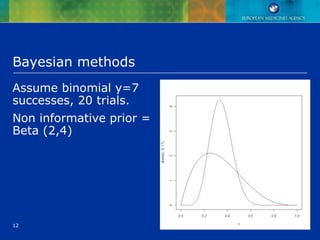
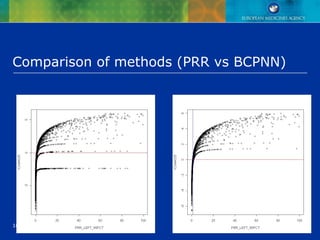
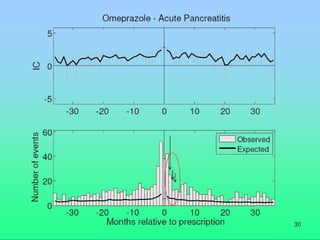
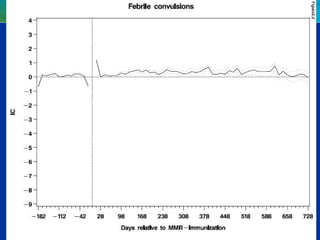
This document discusses quantitative methods for signal detection in pharmacovigilance. It describes current methods like proportional reporting ratios (PRR) that measure disproportionate reporting of adverse drug reactions. Bayesian methods and log-linear models are presented as improvements. Longitudinal health records are discussed as an alternative that avoids some biases but is difficult to implement. Future directions may include using all available data on reports and incorporating pharmacokinetic properties of drugs.






![8
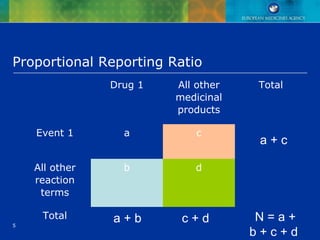
Disproportionality analysis (example)
. CNS drug for which the total No of reports is
400, of these 20 reports of diarrhoea
. All other products in the database (1 million
reports excluding reports involving drug A), of
these 50,000 reports of diarrhoea.
PRR = [20/400] / [50,000/1,000,000] = 1 (no
SDR)](https://image.slidesharecdn.com/quantitativemethodsofsignaldetection-151203222151-lva1-app6891/85/Quantitative-methods-of-signal-detection-8-320.jpg)
![9
Disproportionality analysis (example)
CNS drug for which the total No of reports is 400,
of these 40 reports of drowsiness
. All other products in the database (1 million
reports excluding reports involving drug A), of
these 25,000 reports of diarrhoea.
PRR = [40/400] / [25,000/1,000,000] = 4
(presence of a SDR)](https://image.slidesharecdn.com/quantitativemethodsofsignaldetection-151203222151-lva1-app6891/85/Quantitative-methods-of-signal-detection-9-320.jpg)













![28
Bibliography
Guideline on the use of statistical signal detection methods in EVDAS
http://www.ema.europa.eu/pdfs/human/phvwp/10646406enfin.pdf
Bate & Evans quantitaitve signal detection using ADR reporting. PDS
2009.
Hauben & Bate. Decision support methods for the detection of adverse
events in post-marketing data. Drug Discovery Today. April 2009.
Evaluation of Signal Detection Methods for use in Prospective Post
Licensure Medical Product Safety Surveillance [FDA Sentinel Initiative
Safety Signal Identification Contract] – Report
http://www.fda.gov/Safety/FDAsSentinelInitiative/ucm149343.htm](https://image.slidesharecdn.com/quantitativemethodsofsignaldetection-151203222151-lva1-app6891/85/Quantitative-methods-of-signal-detection-28-320.jpg)