Liposomes
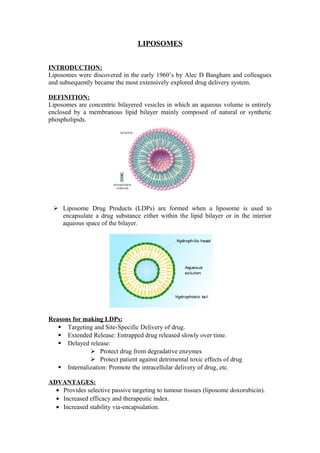
- 1. LIPOSOMES INTRODUCTION: Liposomes were discovered in the early 1960’s by Alec D Bangham and colleagues and subsequently became the most extensively explored drug delivery system. DEFINITION: Liposomes are concentric bilayered vesicles in which an aqueous volume is entirely enclosed by a membranous lipid bilayer mainly composed of natural or synthetic phospholipids. Liposome Drug Products (LDPs) are formed when a liposome is used to encapsulate a drug substance either within the lipid bilayer or in the interior aqueous space of the bilayer. Reasons for making LDPs: Targeting and Site-Specific Delivery of drug. Extended Release: Entrapped drug released slowly over time. Delayed release: Protect drug from degradative enzymes Protect patient against detrimental toxic effects of drug Internalization: Promote the intracellular delivery of drug, etc. ADVANTAGES: · Provides selective passive targeting to tumour tissues (liposome doxorubicin). · Increased efficacy and therapeutic index. · Increased stability via-encapsulation.
- 2. · Reduction in toxicity of the encapsulated agent. · Site avoidance effect. · Improved pharmacokinetic effects (reduced elimination, increased circulation life times). · Flexibility to couple with site-specific ligands to achieve active targeting. DISADVANTAGES: · Production cost is high. · Leakage and fusion of encapsulated drug/molecules. · Sometimes phospholipids undergo oxidation and hydrolysis like reaction. · Short half-life · Low solubility. STRUCTURAL COMPONENTS OF LIPOSOMES: The main components of liposomes are: · Phospholipids · Cholesterol The phospholipids possess a hydrophobic tail structure and a hydrophilic head component.When dissolved in water, the hydrophobic tails mutually attract while the hydrophilic heads contact with the aqueous medium external and internal to the liposome surface. In this way, double lipid layers are formed which seal off to form small vesicles similar to the body cells and organelles. Natural phospholipids: PC-Phosphatidylcholine PE-Phosphatidylethanolamine PS-Phosphatidylserine Synthetic phospholipids: DOPC-Dioleoylphosphatidylcholine DSPC-Disteroylphosphotidylcholine DOPE-Dioleoylhosphatidylethanolamine DSPE-Disteroylphosphotidylethanolamine Cholesterol acts as a fluidity buffer, i.e below the phase transition temperature, it makes the membrane less ordered and slightly more permeable; above the phase transition temperature it makes the membrane more ordered and stable.
- 3. CLASSIFICATION OF LIPOSOMES: Based on structural parameters: (1) MLV-Multilamellar vesicles(>0.5μm) (2) OLV-Oligolamellar vesicles(0.1-1μm) (3) UV Unilamellar vesicles(all size ranges) a. MUV-Medium Unilamellar Vesicles b. SUV-Small Unilamellar Vesicles(20-100nm) c. GUV-Giant Unilamellar Vesicles(>1μm) d. LUV-Large Unilamellar Vesicles(>100nm) (4) MVV/MV Multivesicular vesicles(>1μm) Based on method of preparation: (1) REV→SUVs/OLVs made by reverse-phase evaporation method (2) MLV-REV→MLVs made by reverse-phase evaporation method (3) SPLV→Stable plurilamellar vesicles (4) FATMLV→Frozen and thawed MLV (5) VET→Vesicles prepared by extrusion technique (6) DRV→De-hydrated rehydration method Based upon composition and applications: (1) Conventional liposomes (CL) (2) Fusogenic liposomes (3) pH sensitive liposomes (4) Cationic liposomes (5) Long circulatory(stealth) liposomes (LCL) (6) Immuno-liposomes PREPARATION OF LIPOSOMES: Generally two methods are followed 1. Passive loading 2. Remote loading Passive loading: This method involves the loading of the entrapped agents before (or) during the manufacture procedure. Remote loading: Certain types of compounds with ionisable groups, and those, which display both lipid and water solubility, can be introduced into the
- 4. liposomes after the formation of intact vesicles. (A)PASSIVE LOADING TECHNIQUES: It includes three different groups of methods working on different principles. Mechanical dispersion Solvent dispersion Detergent solubilisation (1) MECHANICAL/PHYSICAL DISPERSION: Four basic methods of mechanical dispersion Hand-shaken multilamellar vesicles Non-shaken vesicles Pro-liposomes Freeze drying Hand-shaking method: Simplest and most widely used method of physical dispersion Basic method involves Dissolution of the lipid mixture and charge components in chloroform:methanol solvent. Evaluation of the solvent in rotary evaporator or by hand shaking to form a film. Further drying of the film by attaching the flask to the manifold of the lyophilizer. Casted film is then dispersed in an aqueous medium. Upon hydration, lipid swell and peel off the wall of the flask and vesiculate forming multilamellar vesicles (MLVs). Non Shaking Method: The procedure differs from hand shaken method in that it uses a stream of nitrogen to provide agitation rather than the rotationary movements. Here the lipid film is exposed to watersaturated nitrogen for (15 – 20min). After Hydration, lipid is swelled by addition of bulk fluid, 10-20ml of 0.2 M sucrose in distilled water (degassed) is introduced. The flask is flushed with nitrogen, sealed & allowed to stand for 2 hrs at 37°C
- 5. After swelling, the vesicles are harvested by swirling the contents of the flask gently, to yield a milky‐suspension. Centrifugation LUVs. Pro-liposomes: To increase the surface area of dried lipid film and to facilitate instantaneous hydration, the lipid is dried over a finely divided particulate support, such as powdered Nacl, (or) sorbitol (or) other polysaccharides. These dried lipid coated particulates are called pro-liposomes. Pro-liposomes form dispersion of MLVs on adding water into them, where support is rapidly dissolved and lipid film hydrates to form MLVs. This method overcomes the stability problems of liposomes encountered during their storage as dispersion, dry (or) frozen form. Freeze Drying Method: Another method of dispersing the lipid in a finely divided form, prior to addition of aqueous medium is to freeze dry the lipid dissolved in a suitable organic solvent. Tertiary butanol is considered to be the most ideal solvent. After obtaining the dry lipid, which is expanded foam like structure, water or saline can be added with rapid mixing above the phase transition temperature to give MLVs. After these methods, other processing methods are used to modify the type of vesicles that are produced such as: Micro emulsification liposomes (MEL) Sonicated unilamellar vesicles (SUVs) French Pressure Cell liposomes Membrane extrusion liposomes Dried reconstituted vesicles (DRVs) Freeze thaw sonication pH induced vesiculation Calcium induced fusion These methods are known as “the mechanical treatment of MLVs” or “Processing of lipids hydrated by physical means” (2) SOLVENT DISPERSION METHODS: In this method, lipids are first dissolved in an organic solution, which then brought into contact with an aqueous phase containing materials to be entrapped within the Liposomes. Ethanol injection: This method is used as one of the alternatives used for the preparation of SUVs without sonication. An ethanol solution of lipids is injected rapidly through a fine needle into an excess of saline or other aqueous medium. The procedure yields a high proportion of SUVs (~25nm). This method is extremely simple and has low risk of degradation of sensitive lipids.
- 6. Ether injection: It is similar to the ethanol injection. It involves injecting the immiscible organic solution very slowly into an aqueous phase through a narrow needle at the temperature of vaporizing the organic solvent. This method may also treat sensitive lipids very gently. It has little risk of causing oxidative degradation provided ether is free from peroxides. Rapid solvent exchange vesicles(RSEVs): The lipid mixture is quickly transferred between an essentially pure solvent environment and a pure aqueous environment. The method involves passing the organic solution of the lipids through the orifice of blue-tipped syringe under the vacuum into a tube containing aqueous buffer. The tube is mounted on the vortexer. Bulk solvent vaporizes and is removed within seconds before coming in contact with aqueous environment. Reverse phase evaporation vesicles: In this method removal of solvent is from an emulsion by evaporation. The droplets are formed by bath sonication of mixture of two phases. And the emulsion is dried down to a semisolid gel in a rotatory evaporator under reduced pressure. And bring water droplets to get collapsed by vigorous mechanical shaking using a vortex mixer. The aqueous content of the collapsed droplet provides the medium required for dispersion of these newly formed liposomes. And conversion of gel into a homogenous free flowing fluid. And the vesicles generally formed are unilamellar.
- 7. Stable plurilamellar vesicles (SPLVs): In this method, preparation of water-inorganic phase dispersion with an excess of lipid followed by drying under continued bath sonication with an intermittent stream of nitrogen. The redistribution and equilibration of aqueous solvent and solute occurs in between the various bilayers in each plurilamellar vescile. (3) DETERGENT SOLUBILIZATION: In this method, the phospholipids are brought into contact with the aqueous phase via detergents, which associate with phospholipid molecules and serve to screen the hydrophobic portions of the molecule. The structures thus formed are known as micelles. As the detergent concentration increases further, the micelles are reduced in size until they become saturated with detergent, whereupon the concentration of free molecules equals the CMC & simple detergent micelles are formed. The basic feature is to remove the detergent from preformed mixed micelles containing phospholipid, whereupon unilamellar vesicles form spontaneously. To remove the detergents, and all the transition of mixed micelles to concentric bilayered form, three methods can be employed: Dialysis Column chromatography Use of biobeads (B) REMOTE (ACTIVE) LOADING: Load drug molecules into preformed liposomes using pH gradients and potential difference across the liposomal membranes. A concentration difference in proton concentration across the membrane of liposomes can drive the loading of amphipathic molecules. CHARACTERIZATION OF LIPOSOMES: There are mainly three types of characterization of liposomes. They are:
- 8. 1.Physical characterization 2.Biological characterization 3.Chemical characterization Physical Characterization: S.NO. Characterization parameters Analytical method/Instrument 1. Vesicle shape and surface morphology Transmission electron microscopy, Freeze-fracture electron microscopy 2. Mean vesicle size and size distribution (submicron and micron range) Dynamic light scattering, zetasizer, Photon correlation spectroscopy, laser light scattering, gel permeation and gel exclusion 3. Surface charge Free-flow electrophoresis 4. Electrical surface potential and surface pH Zeta potential measurements & pH sensitive probes 5. Lamellarity Small angle X-ray scattering, 31P-NMR, Freeze-fracture electron microscopy 6. Phase behavior Freeze-fracture electron microscopy, Differential scanning colorimetery 7. Percent of free drug/ percent capture Mini column centrifugation, ion-exchange chromatography, radiolabelling 8. Drug release Diffusion cell/ dialysis Biological Characterization: S.NO. Characterization parameters Analytical method/Instrument 1. Sterility Aerobic or anaerobic cultures 2. Pyrogenicity Limulus Amebocyte Lysate (LAL) test 3. Animal toxicity Monitoring survival rates, histology and pathology Chemical Characterization: S.NO. Characterization parameters Analytical method/Instrument 1. Phospholipid concentration Barlett assay, stewart assay, HPLC
- 9. 2. Cholesterol concentration Cholesterol oxidase assay and HPLC 3. Phopholipid peroxidation UV absorbance, Iodometric and GLC 4. Phospholipid hydrolysis, Cholesterol auto-oxidation. HPLC and TLC 5. Osmolarity Osmometer APPLICATIONS OF LIPOSOMES 1. Liposomes as drug/protein delivery vehicles Controlled & sustained drug release Enhanced drug stabilization Altered pharmacokinetics and biodistribution Enzyme replacement therapy and liposomal storage disorders 2. Liposomes in antimicrobial, antifungal (lung therapeutics) and antiviral (anti- HIV therapy) Liposomal drugs Liposomal biological response modifier 3. Liposomes in tumour therapy Carrier of small cytotoxic molecules Vehicle for macromolecules as cytokines or genes 4. Liposomes in gene delivery Gene and antisense therapy Genetic (DNA) vaccination 5. Liposomes in immunology Immunoadjuvant Immunomodulator immunodiagnosis 6. Liposomes as artificial blood surrogates 7. Liposomes as radiopharmaceutical and radio-diagnostic carriers 8. Liposomes in cosmetics and dermatology 9. Liposomes in enzyme immobilization and bio-reactor technology References: Targeted & Controlled Drug Delivery by S.P.Vyas & R.K.Khar www.pharmainfo.net www.pharmxchange.info
- 10. 2. Cholesterol concentration Cholesterol oxidase assay and HPLC 3. Phopholipid peroxidation UV absorbance, Iodometric and GLC 4. Phospholipid hydrolysis, Cholesterol auto-oxidation. HPLC and TLC 5. Osmolarity Osmometer APPLICATIONS OF LIPOSOMES 1. Liposomes as drug/protein delivery vehicles Controlled & sustained drug release Enhanced drug stabilization Altered pharmacokinetics and biodistribution Enzyme replacement therapy and liposomal storage disorders 2. Liposomes in antimicrobial, antifungal (lung therapeutics) and antiviral (anti- HIV therapy) Liposomal drugs Liposomal biological response modifier 3. Liposomes in tumour therapy Carrier of small cytotoxic molecules Vehicle for macromolecules as cytokines or genes 4. Liposomes in gene delivery Gene and antisense therapy Genetic (DNA) vaccination 5. Liposomes in immunology Immunoadjuvant Immunomodulator immunodiagnosis 6. Liposomes as artificial blood surrogates 7. Liposomes as radiopharmaceutical and radio-diagnostic carriers 8. Liposomes in cosmetics and dermatology 9. Liposomes in enzyme immobilization and bio-reactor technology References: Targeted & Controlled Drug Delivery by S.P.Vyas & R.K.Khar www.pharmainfo.net www.pharmxchange.info
