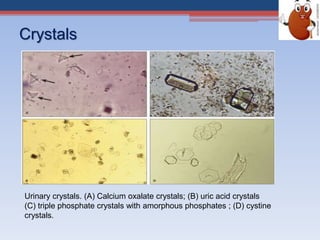
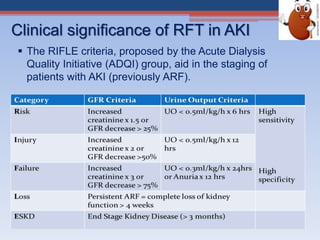
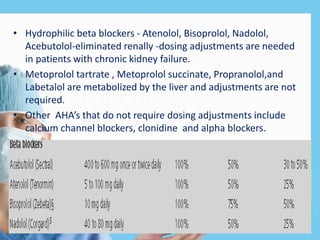
Renal function tests assess kidney function and detect impairment. They include tests of glomerular filtration rate (GFR) using creatinine, urea and inulin clearance. GFR is calculated using creatinine levels and equations factoring age, weight and sex. Other tests evaluate tubular function through urine concentration, osmolality, and checking for proteins, glucose and amino acids in urine. Together these tests provide valuable information about both glomerular and tubular integrity and kidney health.