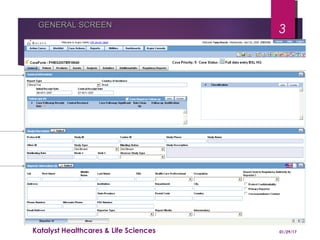
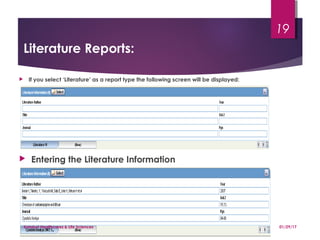
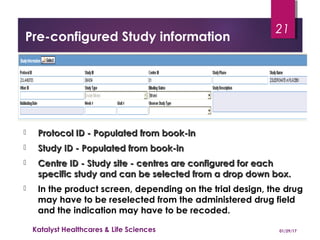
This document provides guidelines for entering and managing case reports within Argus Safety, covering key aspects such as report types, data fields, and processing workflow. It details how to categorize cases, manage reporter information, and the importance of confidentiality, particularly for consumer reports. Additionally, it outlines specific procedures for handling cases from license partners and other companies.