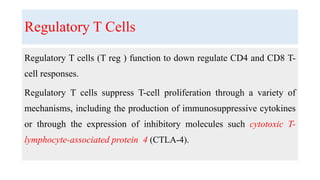
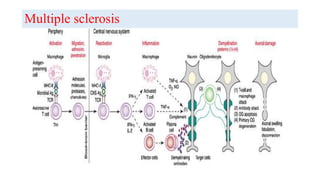
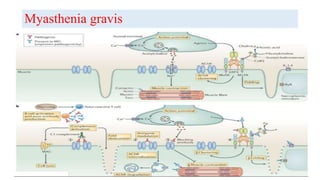
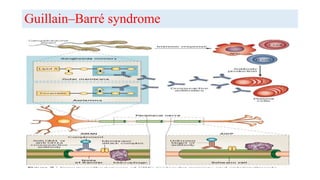
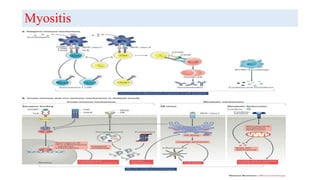
The document discusses neuroimmunology and provides information on the immune system and its normal functions and disorders. It describes the innate and adaptive immune systems, including skin, phagocytes, natural killer cells, the complement system, antibodies, B cells, antigen presenting cells, major histocompatibility complex, toll-like receptors, T lymphocytes, cluster of differentiation markers, cytokines, chemokines, initiation and regulation of the immune response, termination of the immune response, self-tolerance, central tolerance, peripheral tolerance, anergy, regulatory T cells, immune privilege in the central nervous system, and several immune-mediated disorders of the nervous system including multiple sclerosis, myasthenia gravis, Guillain-Barré syndrome



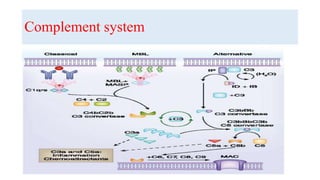
![Complement system
• They are protein synthesized in the liver.
• They are inactivated proteolysis enzymes.
• Numbered from [1- 9 ] according cascade of activation.
• When activated complement occurs it breaks down into portion eg: C5a and
C5b.
• The b segment is the large one and in turn will activate the next step in the
cascade.](https://image.slidesharecdn.com/neuro-immunologybasics-191012103840/85/Neuro-immunology-basics-5-320.jpg)


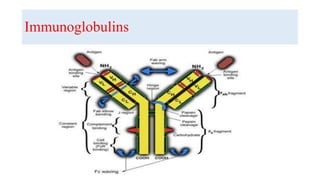
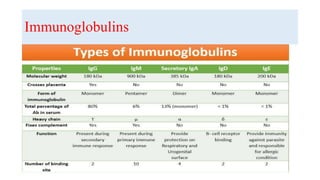
![Immunoglobulins
4. Each molecule consists of two identical polypeptide light chains
(kappa [κ] or lambda [λ]) linked to two identical heavy chains.
6. According to the biochemical nature of the heavy chain, Igs are
divided into five main classes: IgM, IgD, IgG, IgA, and IgE. These may
be further divided into subclasses depending on differences in the heavy
chain.](https://image.slidesharecdn.com/neuro-immunologybasics-191012103840/85/Neuro-immunology-basics-9-320.jpg)



![Toll like receptors [ TLR]
TLR are pathogen recognition
receptors .
They recognize pathogen associated
molecular pattern.
They present on cell surface and
intracellular.](https://image.slidesharecdn.com/neuro-immunologybasics-191012103840/85/Neuro-immunology-basics-18-320.jpg)


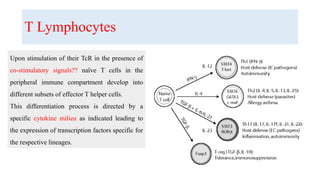
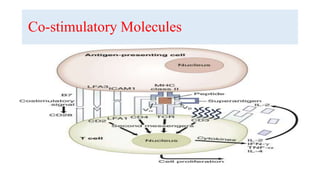
![Cluster of differentiation [ CD]
They are group of proteins
presents on cell surface which are
expressed during different stages
of maturation and differentiation.
CD were numbered 1-340
according to discovery order.](https://image.slidesharecdn.com/neuro-immunologybasics-191012103840/85/Neuro-immunology-basics-22-320.jpg)
![Cluster of differentiation [ CD]
CD3 – 4- 8 on T cells](https://image.slidesharecdn.com/neuro-immunologybasics-191012103840/85/Neuro-immunology-basics-23-320.jpg)