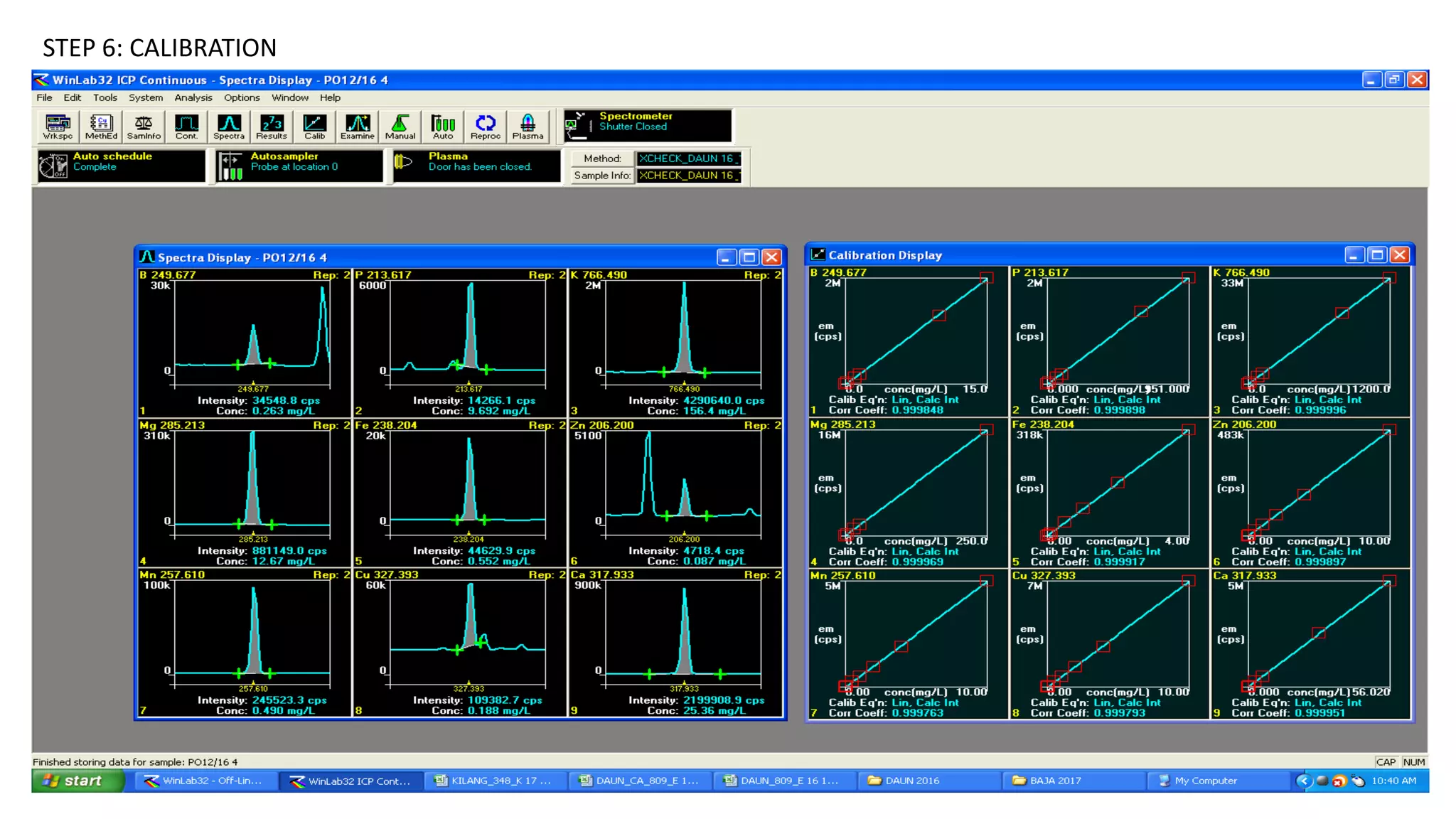
This document provides information about industrial training on inductively coupled plasma optical emission spectrometry (ICP-OES). It discusses the theory behind ICP-OES, including that it is a type of emission spectroscopy that uses inductively coupled plasma to excite sample elements, and the intensity of emission is used to determine concentration. It also outlines the sample preparation, instrumentation components like the nebulizer and torch, operating procedures for the ICP-OES, an example analysis of fertilizer samples, and calculations for determining element concentrations.












![DATA AND CALCULATION: BASED ON ONE SAMPLE
OF FERTILIZER
Sample ID A/S Loc Date Time
Analyte
Name Int (Corr)
RSD
(Corr Int)
SD (Corr
Int)
Conc
(Calib)
Conc
(Samp)
Reported
Conc
(Calib) Reported Conc (Samp)
Calib Blank 1 1 05-02-17 1:52:54 PM P 213.617 14.4235457 6.457346 0.931378 [0.00]
Calib Blank 1 1 05-02-17 1:52:54 PM K 766.490 18820.7249 2.285483 430.1444 [0.00]
Calib Blank 1 1 05-02-17 1:52:54 PM Mg 285.213 -463.70613 6.533916 30.29817 [0.00]
Calib Blank 1 1 05-02-17 1:52:54 PM B 249.677 30111.9796 0.244815 73.71873 [0.00]
S1 33 05-02-17 1:57:27 PM P 213.617 351082.482 0.671362 2357.034 [158.4]
S1 33 05-02-17 1:57:27 PM K 766.490 6006884.89 0.581678 34940.74 [200]
S1 33 05-02-17 1:57:27 PM Mg 285.213 2299070.57 0.009609 220.9133 [30]
S1 33 05-02-17 1:57:27 PM B 249.677 360518.244 0.765572 2760.028 [2]
S2 34 05-02-17 2:01:15 PM P 213.617 1537716.75 0.056084 862.4073 [634.17]
S2 34 05-02-17 2:01:15 PM K 766.490 25495615.6 0.424443 108214.3 [800]
S2 34 05-02-17 2:01:15 PM Mg 285.213 11193576.8 0.859641 96224.63 [150]
S2 34 05-02-17 2:01:15 PM B 249.677 1989438.61 1.955102 38895.55 [10]
S3 35 05-02-17 2:05:03 PM P 213.617 2267715.79 0.252416 5724.087 [951]
S3 35 05-02-17 2:05:03 PM K 766.490 37909821.9 0.816761 309632.8 [1200]
S3 35 05-02-17 2:05:03 PM Mg 285.213 17467052 0.27787 48535.69 [250]
S3 35 05-02-17 2:05:03 PM B 249.677 3050847.97 0.653446 19935.66 [15] %P2O5 %K2O %MgO %B2O3
QC F15/10 5 05-02-17 2:08:56 PM P 213.617 257151.976 2.530687 6507.711 110.9262 110.9262 110.9 110.9 5.07922
QC F15/10 5 05-02-17 2:08:56 PM K 766.490 27132931.3 0.079279 21510.65 857.8097 857.8097 857.8 857.8 20.67298
QC F15/10 5 05-02-17 2:08:56 PM Mg 285.213 5155926.77 1.686524 86955.92 70.70965 70.70965 70.71 70.71 2.347572
QC F15/10 5 05-02-17 2:08:56 PM B 249.677 862541.565 0.646395 5575.429 4.35837 4.35837 4.358 4.358 0.280655
BLANK 38 05-02-17 2:12:53 PM P 213.617 86.9382419 18.04484 15.68787 4.097573 4.097573 4.098 4.098 0.000938
BLANK 38 05-02-17 2:12:53 PM K 766.490 14654.35 3.597323 527.1643 4.292732 4.292732 4.293 4.293 0.000517
BLANK 38 05-02-17 2:12:53 PM Mg 285.213 36282.3952 0.113541 41.19531 -2.18262 -2.18262 -2.183 -2.183
-
0.000362
BLANK 38 05-02-17 2:12:53 PM B 249.677 30602.727 1.794585 549.1918 0.278291 0.278291 0.278 0.278 8.95E-05
QC F15/10 39 05-02-17 2:17:20 PM P 213.617 250696.412 0.442589 1109.555 108.2434 108.2434 108.2 108.2 4.95556
QC F15/10 39 05-02-17 2:17:20 PM K 766.490 26057616.9 0.948126 247059 823.9654 823.9654 824 824 19.8584
QC F15/10 39 05-02-17 2:17:20 PM Mg 285.213 4935491.66 0.368125 18168.8 67.57115 67.57115 67.57 67.57 2.243324
QC F15/10 39 05-02-17 2:17:20 PM B 249.677 723577.686 0.783142 5666.643 3.676849 3.676849 3.677 3.677 0.236799
BKILANG_R4_272/K_17 40 05-02-17 2:21:11 PM P 213.617 131308.767 0.221019 290.2168 58.62947 58.62947 58.63 58.63 2.685254
BKILANG_R4_272/K_17 40 05-02-17 2:21:11 PM K 766.490 10553758.9 0.750208 79175.18 335.9991 335.9991 336 336 8.0976
BKILANG_R4_272/K_17 40 05-02-17 2:21:11 PM Mg 285.213 1123883.39 0.329213 3699.975 13.30239 13.30239 13.3 13.3 0.44156
BKILANG_R4_272/K_17 40 05-02-17 2:21:11 PM B 249.677 34466.5758 1.223829 421.8121 0.29724 0.29724 0.297 0.297 0.019127](https://image.slidesharecdn.com/icp-oes-170301010048/75/Icp-oes-14-2048.jpg)

![• FACTOR P2O5
= (Molecular Weight P2O5)
Atomic Weight 2P
= [2(30.973762) + 5(15.9994)]
2(30.973762
= 2.29
CALCULATION: FACTOR P2O5](https://image.slidesharecdn.com/icp-oes-170301010048/75/Icp-oes-16-2048.jpg)
