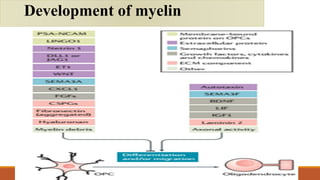
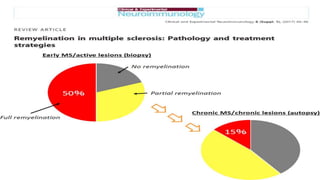
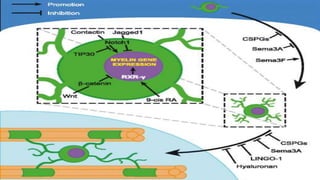
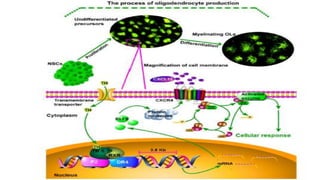
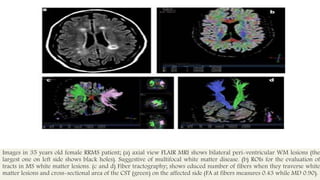
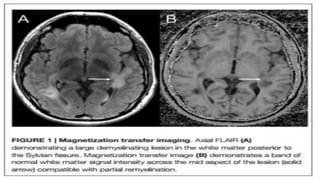
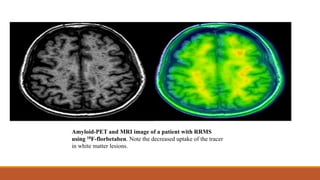
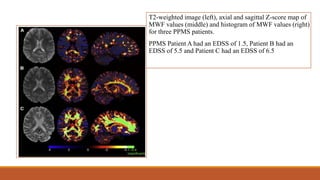
This document discusses remyelinating therapies for multiple sclerosis (MS). It begins by explaining how MS results in demyelination and how remyelination can restore neuronal function. Several potential remyelinating therapies currently in preclinical or clinical trials are described, including clobetasol, opicinumab, guanabenz, and olesoxime. Biomarkers for measuring remyelination like diffusion tensor imaging, magnetization transfer imaging, and positron emission tomography are also summarized. The document concludes that while challenges remain, promising remyelinating strategies exist to provide benefit throughout the entire course of MS.