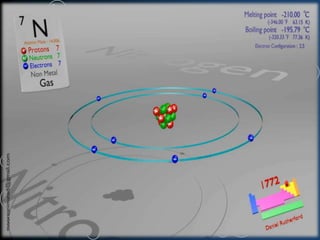
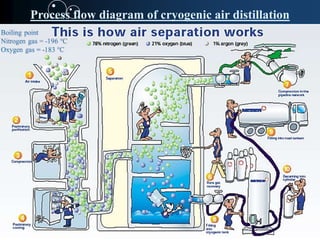
This document discusses nitrogen gas, including its discovery, properties, production via cryogenic distillation, and uses. Nitrogen makes up 78% of the atmosphere and is essential for life. It is produced commercially through cryogenic distillation of air, which involves compressing, cooling, and distilling air into its primary components in distillation columns. Nitrogen gas is nonflammable, odorless and colorless. It has various industrial and medical applications such as food preservation, welding, fertilizers, and medical anesthesia.