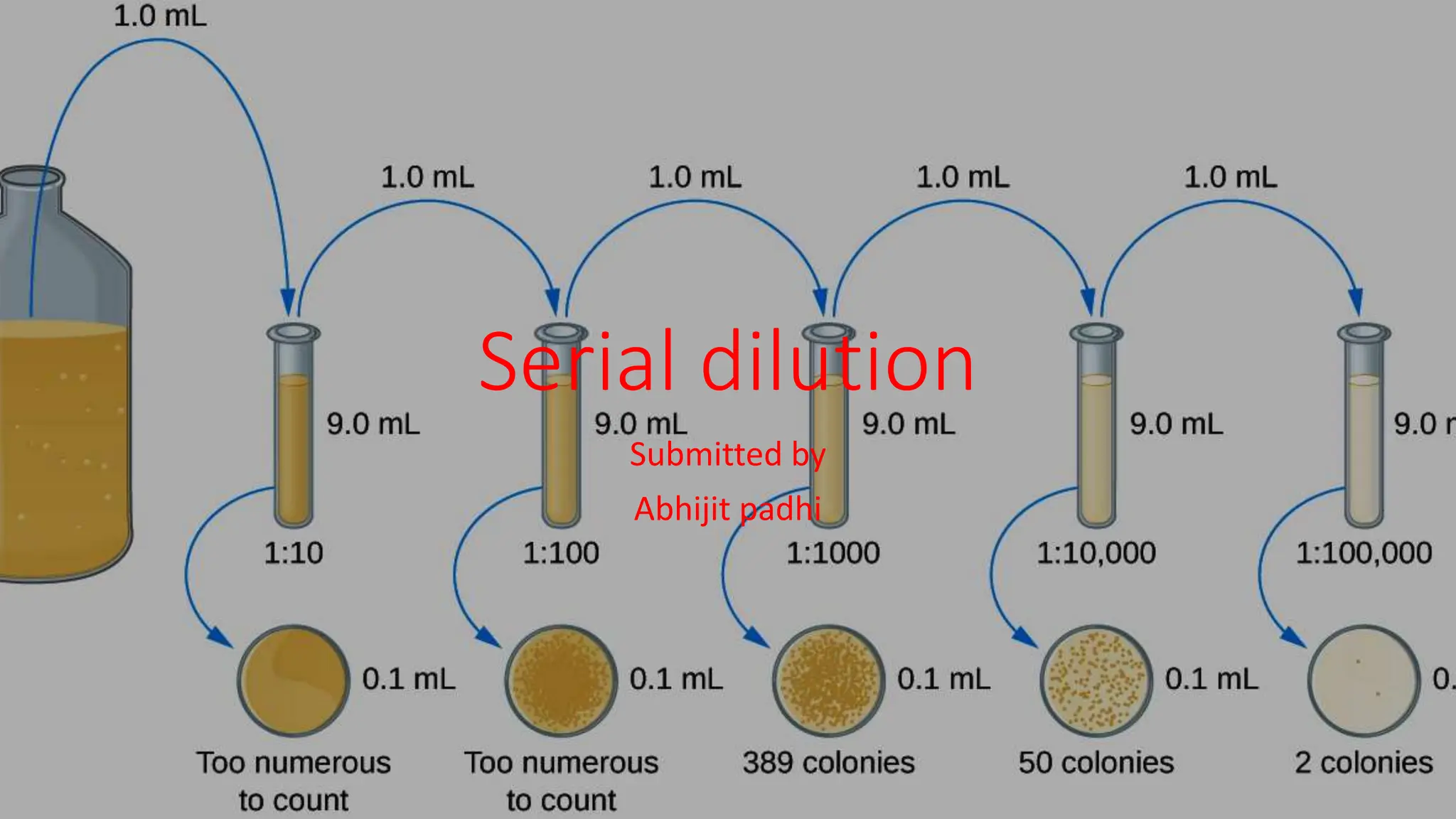
Serial dilution is a method of sequentially diluting a concentrated solution to a more usable concentration, commonly used in biological contexts to estimate the concentration of cells or organisms. The process involves taking an initial sample and repeatedly diluting it in sterile solutions, allowing for more manageable counting of cultured colonies. This technique has applications in multiple scientific fields, including microbiology, biochemistry, pharmacology, and homeopathy.