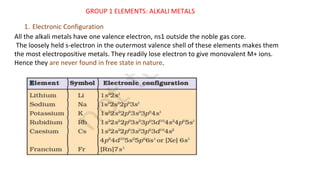
The s-block elements are those where the last electron enters the s-orbital. Groups 1 and 2 belong to the s-block. Group 1 elements are alkali metals including lithium, sodium, potassium, rubidium, cesium, and francium. Group 2 elements are alkaline earth metals including beryllium, magnesium, calcium, strontium, barium, and radium. The alkali metals have one valence electron and readily lose electrons to form M+ ions, so they are never found in free state in nature.



![ The general electronic configuration of s-block elements
[noble gas]ns1 for alkali metals
[noble gas] ns 2 for alkaline earth metals
ANOMALOUS PROPERTIES OF LITHiUM AND BERYLLIUM
• they resemble the second element of the following
group.
• Thus, lithium shows similarities to magnesium and
beryllium to aluminium in many of their properties.
• This type of diagonal similarity is commonly referred to
as diagonal relationship in the periodic table.
• The diagonal relationship is due to the similarity in
ionic sizes and or charge/radius ratio of the elements.](https://image.slidesharecdn.com/part1-230122033807-9096ed0d/85/s-block-elements-pptx-4-320.jpg)