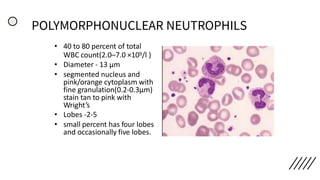
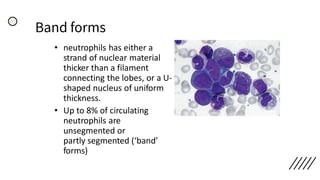
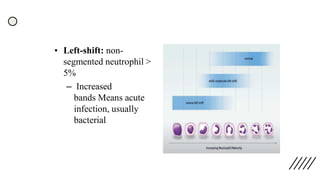
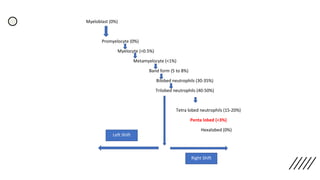
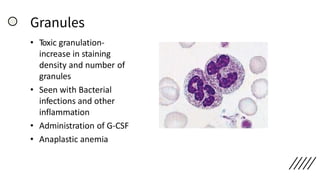
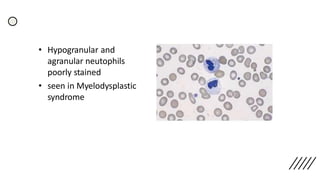
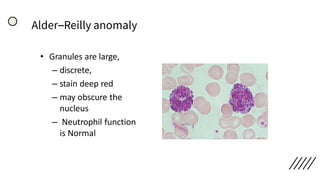
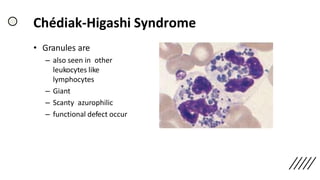
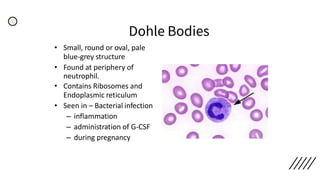
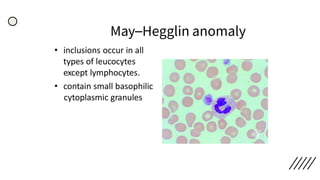
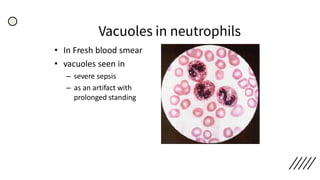
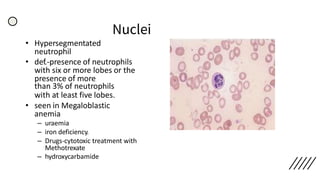
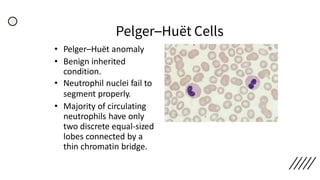
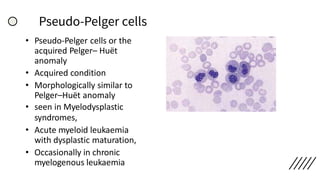
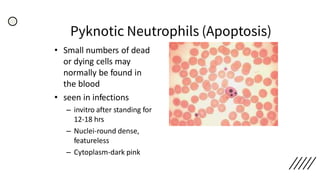
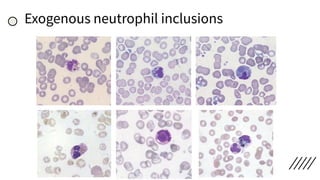
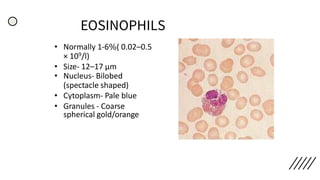
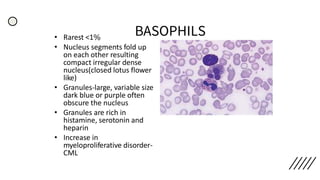
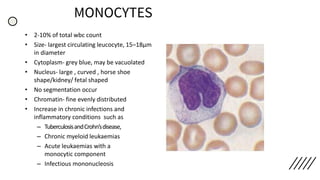
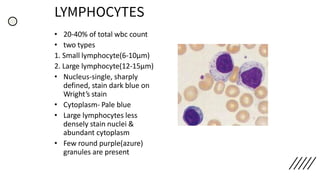
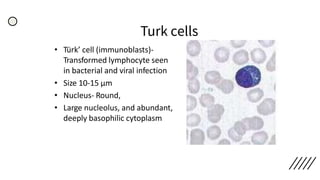
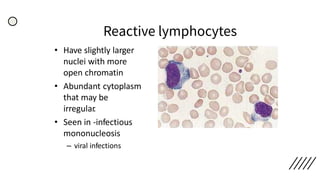
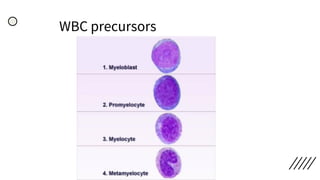
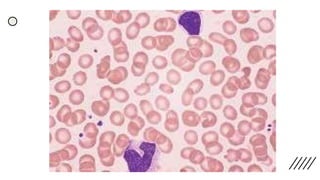
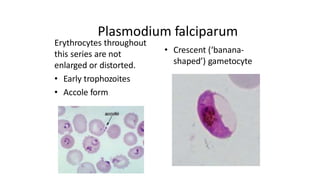
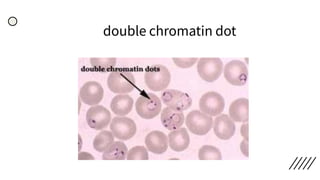
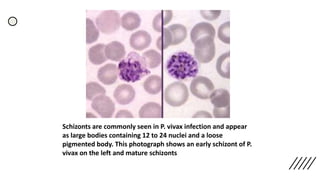
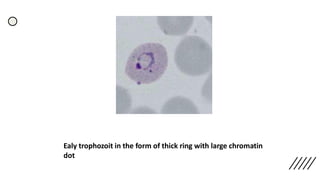
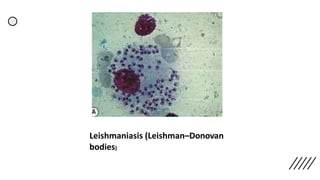
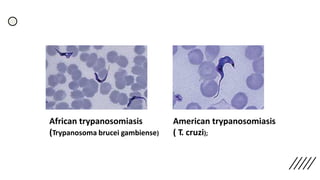
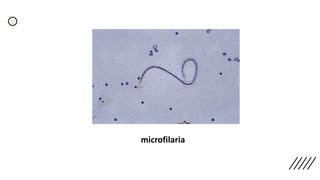
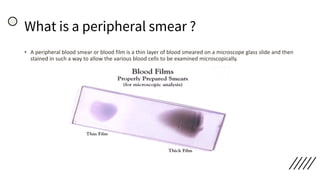
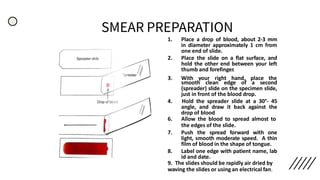
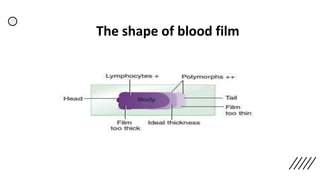
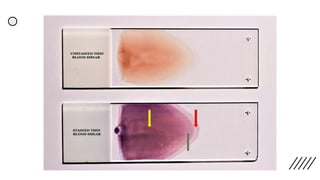
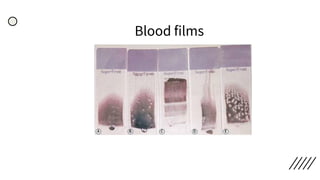
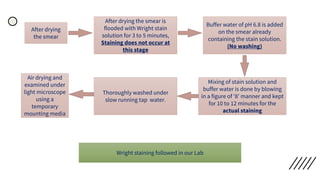
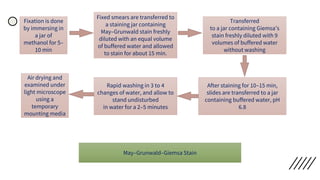
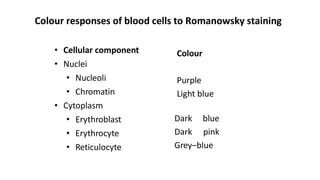
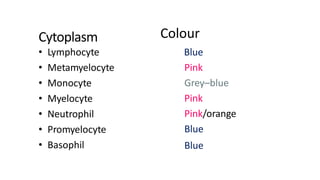
This document provides information on preparing and staining peripheral blood smears (PBS). It discusses how to make a wedge blood smear using the correct technique and equipment. It also describes how to evaluate a quality smear and identify common causes of poor smears. The document outlines the staining process for PBS, including the history and components of Romanowsky staining methods like Wright-Giemsa and May-Grunwald Giemsa. Factors that can influence staining and cause faulty results are discussed. Finally, it provides guidance on examining a PBS under the microscope, including evaluating red blood cells, white blood cells, platelets, and identifying any parasites.







![Brief History
• The beginnings of modern-day blood staining can be traced back to Ehrlich,
who in 1877, was the first scientist to divide the aniline dyes into acidic and
basic categories.
• Three years later , the malarial parasite was discovered, and a rigorous search
for an improved blood stain ensued – In 1888, Chenzinsky discovered a stain
composed of cationic dye methylene blue and anionic eosin.
• Malachowski modified that stain with a markedly improved color range
and depth
• Regrettably, when he published his results in Berlin in 1891, he did not give the
necessary details as to how his method might be reproduced.
• 3 weeks later the young Russian protozoologist Romanowsky published similar
findings in an article so celebrated that all future stains of analogous
composition were named as Romanowsky-type stains
[Romanowsky noted that a neglected, moldy methylene blue solution was more effective
in producing the desired red plasmodial chromatin bodies than was a fresh solution]](https://image.slidesharecdn.com/peripheralsmear-stainingandmorphology-200901120406/85/Peripheral-smear-staining-and-morphology-14-320.jpg)












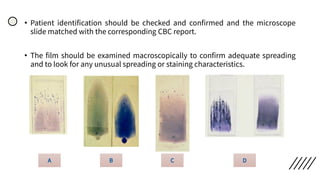








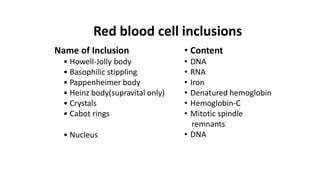
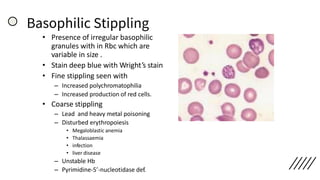
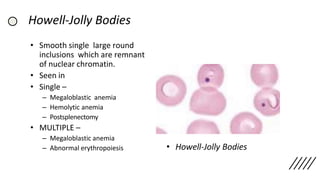
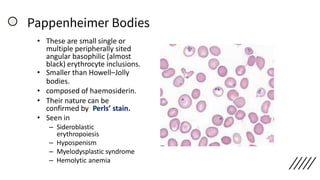
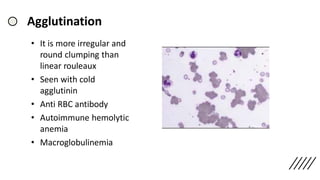
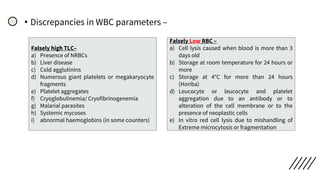
![• Discrepancies in Red cell parameters –
Falsely high RBC –
a) Numerous large platelets
b) Hyperlipidaemia
c) Cryoglobulinemia/
Cryofibrinogenemia
Falsely Low RBC –
a) Cold agglutinins (Rarely warm
autoantibodies)
b) EDTA‐dependent pan-
agglutination
c) In vitro red cell lysis due to
mishandling of Extreme
microcytosis or fragmentation
Falsely high MCV –
a) Storage of blood at room
temperature
b) Cold agglutinins and EDTA‐
dependent pan-agglutinins
c) very high WBC count
d) Hyperosmolar states (e.g.
hypernatraemia, )
e) Excess K2EDTA
Falsely Low MCV –
a) Increase in ambient
temperature
b) Hypo‐osmolar states (e.g.
hyponatraemia
c) Repeated mixing of sample
leading to increased
oxygenation
Falsely high RBC
a) Poorly mixed specimen
b) High WBC
c) Hyperlipidaemia,
[endogenous or due to
parenteral nutrition]
d) hypergammaglobuline
mia](https://image.slidesharecdn.com/peripheralsmear-stainingandmorphology-200901120406/85/Peripheral-smear-staining-and-morphology-71-320.jpg)