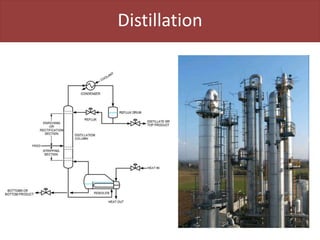
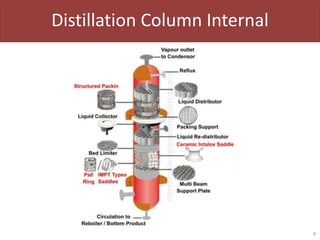
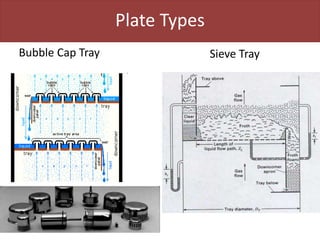
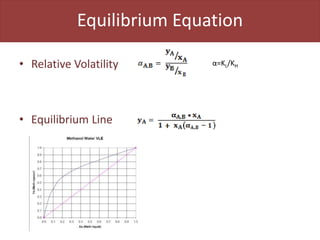
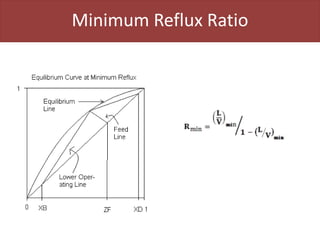
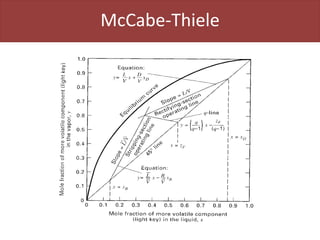
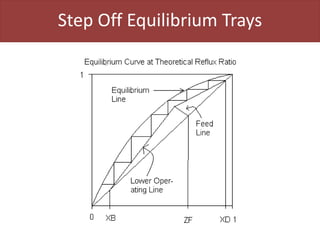
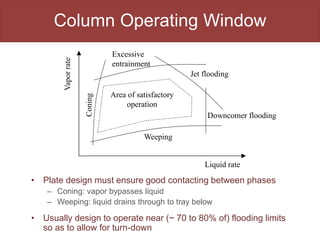
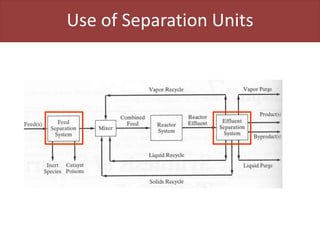
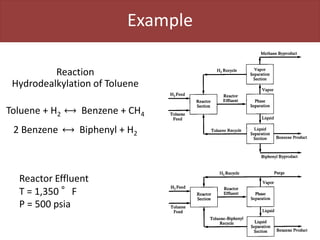
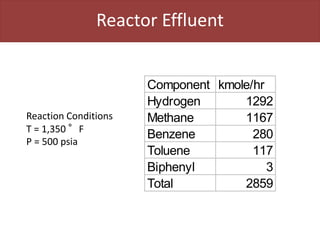
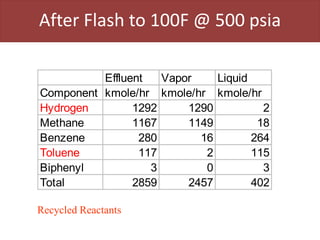
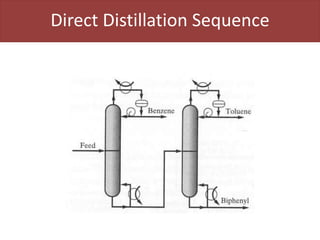
This document provides an overview of distillation columns and separation train design. It discusses different types of distillation columns including plate columns with bubble cap trays and sieve trays, and packed columns. It also covers concepts like equilibrium lines, operating lines, minimum reflux ratio, and McCabe-Thiele diagrams. The document discusses methods for determining the number of columns needed, possible sequences, and how to evaluate sequences using the marginal vapor rate method. Finally, it provides heuristics for sequencing separation points in a distillation train.




![Short cut to Selecting a Column Design
• Minimum Cost for Distillation Column will
occur when you have a
– Minimum of Total Vapor Flow Rate for column
– Occurs at
• R = 1.2 Rmin @ N/Nmin= 2
• Nmin= log[(dLK/bLK)(bHK/dHK)]/log[αLK,HK]
• Rmin ≈ (F/D)/(α-1)
– V = D(R+1)
• V = Vapor Flow Rate
• D = Distillate Flow Rate (Production Rate)
• R = Reflux Ratio](https://image.slidesharecdn.com/l09-separationsandcolumnsimulation-220917223705-3062eaf1/85/L09-Separations-and-Column-Simulation-pptx-12-320.jpg)




![Column Sequences
• Number of Columns
– Nc = P – 1
• P = Number of Products
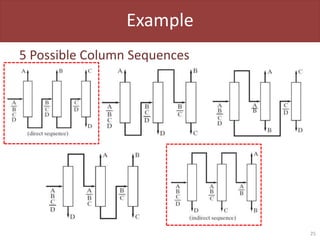
• No. of Possible Column Sequences
– Ns = [2(P–1)]! / [P!(P–1)!]
• P = Number of Products
P = 3, Nc = 2, Ns = 2
P = 4, Nc = 3, Ns = 5
P = 5, Nc = 4, Ns = 14
P = 6, Nc = 5, Ns = 42
P = 7, Nc = 6, Ns = 132
Number of possible
column sequences
becomes very large
very quickly!](https://image.slidesharecdn.com/l09-separationsandcolumnsimulation-220917223705-3062eaf1/85/L09-Separations-and-Column-Simulation-pptx-23-320.jpg)
![Example
• P = Number of Products = 4 ( A, B, C, D)
• Number of Columns
– Nc = P – 1= 4-1=3
• No. of Possible Column Sequences
– Ns = [2(P–1)]! / [P!(P–1)!]
= [2(4–1)]! / [4!(4–1)!] )
= [2X3]! / [4!X3!]
= 6! / [4!X3!]
= 720 / [24X6]
= 720 / 144
= 5
24](https://image.slidesharecdn.com/l09-separationsandcolumnsimulation-220917223705-3062eaf1/85/L09-Separations-and-Column-Simulation-pptx-24-320.jpg)