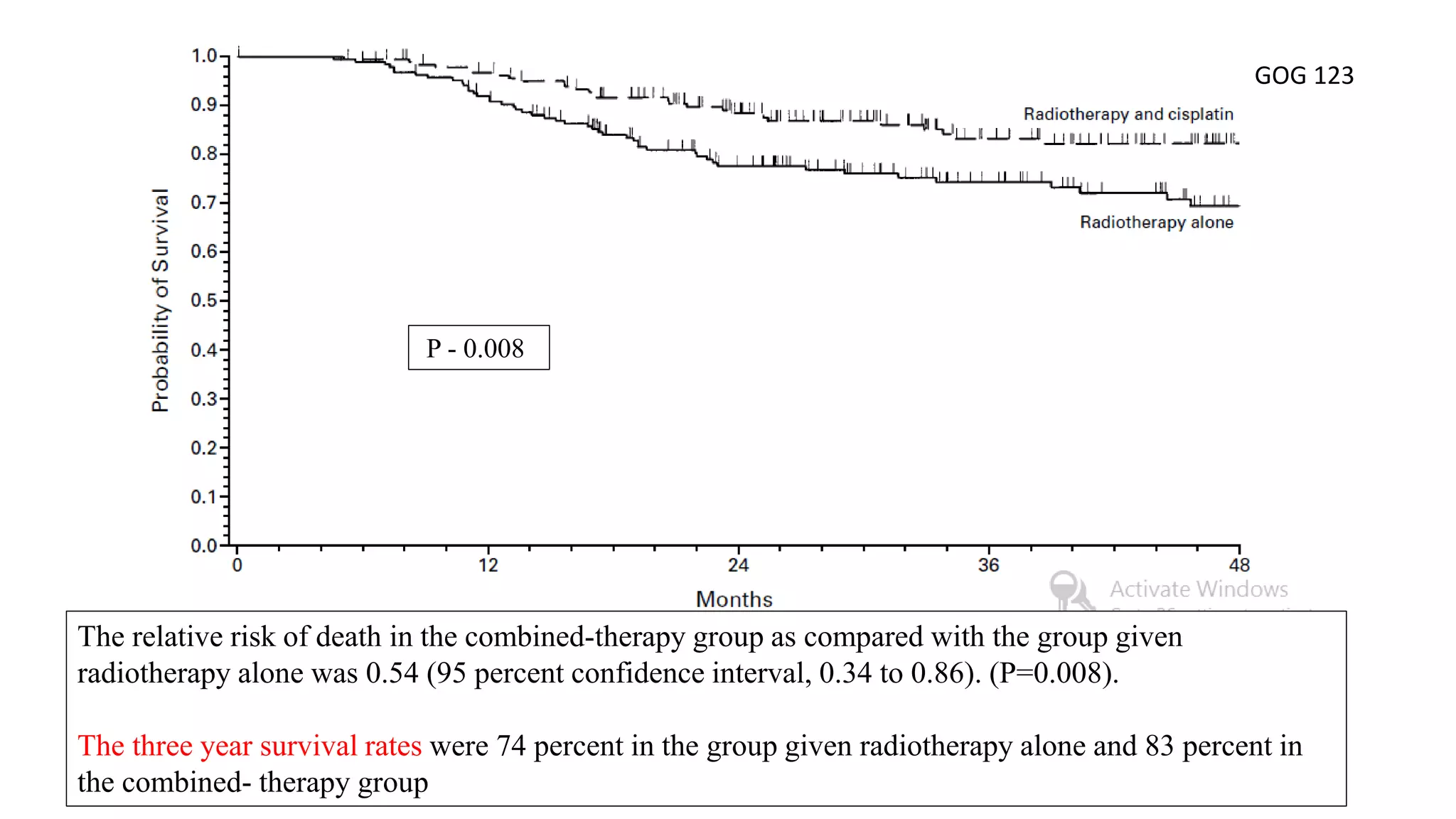
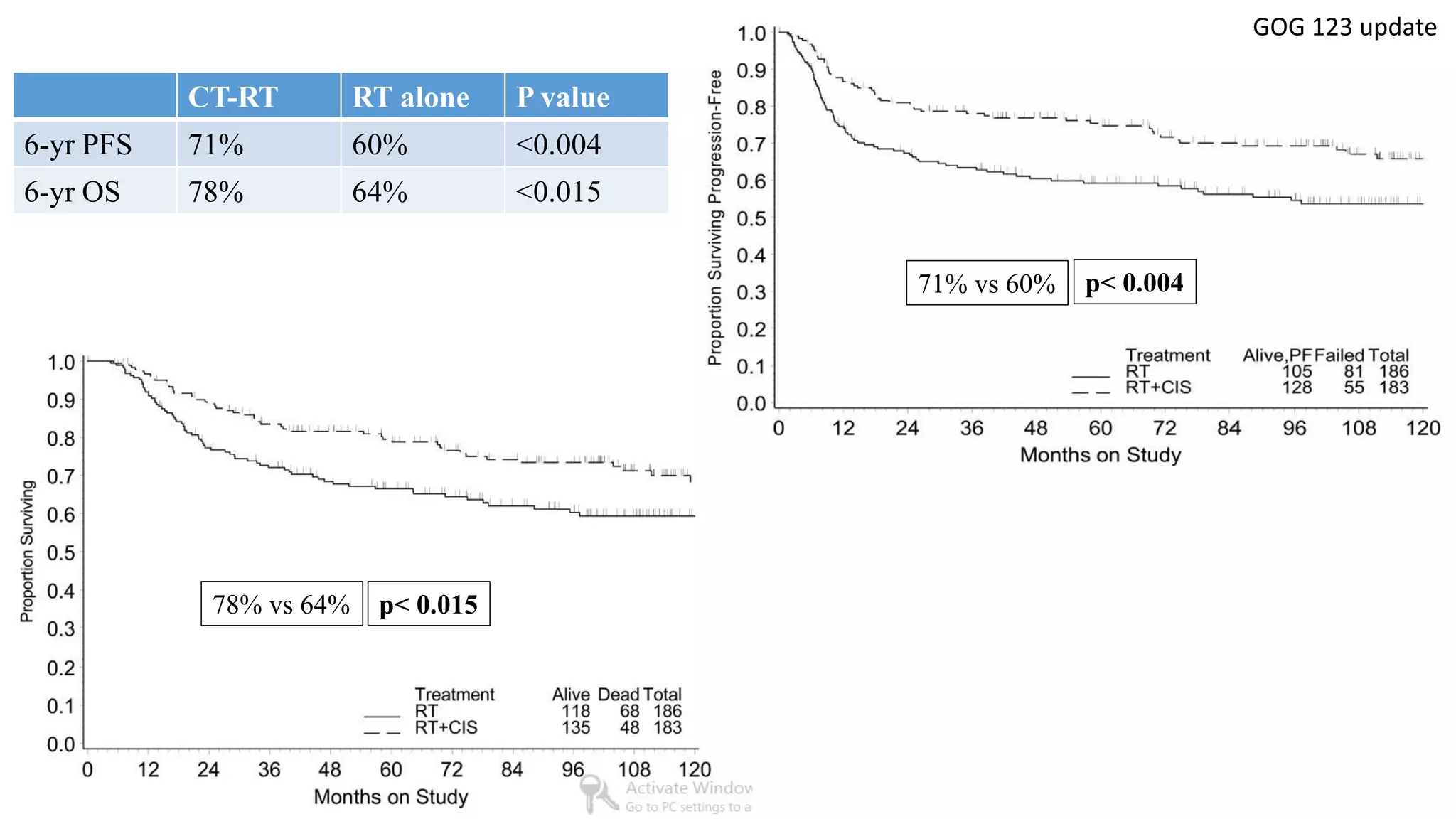
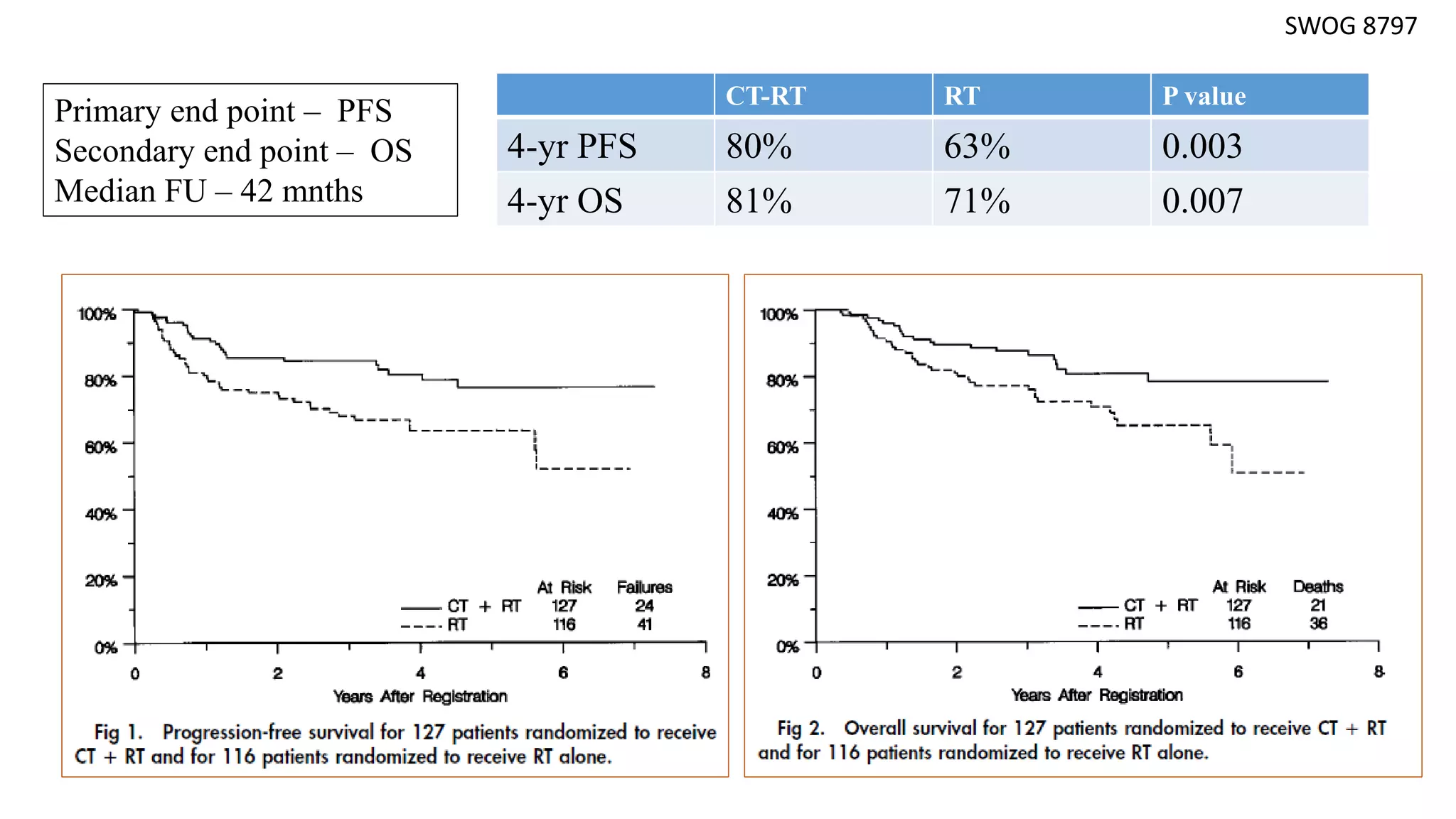
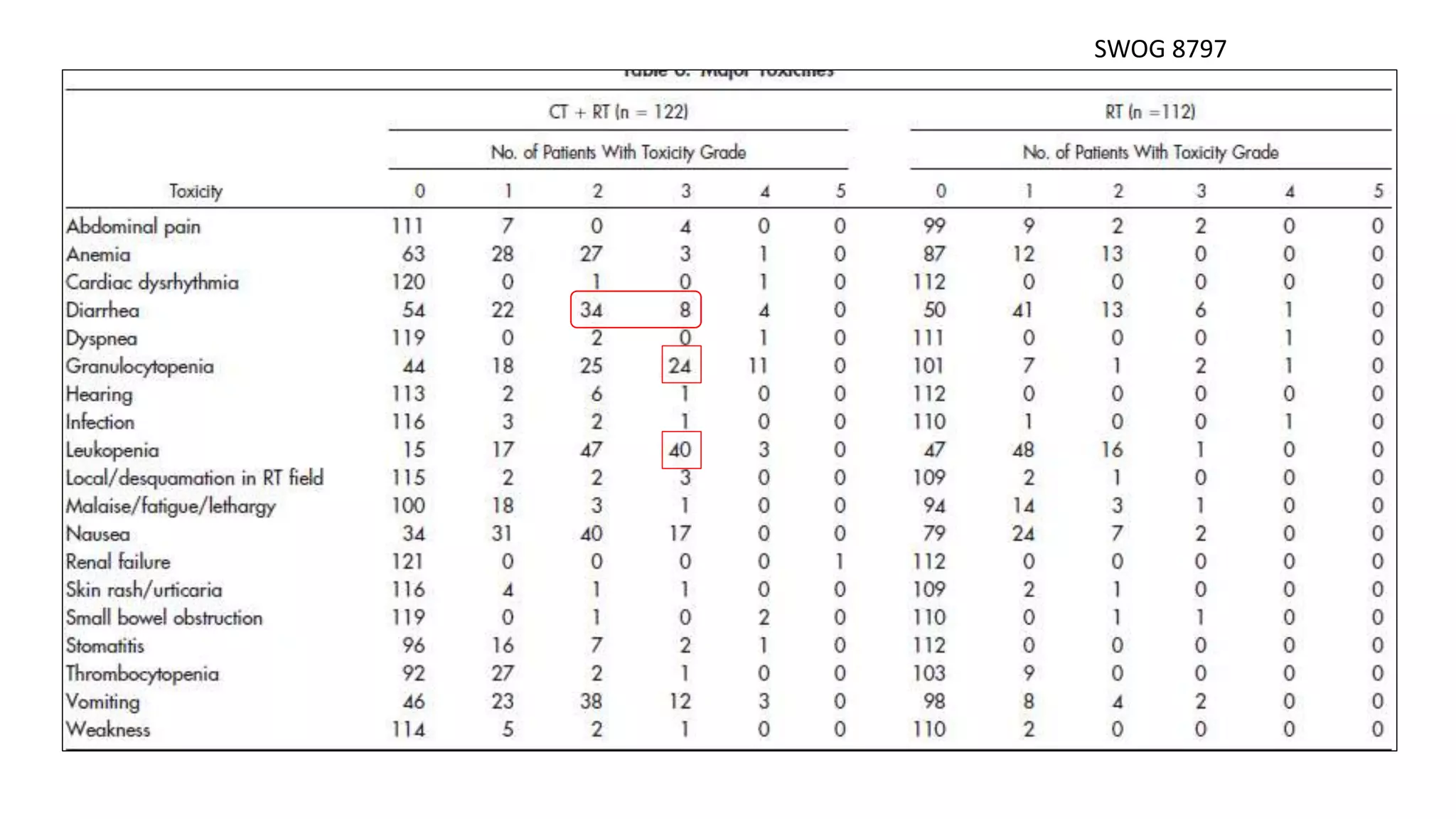
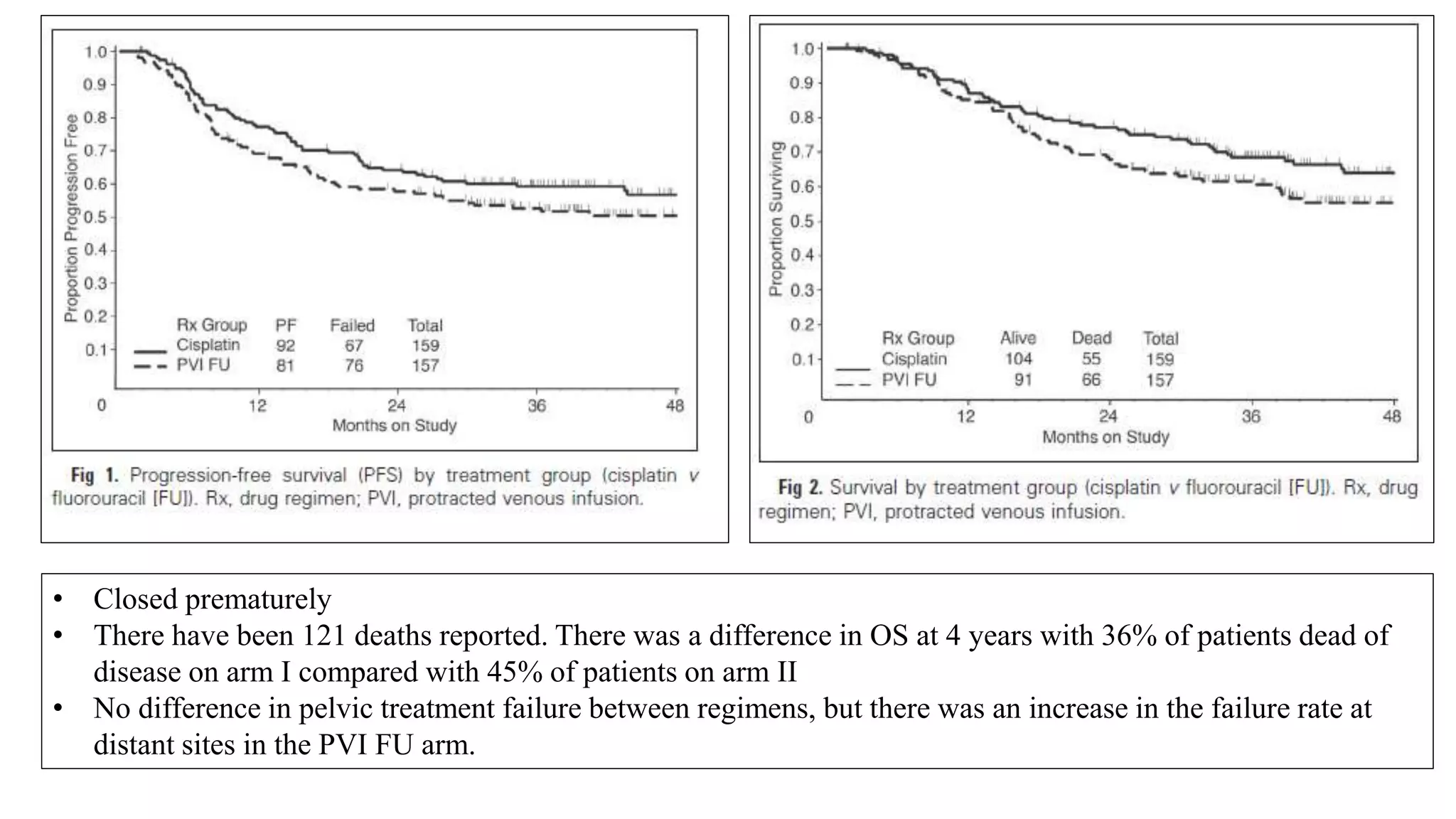
• Collectively, 1894 women were analysed and compared cisplatin based chemoRT to RT
alone (RTOG 9001, GOG 123, SWOG 87-97) and hydroxyurea (GOG 85 and 120). All
showed a significant reduction in the risk of recurrence and death with cisplatin-based
chemoRT.
GOG 85 [1] Whitney et al, JCO 1999
RTOG – 9001 [2] Morris M et al, NEJM 1999
GOG 120 [3] Rose PG et al, NEJM 1999
SWOG 8797/GOG 109 [4] Peters WA et al, Gynecol Oncol 1999
GOG 123 [5] Keys HM et al, NEJM 1999
1. Whitney CW, Sause W, Bundy BN, et al: Randomised comparison of fluorouracil plus cispla- tin versus hydroxyurea in stage IIB/IVA in carcinoma of the cervix. J Clin Oncol 17:1339-
1348, 1999
2. Morris M, Eifel PJ, Lu J, et al: Pelvic radiation with concurrent chemotherapy compared with plevic and para-aortic radiation for high-risk cervical cancer. N Engl J Med 340:1137-1143,
1999
3. Rose PG, Bundy BN, Watkins EB, et al: Con- current cisplatin-based radiotherapy and chemother- apy for locally advanced cervical cancer. N Engl J Med 340:1144-1153, 1999
4. Peters WA, Liu PY, Barrett RGW, et al: Cis- platin, 5-Fluorouracil plus radiation therapy are supe- rior to radiation therapy as adjunctive therapy in high risk, early stage carcinoma of the
cervix after radical hysterectomy and pelvic lymphadenectomy: Report of a Phase III inter group study. Presented at Soc Gynecol Oncol 30th Annual Meeting, San Fransisco, CA, February
5-9, 1999
5. Keys HM, Bundy BN, Stehman FB, et al: Cisplatin, radiation and adjuvant hysterectomy com- pared with radiation and adjuvant hysterectomy for bulky stage IB cervical carcinoma. N
Engl J Med 340:1154-1161, 1999](https://image.slidesharecdn.com/satyajeetcervixconcurrentchemo-radiotherapy-161219172653/75/Satyajeet-cervix-concurrent-chemo-radiotherapy-6-2048.jpg)

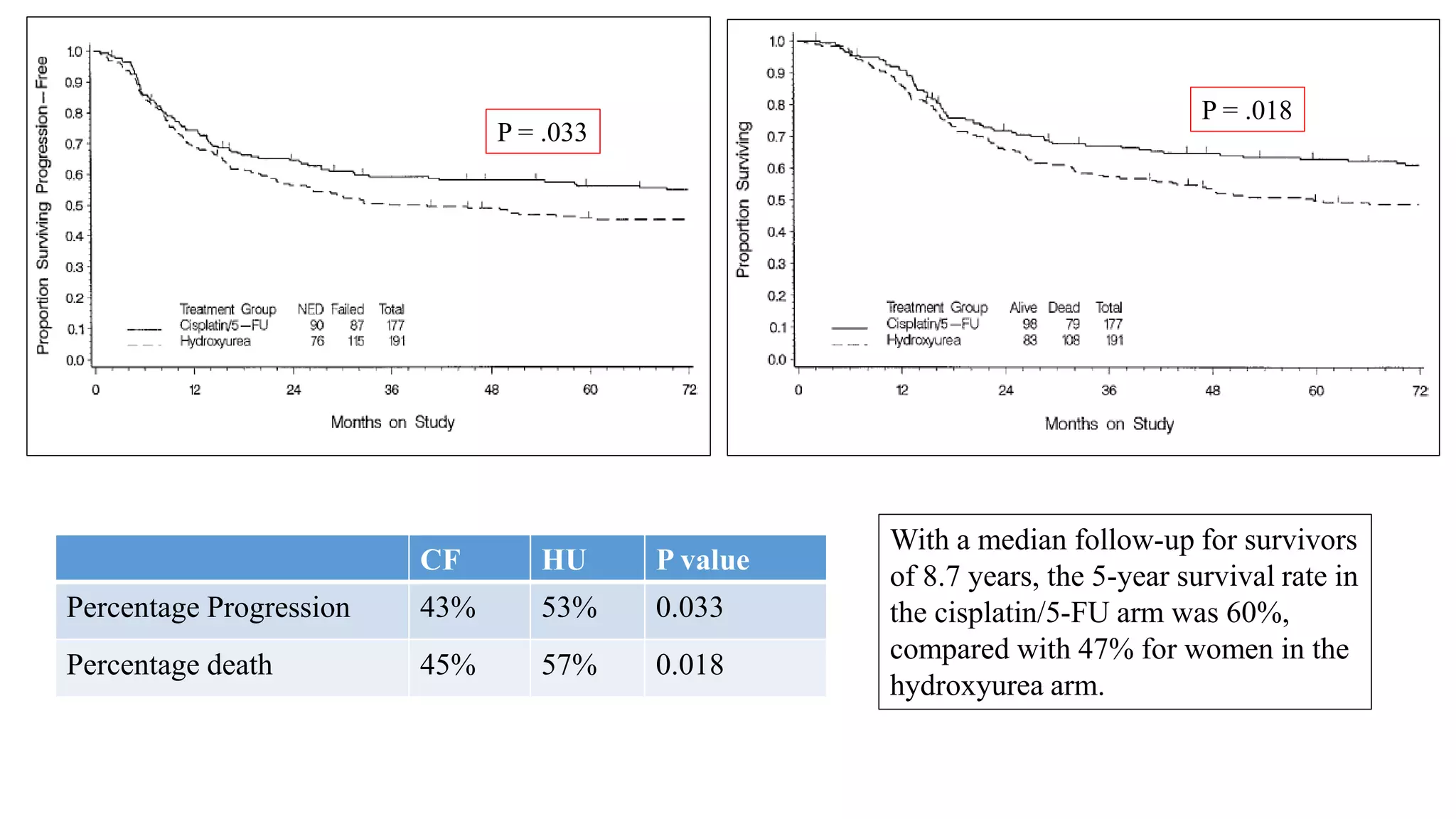
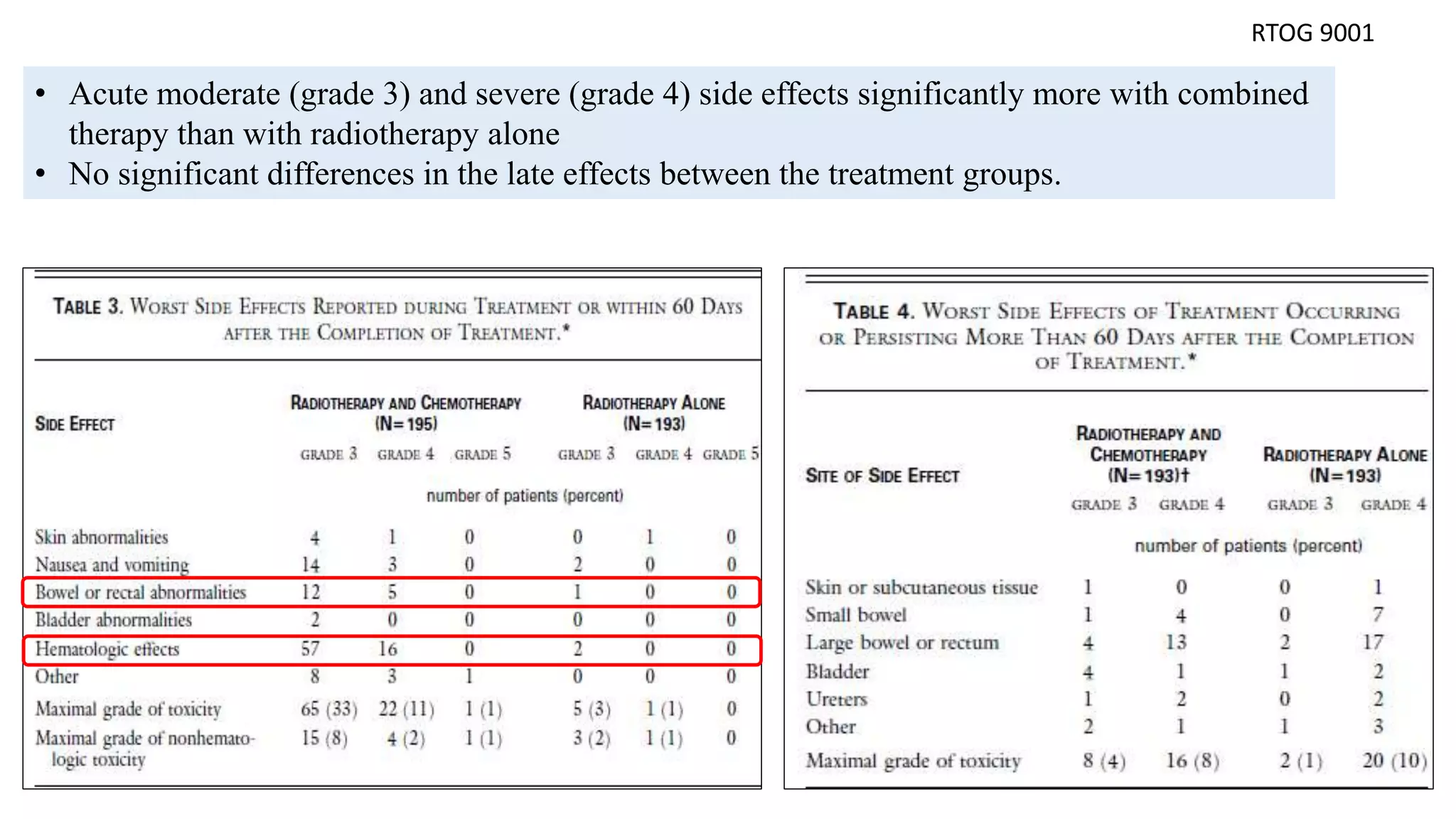
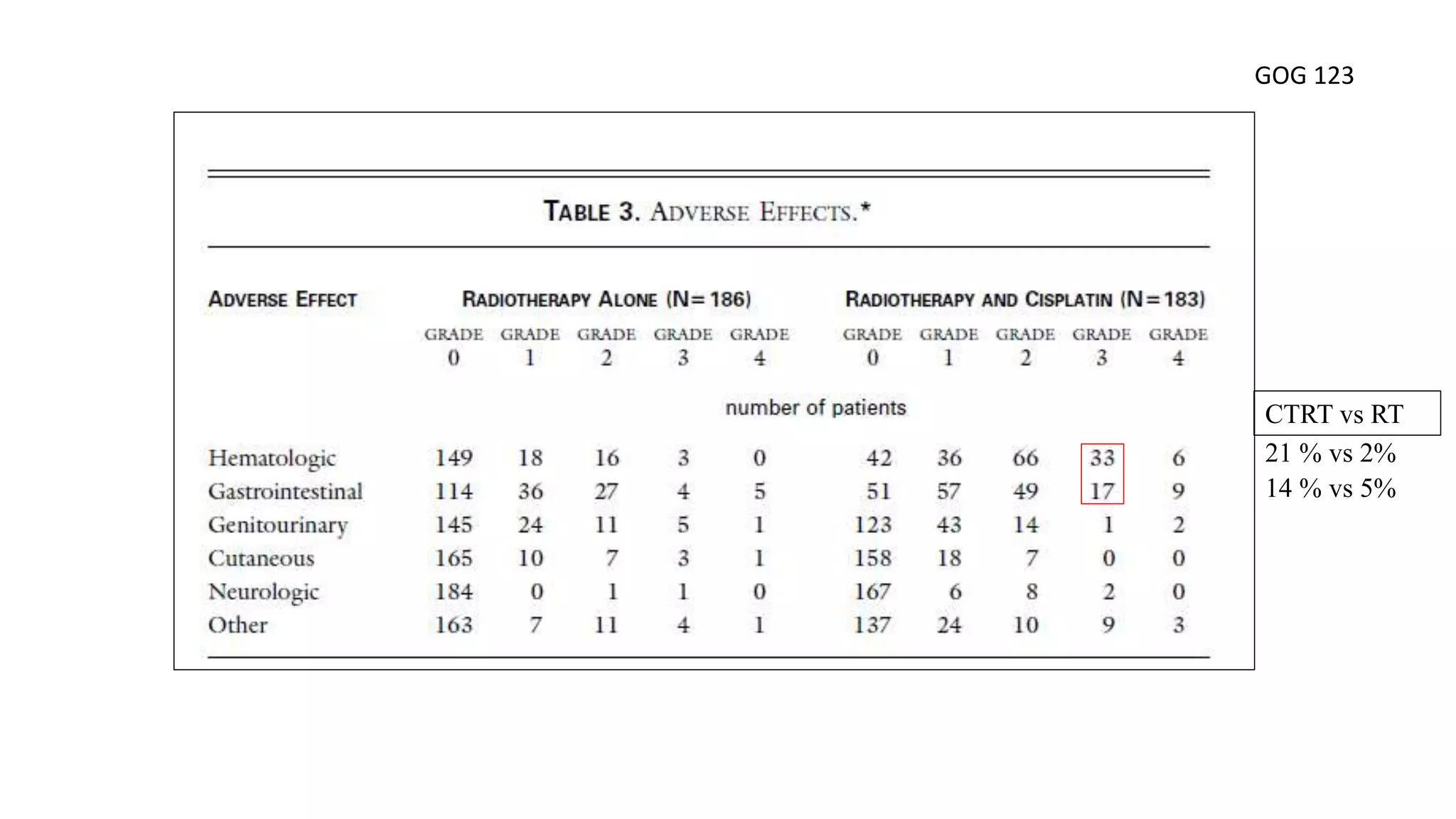
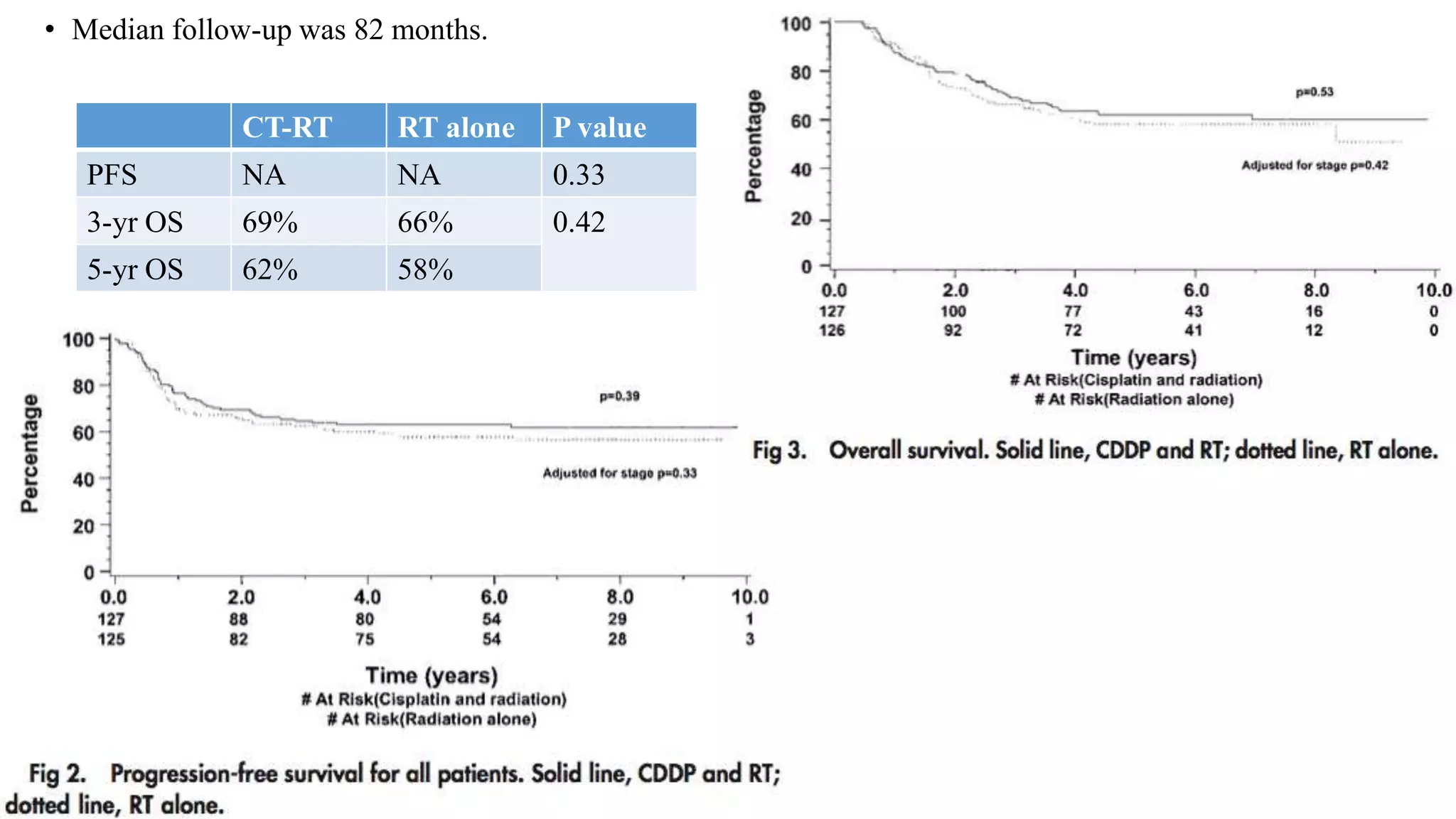
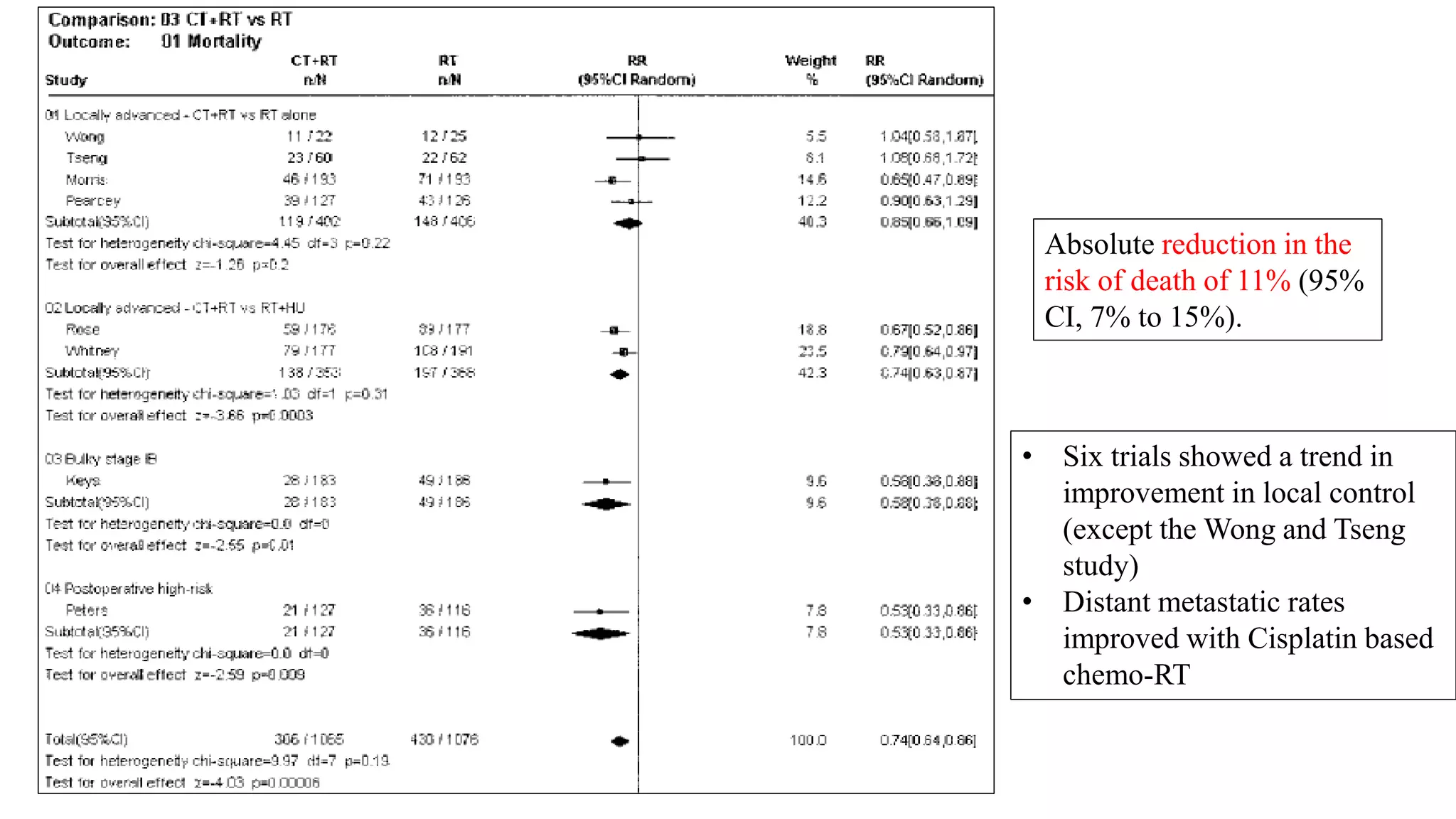
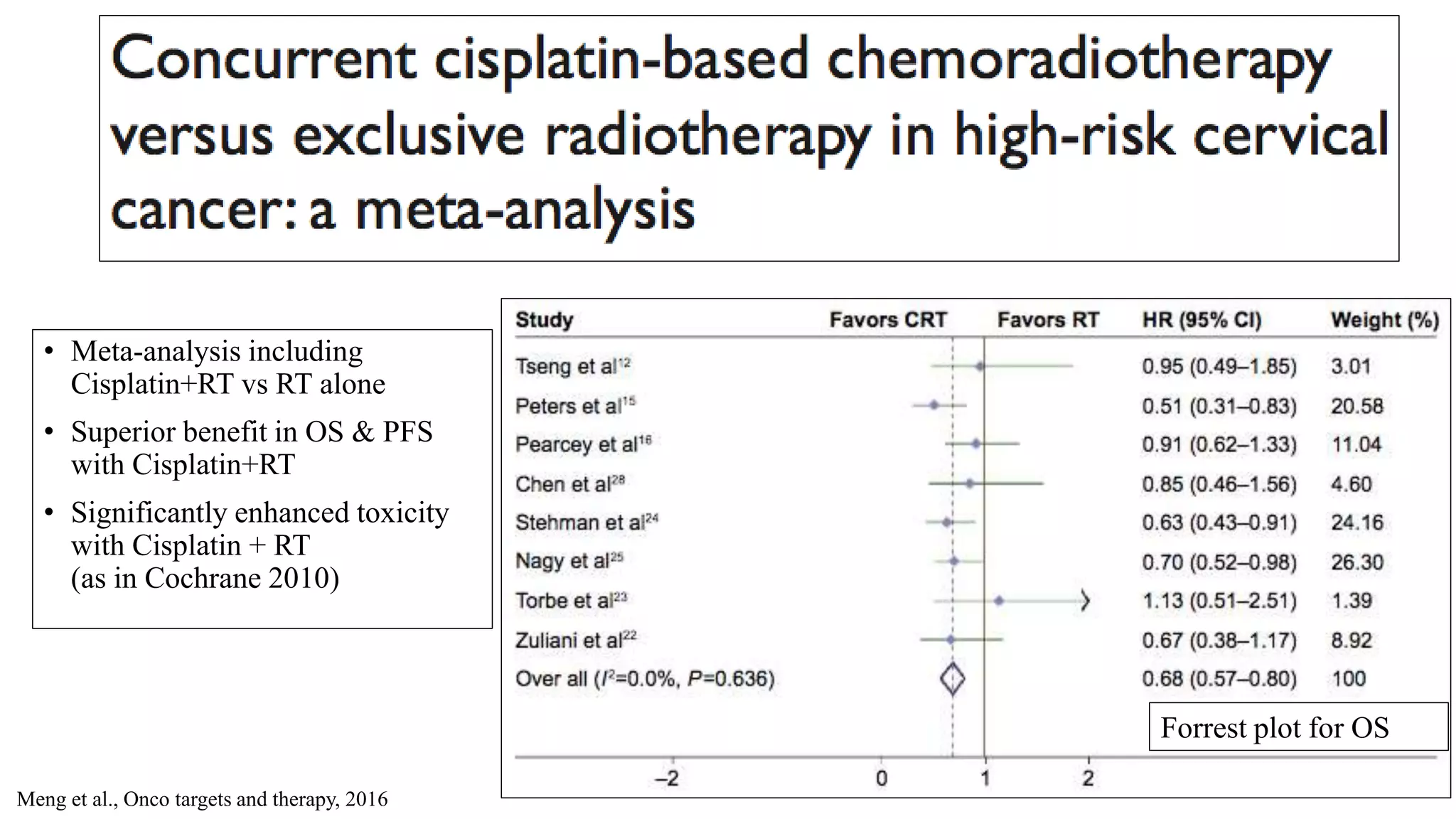
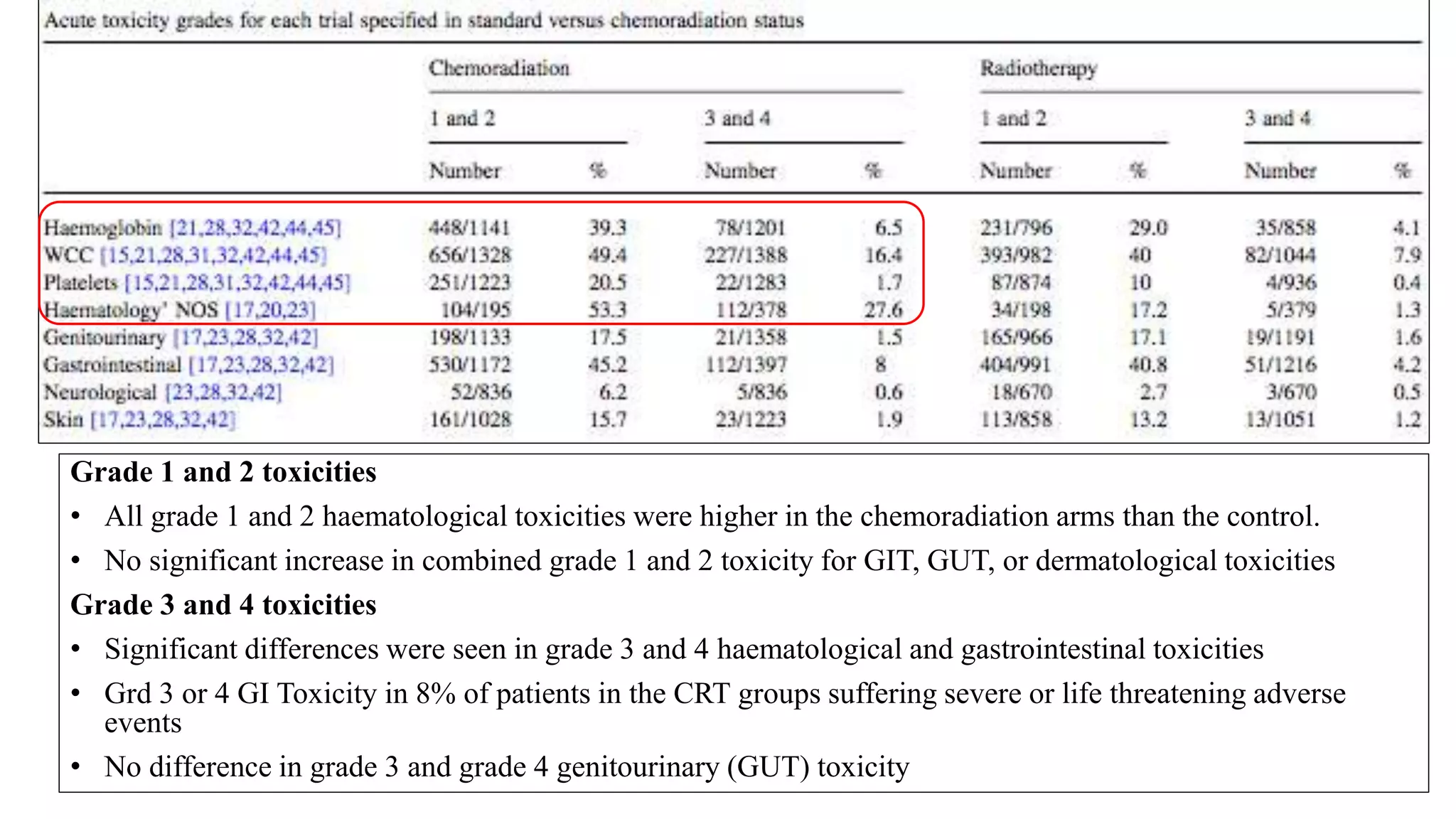
![Adverse effects
• predominately hematologic or gastrointestinal in both treatment groups.
• Severe life threatening Leukopenia was more common in the HU regimen (P , .00001).
[only six CF patients (4%) compared with 46 HU patients (24%)].
• Grade 3 or 4 gastrointestinal toxicity was slightly more common (not statistically significant)
for patients randomized to CF (8%) than for the HU group (4%).
• The late major complications rate (grade 3 and grade 4) was 16.2% at 3 yr for CF group and
16.5% at 3 yr for HU group.
GOG 85
Staged as IIB - receive 40.8-Gy/24 fraction EBRT to the whole pelvis, 40 Gy was to be delivered to
point A via one or two intracavitary applications (tandem and colpostats) of radium or its equivalent. If
necessary, a parametrial boost was given to bring them point-B dose to 55 Gy.
Staged as III or IVA - receive 51 Gy in 30 fractions, Point A received 30 Gy from one or two
Intracavitary implants. Point B received 60 Gy from both sources with or without a parametrial boost.](https://image.slidesharecdn.com/satyajeetcervixconcurrentchemo-radiotherapy-161219172653/75/Satyajeet-cervix-concurrent-chemo-radiotherapy-9-2048.jpg)















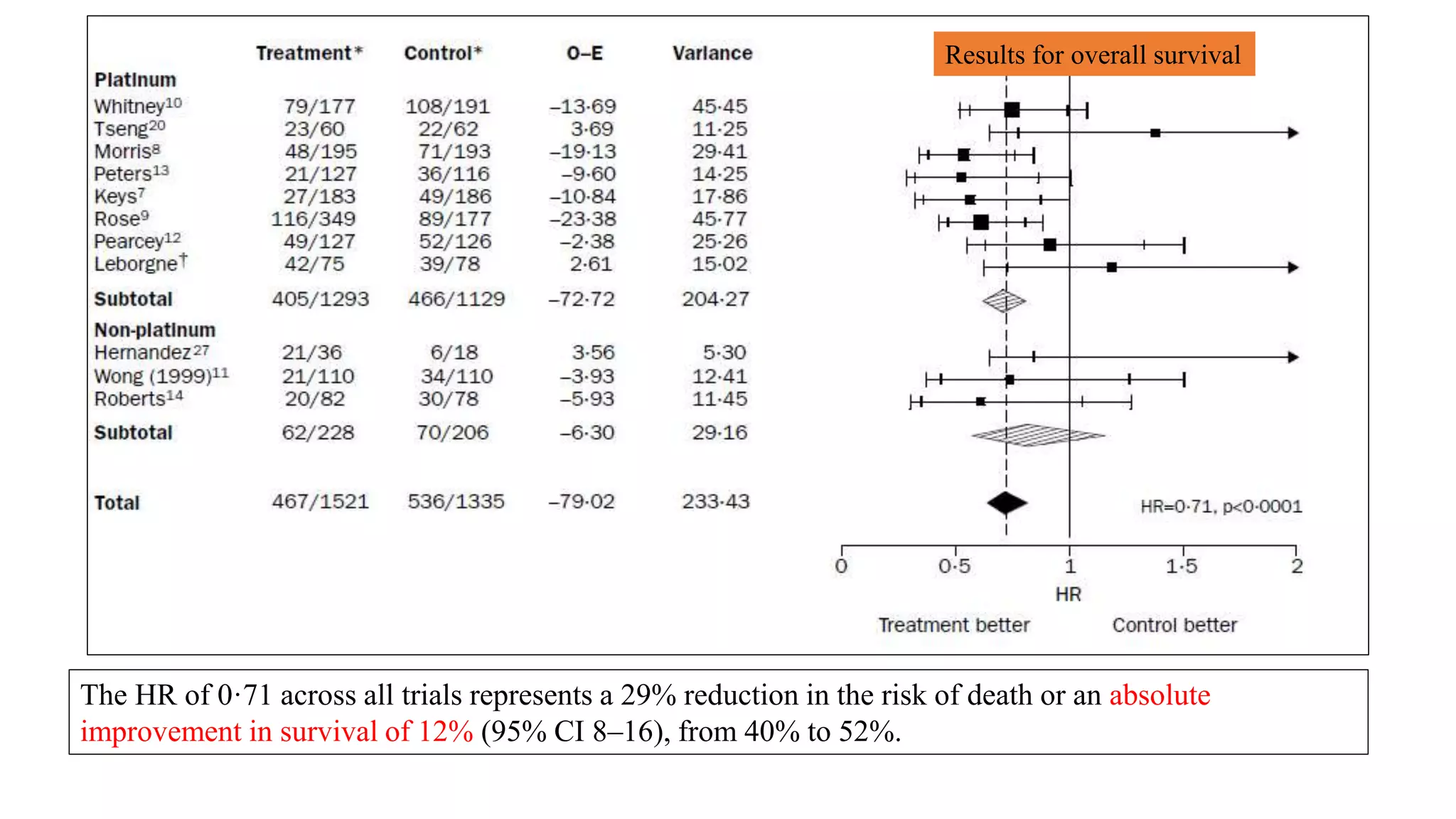

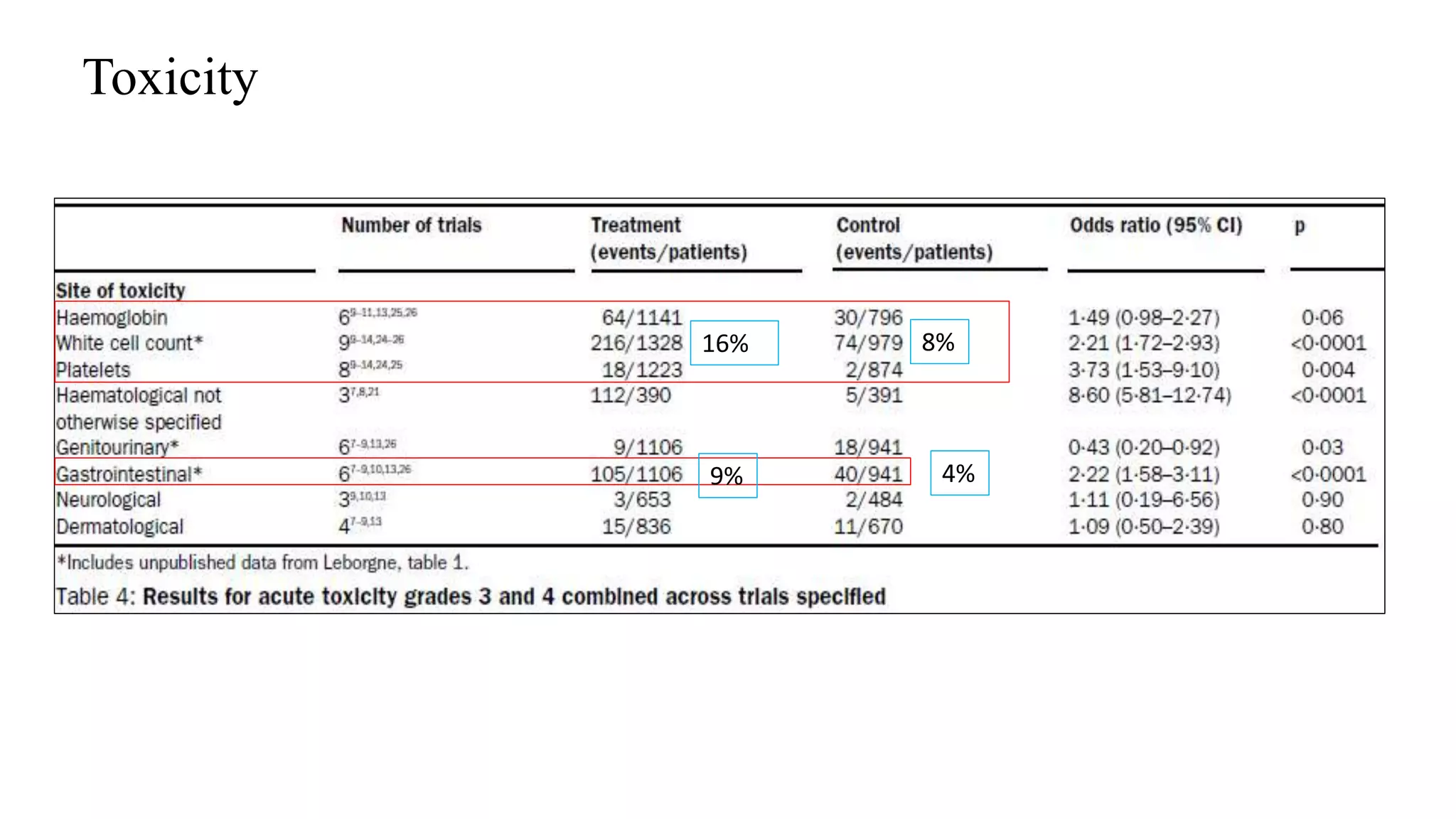
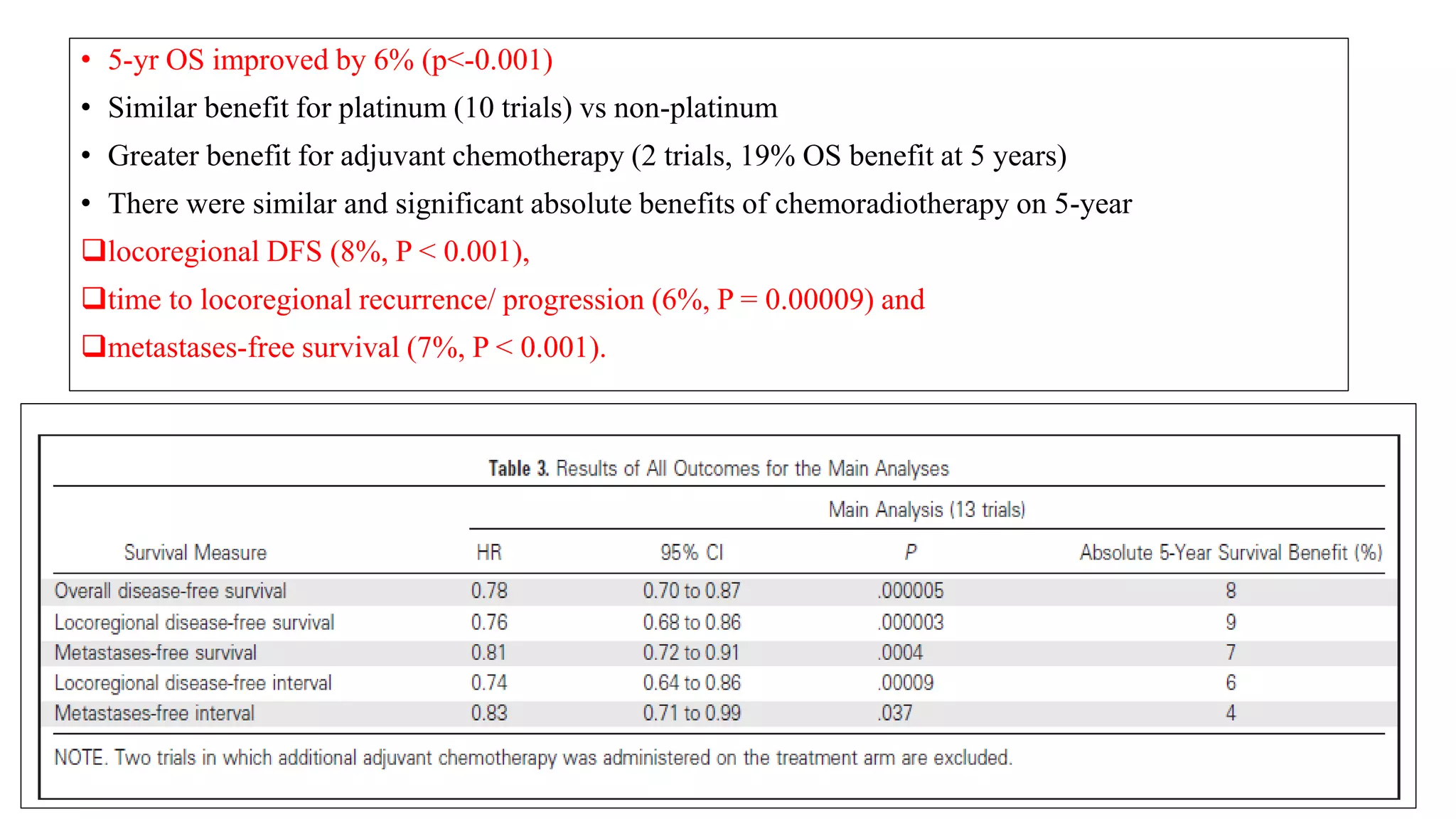
![Conclusions :
• Chemoradiation improves overall survival (hazard ratio 0·71, p<0·0001)
[whether platinum was used (0·70, p<0·0001) or not (0·81, p=0·20).]
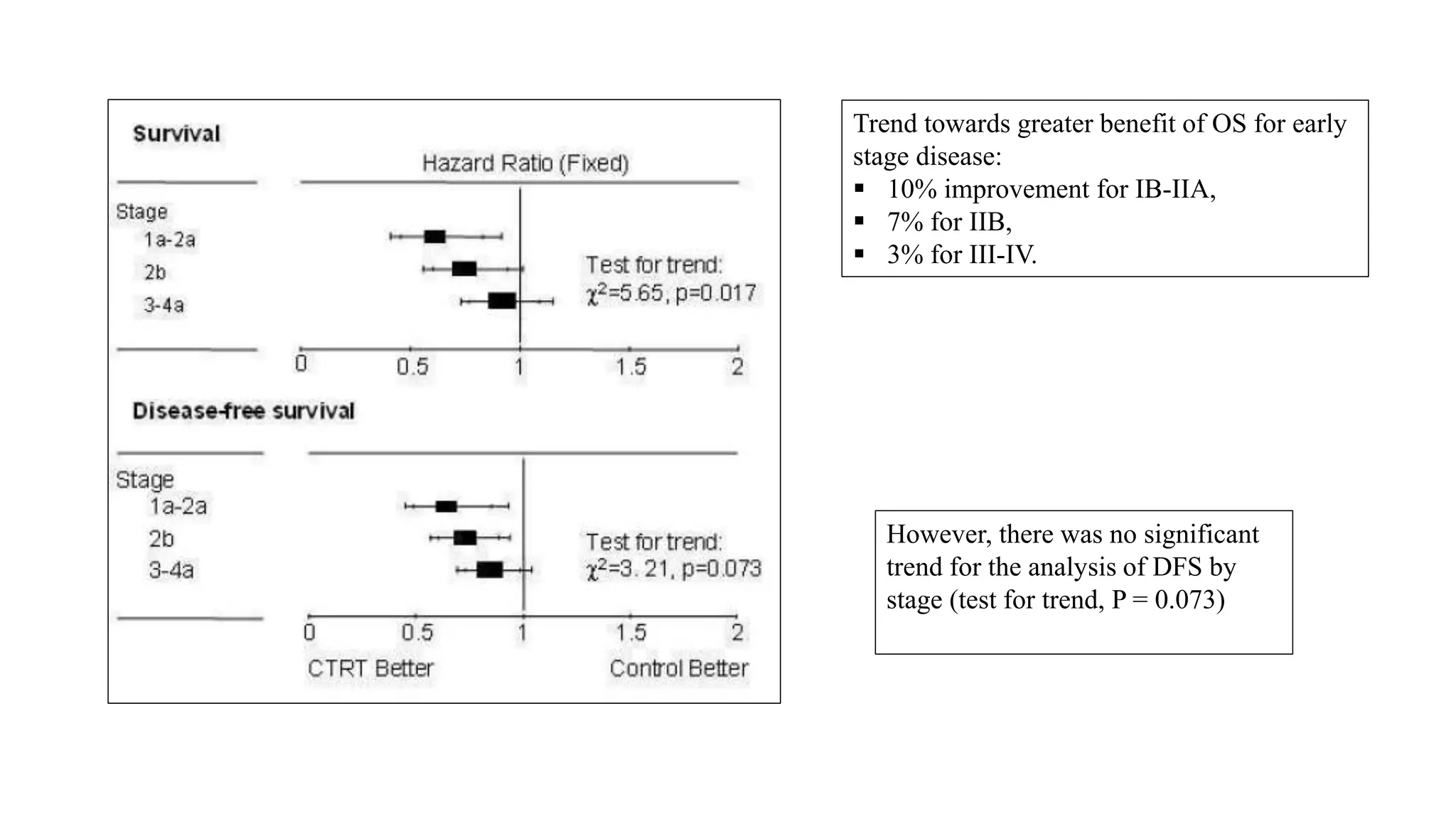
• Greater beneficial effect was seen in trials that included a high proportion of stage I and II patients
(p=0·009).
• Absolute benefit
• PFS – 16%
• OS – 12%
• A significant benefit of chemoradiation on both
• Local (p<0·0001) and
• Distant recurrence (p<0·0001)
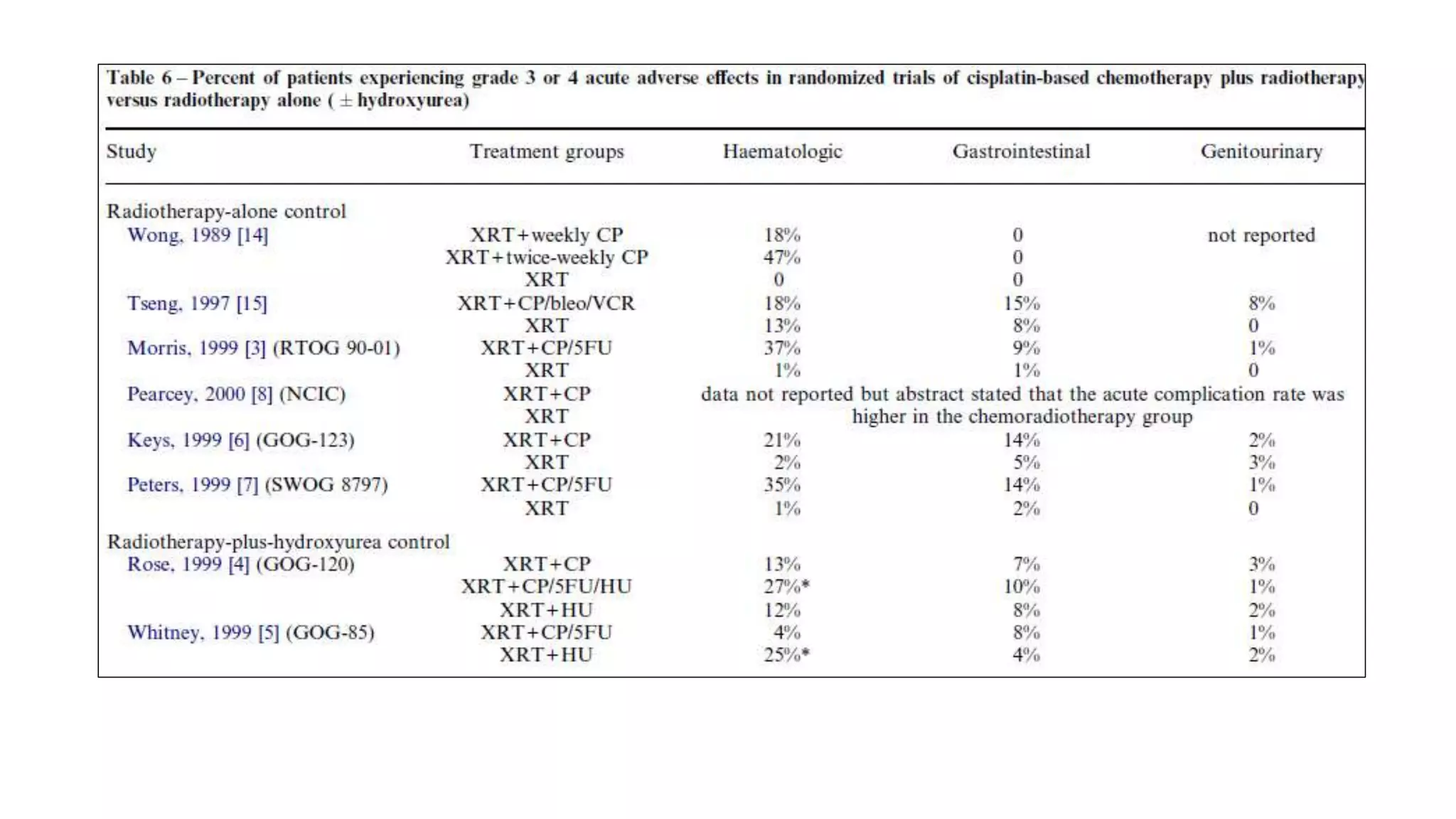
• Grade 3 or 4 haematological and gastrointestinal toxicities were significantly greater in the
concomitant chemoradiation group than the control group.
• There was insufficient data to establish whether late toxicity was increased in the concomitant
chemoradiation group.](https://image.slidesharecdn.com/satyajeetcervixconcurrentchemo-radiotherapy-161219172653/75/Satyajeet-cervix-concurrent-chemo-radiotherapy-42-2048.jpg)










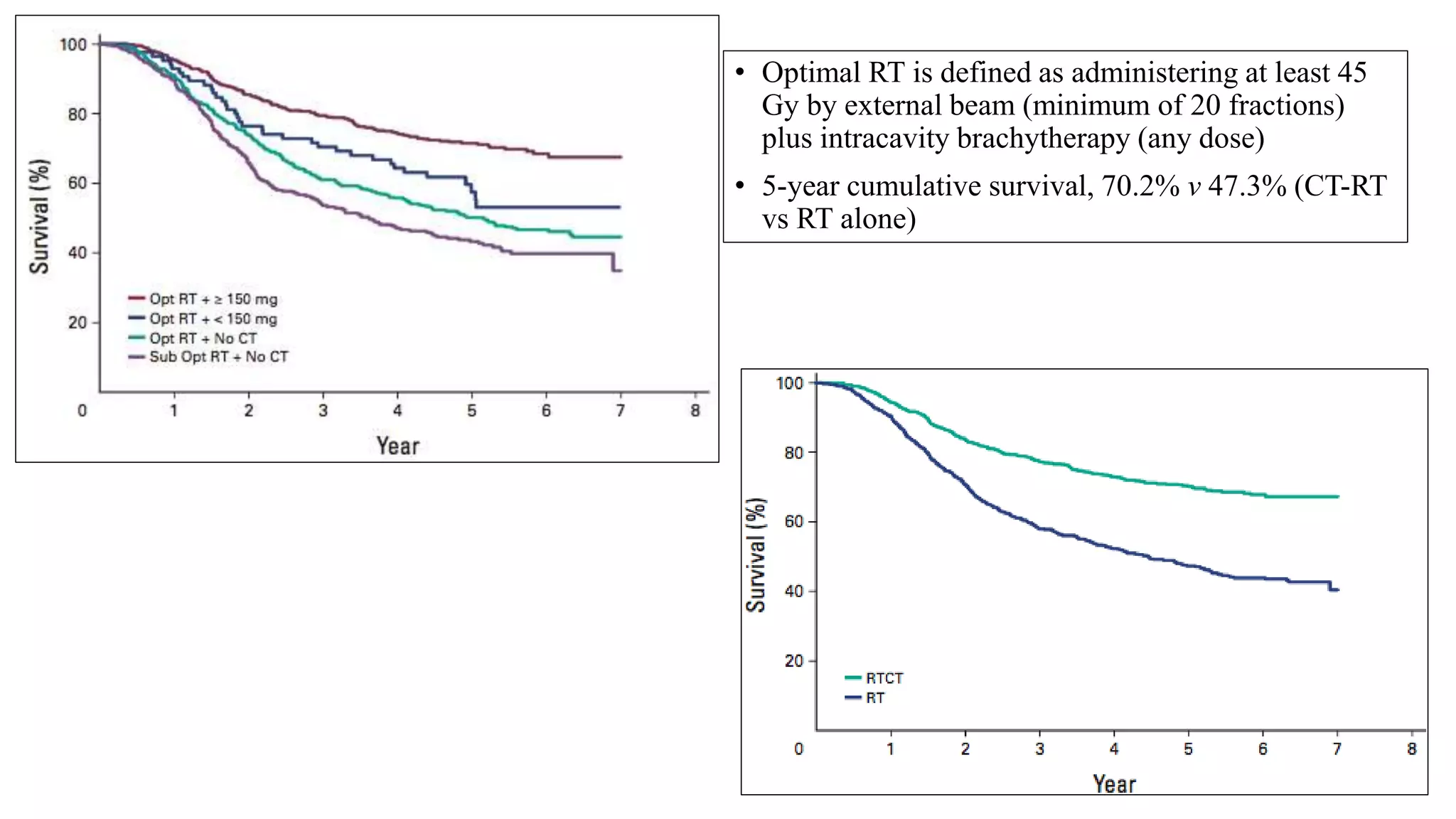


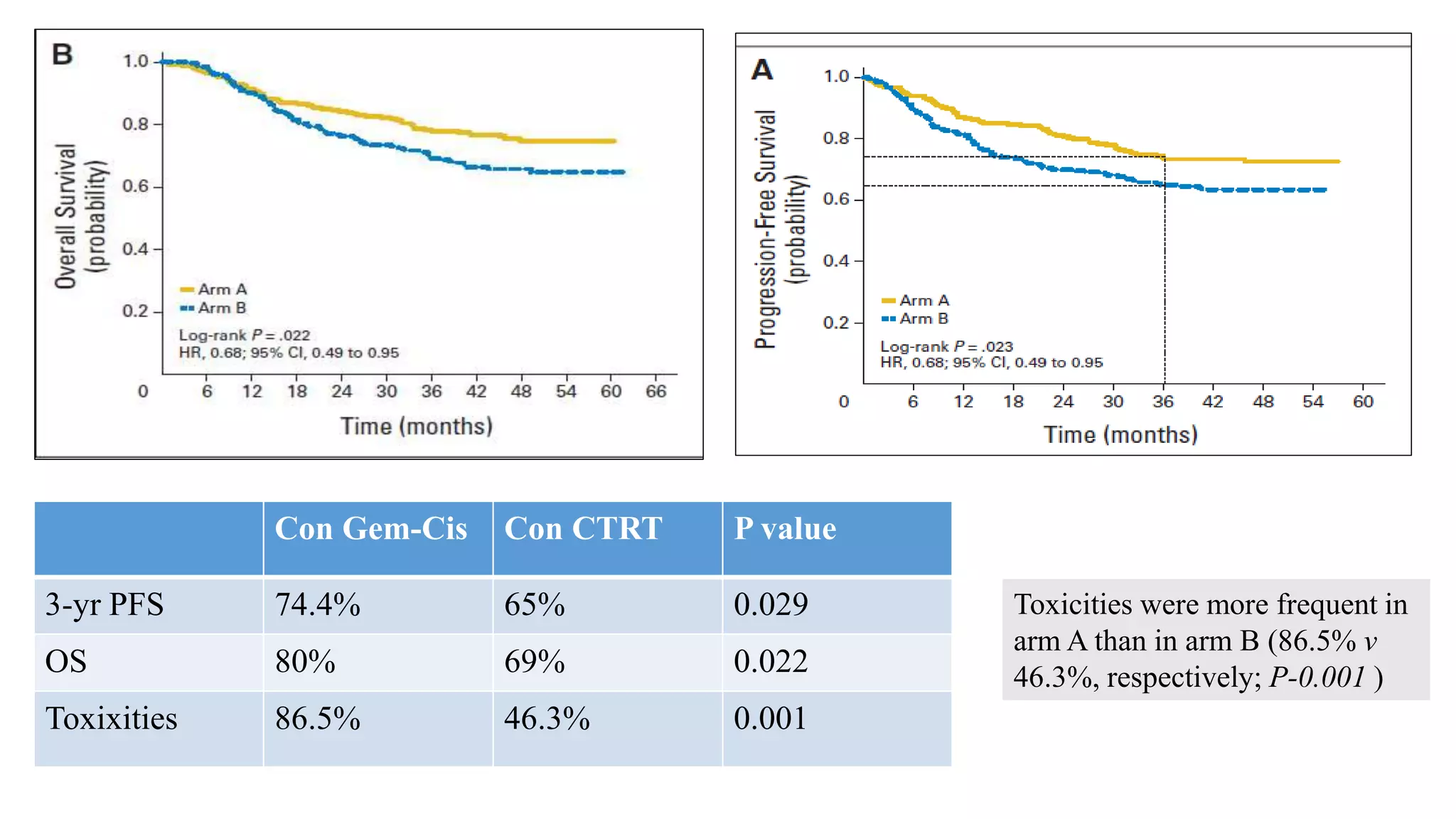
![Gonzalez et al
J Clin Oncol 29:1678-1685. © 2011
Arm A: cisplatin 40 mg/m2 and gemcitabine 125 mg/m2
weekly for 6 weeks with concurrent external-beam
radiotherapy [XRT] 50.4 Gy in 28 fractions, followed by
brachytherapy [BCT] 30 to 35 Gy in 96 hours, and then
two adjuvant 21-day cycles of cisplatin, 50 mg/m2 on day
1, plus gemcitabine, 1,000 mg/m2 on days
1 and 8) or
Arm B: cisplatin and concurrent XRT followed by BCT
only; dosing same as for arm A.](https://image.slidesharecdn.com/satyajeetcervixconcurrentchemo-radiotherapy-161219172653/75/Satyajeet-cervix-concurrent-chemo-radiotherapy-64-2048.jpg)