Igcse biology edexcel 2.39 2.43
•Download as PPTX, PDF•
13 likes•18,431 views
Edexcell ppt Biology 2.39-2.43 Used in lessons to scaffold class teaching and as a revision resource for students
Report
Share
Report
Share
Recommended
Laboratory apparatus and safety measures

This Slides Share discuss common laboratory apparatus as well as safety measures while inside a laboratory room. It includes images so that students would be able to visualize the equipment.
Gas Laws

this presentation contains Boyle's law, Charles's law, Avogadro's law, ideal gas equation, units of gas constant R and significance of PV=nRT
Recommended
Laboratory apparatus and safety measures

This Slides Share discuss common laboratory apparatus as well as safety measures while inside a laboratory room. It includes images so that students would be able to visualize the equipment.
Gas Laws

this presentation contains Boyle's law, Charles's law, Avogadro's law, ideal gas equation, units of gas constant R and significance of PV=nRT
Chemical Thermodynamics

Basic Terminology,Heat, energy and work, Internal Energy (E or U),First Law of Thermodynamics, Enthalpy,Molar heat capacity, Heat capacity,Specific heat capacity,Enthalpies of Reactions,Hess’s Law of constant heat summation,Born–Haber Cycle,Lattice energy,Second law of thermodynamics, Gibbs free energy(ΔG),Bond Energies,Efficiency of a heat engine
Anatomy/Physiology Slideshow: The Respiratory System

Learner High School student Azabelle Peters has created this slide show fo
IDEAL GAS AND LAWS

Slides consists the ideal gas and ideal gas laws.This presentation was presented by Farhan Qasim. For any suggestion Farhanqasim373@gmail.com
Laws of Chemical Combination and Balancing Chemical Equation.pptx

The verification of each of the following chemical laws was well established with examples to simplify it.
THE LAW OF CONSERVATION OF MASS
This Law was established by Lavoisier, a French Chemist. This law states that matter is neither created nor destroyed during chemical reaction but changes from one form to another. This means that in a chemical reaction, the total mass of all reacting substances (i.e. the reactants) is equal to the total mass of the products.
THE LAW OF DEFINITE PROPORTIONS OR CONSTANT COMPOSITION
This Law was proposed by Proust (1755-1826). The Law of Definite Proportions states that all pure samples of a particular chemical compound contain similar elements combined in the same proportion by mass.
THE LAW OF MULTIPLE PROPORTIONS
The law of Multiple Proportions states that if two elements, A and B, combine to form more than one chemical compound, the various masses of one element, A which combine separately with a fixed mass of the other element, B, are in simple multiple ratios.
BALANCING CHEMICAL EQUATION WITH CALCULATION
A chemical equation is a shorthand expression for a chemical change or reaction. It shows among other things the arrangement of atoms that are involved in the reaction.
When balancing an equation, you must remember the following:
Know the reacting substances and the products formed.
Know the chemical formulae for all the substances.
Write, in front of the formulae, coefficients that will balance the equation.
Common gases, such as oxygen, hydrogen, chlorine and nitrogen, in the free state, are diatomic, e.g. O2, H2, Cl2 and N2
Other elements in the free state, such as sodium, potassium, copper and iron, are represented by their atomic symbols, e.g. Na, K, Cu and Fe.
Chem 2 - Chemical Equilibrium I: What is Equilibrium

Chem 2 - Chemical Equilibrium I: What is Equilibrium
Igcse biology edexcel 2.17 2.32

Edexcell ppt Biology 2.17-2.32
Used in lessons to scaffold class teaching and as a revision resource for students
gaseous exchange. Class 10 biology ch#1 gaseous exchange

class 10th chapter#1 gaseous exchange by ahmad ali.
https://youtu.be/8X1EFt0U5PE
like youtube channel for this vedio
Functional groups

Organic chemistry involves the study of the structure, properties, composition, reactions, and preparation of carbon-containing compounds, which include not only hydrocarbons but also compounds with any number of other elements, including hydrogen (most compounds contain at least one carbon–hydrogen bond), nitrogen, oxygen, halogens, phosphorus, silicon, and sulfur.
This branch of chemistry was originally limited to compounds produced by living organisms but has been broadened to include human-made substances such as plastics. The range of application of organic compounds is enormous and also includes, but is not limited to, pharmaceuticals, petrochemicals, food, explosives, paints, and cosmetics.
AS Level Biology - 9) Gas Exchange

Like Membrane, this is one of the shorter topics with less content to go through. More or less an extension of the mamal transport system, the gas exchange system should still be seen as an individual self-serving/ self-functioning system worthy of its own spotlight.
More Related Content
What's hot
Chemical Thermodynamics

Basic Terminology,Heat, energy and work, Internal Energy (E or U),First Law of Thermodynamics, Enthalpy,Molar heat capacity, Heat capacity,Specific heat capacity,Enthalpies of Reactions,Hess’s Law of constant heat summation,Born–Haber Cycle,Lattice energy,Second law of thermodynamics, Gibbs free energy(ΔG),Bond Energies,Efficiency of a heat engine
Anatomy/Physiology Slideshow: The Respiratory System

Learner High School student Azabelle Peters has created this slide show fo
IDEAL GAS AND LAWS

Slides consists the ideal gas and ideal gas laws.This presentation was presented by Farhan Qasim. For any suggestion Farhanqasim373@gmail.com
Laws of Chemical Combination and Balancing Chemical Equation.pptx

The verification of each of the following chemical laws was well established with examples to simplify it.
THE LAW OF CONSERVATION OF MASS
This Law was established by Lavoisier, a French Chemist. This law states that matter is neither created nor destroyed during chemical reaction but changes from one form to another. This means that in a chemical reaction, the total mass of all reacting substances (i.e. the reactants) is equal to the total mass of the products.
THE LAW OF DEFINITE PROPORTIONS OR CONSTANT COMPOSITION
This Law was proposed by Proust (1755-1826). The Law of Definite Proportions states that all pure samples of a particular chemical compound contain similar elements combined in the same proportion by mass.
THE LAW OF MULTIPLE PROPORTIONS
The law of Multiple Proportions states that if two elements, A and B, combine to form more than one chemical compound, the various masses of one element, A which combine separately with a fixed mass of the other element, B, are in simple multiple ratios.
BALANCING CHEMICAL EQUATION WITH CALCULATION
A chemical equation is a shorthand expression for a chemical change or reaction. It shows among other things the arrangement of atoms that are involved in the reaction.
When balancing an equation, you must remember the following:
Know the reacting substances and the products formed.
Know the chemical formulae for all the substances.
Write, in front of the formulae, coefficients that will balance the equation.
Common gases, such as oxygen, hydrogen, chlorine and nitrogen, in the free state, are diatomic, e.g. O2, H2, Cl2 and N2
Other elements in the free state, such as sodium, potassium, copper and iron, are represented by their atomic symbols, e.g. Na, K, Cu and Fe.
Chem 2 - Chemical Equilibrium I: What is Equilibrium

Chem 2 - Chemical Equilibrium I: What is Equilibrium
Igcse biology edexcel 2.17 2.32

Edexcell ppt Biology 2.17-2.32
Used in lessons to scaffold class teaching and as a revision resource for students
gaseous exchange. Class 10 biology ch#1 gaseous exchange

class 10th chapter#1 gaseous exchange by ahmad ali.
https://youtu.be/8X1EFt0U5PE
like youtube channel for this vedio
Functional groups

Organic chemistry involves the study of the structure, properties, composition, reactions, and preparation of carbon-containing compounds, which include not only hydrocarbons but also compounds with any number of other elements, including hydrogen (most compounds contain at least one carbon–hydrogen bond), nitrogen, oxygen, halogens, phosphorus, silicon, and sulfur.
This branch of chemistry was originally limited to compounds produced by living organisms but has been broadened to include human-made substances such as plastics. The range of application of organic compounds is enormous and also includes, but is not limited to, pharmaceuticals, petrochemicals, food, explosives, paints, and cosmetics.
AS Level Biology - 9) Gas Exchange

Like Membrane, this is one of the shorter topics with less content to go through. More or less an extension of the mamal transport system, the gas exchange system should still be seen as an individual self-serving/ self-functioning system worthy of its own spotlight.
What's hot (20)
Anatomy/Physiology Slideshow: The Respiratory System

Anatomy/Physiology Slideshow: The Respiratory System
Laws of Chemical Combination and Balancing Chemical Equation.pptx

Laws of Chemical Combination and Balancing Chemical Equation.pptx
Chem 2 - Chemical Equilibrium I: What is Equilibrium

Chem 2 - Chemical Equilibrium I: What is Equilibrium
gaseous exchange. Class 10 biology ch#1 gaseous exchange

gaseous exchange. Class 10 biology ch#1 gaseous exchange
Viewers also liked
Parts of a microscope

This is just an excerpt from a slideshow that was all ready on slideshare by someone else.
Measurement & Error

I split the presentation for the unit into two, as I added so many slides to help with student questions and misconceptions. This one focuses on mathematical aspects of the unit.
Viewers also liked (20)
IGCSE Biology Revision Quiz - Organisms and Life Processes

IGCSE Biology Revision Quiz - Organisms and Life Processes
Chapter 9: Contact Process, Haber Process and Alloy

Chapter 9: Contact Process, Haber Process and Alloy
Similar to Igcse biology edexcel 2.39 2.43
Photosynthesis and respiration in ecosystems

more sciencebitz resources on iTunesU and iBooks https://itunesu.itunes.apple.com/enroll/DEZ-HWS-HNJ
https://itun.es/gb/ymzI6.n
Greenhouse Effect, Carbon Cycle & Rising Temperatures

Greenhouse Effect, Carbon Cycle & Rising Temperatures .
Respiration Class 10

This ppt is of the topic respiration class 10 cointaining some extra knowledge too.
Effect of Plasmid Addition on the Production of 1-Butanol from CO2 in Isolate...

Effect of Plasmid Addition on the Production of 1-Butanol from CO2 in Isolate...International Journal of Modern Research in Engineering and Technology
For a sustainable future it is essential to close the global carbon cycle. Oxidised forms of carbon, in
particular CO2, can be used to synthesise energy-rich organic molecules. Engineered cyanobacteria have
attracted attention as catalysts for the direct conversion of CO2 into reduced fuel compounds.1-Butanol is a
promising gasoline replacement compared to the more commonly used ethanol due to several advantages.
Specifically, 1-butanol is less corrosive and has a higher energy density than ethanol. In this study,
Synechococcus elongatus was isolated from the sea sediment in Konak and the effects of CO2 isolated from the
same region on the production of 1-butanol was investigated. Addition of plasmid slightly increased the CO2
utilisation rates. The effects of environmental factors (NO3-N, SO4-S, NaCl, H2, O2) on the ratio of 1-butanol
production to CO2 removal were investigated. Under optimized conditions S.elongatus metabolized theCO2
according to Monod kinetic(Ks = 1.5 mg L-1
and µ = 0.21 day-1
). Under highNO3-N, SO4-S, NaCl, and O2
concentrations 1-butanol produced with un-competitive inhibition with a high Ki value of 2.3 mg L-1 indicating
the low inhibition in S.elongatus with plasmid. The competitive inhibition constant (Ki) is low (0.70 mg L-1
)
exhibiting the high competitive inhibition at high concentration for the operational conditions given aboveBiogeochemical cycles

biogeochemical cycle,carbon,nirogen,sulfur and the microbes involved in it.The effect of biogeochemical cycle in environment
Similar to Igcse biology edexcel 2.39 2.43 (20)
Processes controlling carbon dioxide and oxygen in Southern Ocean ecosystems

Processes controlling carbon dioxide and oxygen in Southern Ocean ecosystems
Greenhouse Effect, Carbon Cycle & Rising Temperatures

Greenhouse Effect, Carbon Cycle & Rising Temperatures
Effect of Plasmid Addition on the Production of 1-Butanol from CO2 in Isolate...

Effect of Plasmid Addition on the Production of 1-Butanol from CO2 in Isolate...
More from Marc Rodriguez
GCSE IGCSE Biology by Syllabus points

Edexcell Biology;
Most year 10 & 11 syllabus points by ppt.
Used in lessons to scaffold class teaching and as a revision resource for students
These resources are from many sources
Igcse biology edexcel 5.10 5.20

Edexcell ppt Biology 5.10 - 5.20
Used in lessons to scaffold class teaching and as a revision resource for students
Igcse biology edexcel 5.1 5.9

Edexcell ppt Biology 5.1 - 5.9
Used in lessons to scaffold class teaching and as a revision resource for students
Igcse biology edexcel 4.1 4.17

Edexcell ppt Biology 4.1 - 4.17
Used in lessons to scaffold class teaching and as a revision resource for students
Igcse biology edexcel 3.13 3.33

Edexcell ppt Biology 3.13 - 3.33
Used in lessons to scaffold class teaching and as a revision resource for students
Igcse biology edexcel 3.8 3.12

Edexcell ppt Biology 3.8 - 3.12
Used in lessons to scaffold class teaching and as a revision resource for students
Igcse biology edexcel 2.77 2.90

Edexcell ppt Biology 2.77 - 2.90
Used in lessons to scaffold class teaching and as a revision resource for students
Igcse biology edexcel 2.1 2.16

Edexcell ppt Biology 2.1-2.16
Used in lessons to scaffold class teaching and as a revision resource for students
Igcse biology edexcel 1.1 1.3

Edexcell ppt Biology 1.1-1.3
Used in lessons to scaffold class teaching and as a revision resource for students
More from Marc Rodriguez (16)
Recently uploaded
Observation of Io’s Resurfacing via Plume Deposition Using Ground-based Adapt...

Since volcanic activity was first discovered on Io from Voyager images in 1979, changes
on Io’s surface have been monitored from both spacecraft and ground-based telescopes.
Here, we present the highest spatial resolution images of Io ever obtained from a groundbased telescope. These images, acquired by the SHARK-VIS instrument on the Large
Binocular Telescope, show evidence of a major resurfacing event on Io’s trailing hemisphere. When compared to the most recent spacecraft images, the SHARK-VIS images
show that a plume deposit from a powerful eruption at Pillan Patera has covered part
of the long-lived Pele plume deposit. Although this type of resurfacing event may be common on Io, few have been detected due to the rarity of spacecraft visits and the previously low spatial resolution available from Earth-based telescopes. The SHARK-VIS instrument ushers in a new era of high resolution imaging of Io’s surface using adaptive
optics at visible wavelengths.
What is greenhouse gasses and how many gasses are there to affect the Earth.

What are greenhouse gasses how they affect the earth and its environment what is the future of the environment and earth how the weather and the climate effects.
Comparative structure of adrenal gland in vertebrates

Adrenal gland comparative structures in vertebrates
Richard's aventures in two entangled wonderlands

Since the loophole-free Bell experiments of 2020 and the Nobel prizes in physics of 2022, critics of Bell's work have retreated to the fortress of super-determinism. Now, super-determinism is a derogatory word - it just means "determinism". Palmer, Hance and Hossenfelder argue that quantum mechanics and determinism are not incompatible, using a sophisticated mathematical construction based on a subtle thinning of allowed states and measurements in quantum mechanics, such that what is left appears to make Bell's argument fail, without altering the empirical predictions of quantum mechanics. I think however that it is a smoke screen, and the slogan "lost in math" comes to my mind. I will discuss some other recent disproofs of Bell's theorem using the language of causality based on causal graphs. Causal thinking is also central to law and justice. I will mention surprising connections to my work on serial killer nurse cases, in particular the Dutch case of Lucia de Berk and the current UK case of Lucy Letby.
4. An Overview of Sugarcane White Leaf Disease in Vietnam.pdf

An overview of Sugarcane White Leaf Disease in Vietnam
Citrus Greening Disease and its Management

Citrus Greening was one of the major causes of decline in the citrus production. So, effective management cultural practices should be incorporated
Multi-source connectivity as the driver of solar wind variability in the heli...

The ambient solar wind that flls the heliosphere originates from multiple
sources in the solar corona and is highly structured. It is often described
as high-speed, relatively homogeneous, plasma streams from coronal
holes and slow-speed, highly variable, streams whose source regions are
under debate. A key goal of ESA/NASA’s Solar Orbiter mission is to identify
solar wind sources and understand what drives the complexity seen in the
heliosphere. By combining magnetic feld modelling and spectroscopic
techniques with high-resolution observations and measurements, we show
that the solar wind variability detected in situ by Solar Orbiter in March
2022 is driven by spatio-temporal changes in the magnetic connectivity to
multiple sources in the solar atmosphere. The magnetic feld footpoints
connected to the spacecraft moved from the boundaries of a coronal hole
to one active region (12961) and then across to another region (12957). This
is refected in the in situ measurements, which show the transition from fast
to highly Alfvénic then to slow solar wind that is disrupted by the arrival of
a coronal mass ejection. Our results describe solar wind variability at 0.5 au
but are applicable to near-Earth observatories.
filosofia boliviana introducción jsjdjd.pptx

La filosofía boliviana y la búsqueda por construir pensamientos propios
Recently uploaded (20)
erythropoiesis-I_mechanism& clinical significance.pptx

erythropoiesis-I_mechanism& clinical significance.pptx
Body fluids_tonicity_dehydration_hypovolemia_hypervolemia.pptx

Body fluids_tonicity_dehydration_hypovolemia_hypervolemia.pptx
Observation of Io’s Resurfacing via Plume Deposition Using Ground-based Adapt...

Observation of Io’s Resurfacing via Plume Deposition Using Ground-based Adapt...
What is greenhouse gasses and how many gasses are there to affect the Earth.

What is greenhouse gasses and how many gasses are there to affect the Earth.
Lateral Ventricles.pdf very easy good diagrams comprehensive

Lateral Ventricles.pdf very easy good diagrams comprehensive
ESR_factors_affect-clinic significance-Pathysiology.pptx

ESR_factors_affect-clinic significance-Pathysiology.pptx
Comparative structure of adrenal gland in vertebrates

Comparative structure of adrenal gland in vertebrates
4. An Overview of Sugarcane White Leaf Disease in Vietnam.pdf

4. An Overview of Sugarcane White Leaf Disease in Vietnam.pdf
Multi-source connectivity as the driver of solar wind variability in the heli...

Multi-source connectivity as the driver of solar wind variability in the heli...
In silico drugs analogue design: novobiocin analogues.pptx

In silico drugs analogue design: novobiocin analogues.pptx
Circulatory system_ Laplace law. Ohms law.reynaults law,baro-chemo-receptors-...

Circulatory system_ Laplace law. Ohms law.reynaults law,baro-chemo-receptors-...
Igcse biology edexcel 2.39 2.43
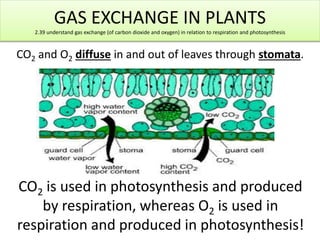
- 1. GAS EXCHANGE IN PLANTS 2.39 understand gas exchange (of carbon dioxide and oxygen) in relation to respiration and photosynthesis CO2 and O2 diffuse in and out of leaves through stomata. CO2 is used in photosynthesis and produced by respiration, whereas O2 is used in respiration and produced in photosynthesis!
- 2. Photosynthesis & Respiration 2.39 understand gas exchange (of carbon dioxide and oxygen) in relation to respiration and photosynthesis Both processes run all the time. So the net amount of glucose the plant produces (i.e. the amount it gets to use for growth etc) is governed by the formula; Net Glucose = Total production – Amount used in respiration The Compensation Point is defined as; 0 = Total production – Amount used in respiration Or Photosynthesis = Respiration
- 3. Light Intensity & Gas Exchange 2.40 understand that respiration continues during the day and night, but that the net exchange of carbon dioxide and oxygen depends on the intensity of light (TA) The amount of glucose the plant uses in respiration in nearly constant. However, glucose production by photosynthesis is not. It is dependent on the rate-limiting factors (i.e. light intensity, CO2 level, water availability, temperature etc). At night photosynthesis is virtually zero. (Net Carbon Dioxide production) In the day the photosynthesis is large. (Net Oxygen production)
- 4. Light Intensity & Gas Exchange 2.40 understand that respiration continues during the day and night, but that the net exchange of carbon dioxide and oxygen depends on the intensity of light (TA)
- 5. Leaf Structure and Photosynthesis 2.41 explain how the structure of the leaf is adapted for gas exchange
- 6. Stomata and Gas Exchange 2.42 describe the role of stomata in gas exchange In sunlight the guard cell becomes turgid Turgid guard cells open the stoma Increases gas exchange Low light causes guard cells to become flaccid Flaccid guard cells close the stoma Decreases water loss
- 7. Stomata and Gas Exchange 2.42 describe the role of stomata in gas exchange Exp 1 Exp 2
- 8. Stomata and Gas Exchange 2.42 describe the role of stomata in gas exchange Potometer 1) You must cut the shoots under water and you must assemble the potometer under water. If air gets into the xylem vessels of the plant, it can form air locks which will prevent the plant taking up water and so prevent steady transpiration. 1) Check all seals are airtight – coat seals with Vaseline jelly 3) The potometers should be left for the leaves to dry. Alternatively dry the leaves gently with a paper towel. The potometer will not work properly until any excess water on the leaves has evaporated or been removed. 2) Adding food colouring to the water makes it easier to see the air bubble in the capillary tube.
- 9. Stomata and Gas Exchange 2.42 describe the role of stomata in gas exchange
- 10. Experiment to Know 2.43 describe experiments to investigate the effect of light on net gas exchange from a leaf, using hydrogen-carbonate indicator (TA) An experiment which will show the effect of light intensity on the rate of gas exchange. - Seal two leaves (still attached to the plant) in separate plastic bags with some bicarbonate indicator solution. - One of the bags is black and the other is translucent. - The leaf in the black bag produces CO2 via respiration and the colour of the bicarbonate indicator changes quickly to yellow. - The leaf in the translucent bag produces O2 via photosynthesis and the bicarbonate indicator solution changes to red slowly. Bicarbonate Indicator colours: Red in the absence of CO2 Yellow in the presence of CO2
