Human Health Risk Assessment Training Module
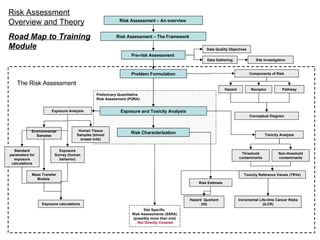
- 1. Risk Assessment – An overview Risk Assessment – The Framework Problem Formulation Exposure and Toxicity Analysis Risk Characterization Conceptual Diagram Components of Risk Exposure Analysis Toxicity Analysis Environmental Samples Threshold contaminants Non-threshold contaminants Risk Assessment Overview and Theory Road Map to Training Module Exposure Survey (human behavior) Human Tissue Samples (blood/ breast milk) Risk Estimate Hazard Receptor Pathway Incremental Life-time Cancer Risks (ILCR) Hazard Quotient (HI) Pre-risk Assessment The Risk Assessment Preliminary Quantitative Risk Assessment (PQRA) Site Specific Risk Assessments (SSRA) (possibly more than one) - Not Directly Covered Data Gathering Data Quality Objectives Site Investigation Standard parameters for exposure calculations Mass Transfer Models Exposure calculations Toxicity Reference Values (TRVs)
- 2. Risk Assessment – An overview (1) Risk assessment is an iterative process leading to the quantification of potential risk. For the POPs project, the emphasis is on risk assessments assessing health risks to humans resulting from exposure to persistent organic chemicals. The assessment looks at multiple exposure scenarios and calculates the incremental risks associated with each scenario, as well as the overall risk attributable to all the scenarios combined. The detailed information provided by risk assessments are needed before appropriate risk management measures can be selected and implemented.
- 3. Risk Assessment – An overview (2) Risk assessment is a process that builds upon itself. Risk assessments start off very simple, using a minimum of data, making simple assumptions and using simple calculations. At this initial stage, all assumptions should err on the side of caution. If the earliest iterations of the risk assessment predicts risk, it doesn’t necessarily mean that there is risk, but additional work needs to be done to improve the risk assessment. In Canada, the earliest iteration is called a Preliminary Quantitative Risk Assessment (PQRA) (Health Canada 2004) . In subsequent iterations of the risk assessment, more data is required, assumptions become more refined, and calculations become more complicated. These subsequent iterations are each called Site Specific Risk Assessments (SSRA) . The iterations/improvements to the risk assessment continue until it is apparent that the risk is negligible or there is potential for risk and the risk assessment can not be refined further or there is potential risk and the risk can be effectively managed. <<Jason please link to the figure two slides forward>>
- 5. Problem Formulation Exposure and Toxicity Analysis Risk Characterization Pre-risk Assessment Preliminary Quantitative Risk Assessment (PQRA) Site Specific Risk Assessment (SSRA) #2 and so on…. Exposure and Toxicity Analysis Risk Characterization Site Specific Risk Assessment (SSRA) #1 No further action Negligible Risk No further action Negligible Risk No further action Negligible Risk Possible Risk Risk Management Gather additional data Refine risk calculations Possible Risk Risk Management Gather additional data Refine risk calculations Risk Assessment Framework Focus of the POPs training Risk Assessment
- 6. Preliminary Quantitative Risk Assessments As its name suggests, a Preliminary Quantitative Risk Assessment (PQRA) is generally the first iteration of a risk assessment. The methods and assumptions prescribed in a PQRA are conservative and generally ensure that risks are not underestimated. Thus, if acceptable or negligible risks are predicted, then it is almost certain that risks are either acceptable or negligible. However, the opposite may not be true. If a PQRA suggests the potential for unacceptable risks, it does not mean that unacceptable risks are present. Instead, further assessment may be necessary to resolve conservatism and uncertainty in risk calculations before the actual health risk can be defined. When risk management activities are implemented based on a PQRA, the result will generally be a greater reduction of risk than was necessary. A more detailed risk assessment will usually result in risk management activities that are more in line with actual risks. The complexity of a risk assessment is often decided based on cost and the feasibility of the proposed risk management approach. More detailed risk assessments are called Site Specific Risk Assessments (SSRA) . Generally in a risk assessment there is only one PQRA iteration. However, there may be multiple SSRA iterations each building upon the last one. In the POPs training package we present a risk assessment approach consistent with a PQRA. Additional reading and/or training will be required before participants can successfully conduct a SSRA.
- 8. Data Quality Objectives (DQO) Data Quality Objectives provide the criteria that a data collection design should satisfy, including: when to collect samples, where to collect samples, the maximum level of error for the study, and how many samples to collect. Using the DQO Process will assure that the type, quantity, and quality of environmental data used in the risk assessment and/or risk management will be appropriate, resulting in decisions that are technically and scientifically defensible. In addition, the DQO Process will help prevent the collection of unnecessary data (USDOE 2008). For more information on DQO’s please visit: http://www.hanford.gov/dqo/index.html
- 9. Data Gathering Before staring the risk assessment process, certain types of data will be required. If previous investigations have been done at the site, some of the required data will be available in printed material. Other information, such as former site use may be available in historical literature. In addition to printed material, a site investigation is always recommended (Environment Canada 2003, USEPA 2008). By visiting the site the Risk Assessor can collect information which may not be available in the printed literature.
- 10. Site Investigation A site investigation can provide the risk assessor with data necessary to carry out a risk assessment. Information can include: Visual Observations – i.e., What might be the contaminant of concern? What organisms might be exposed? How might organisms be exposed? Are there areas where soils are stained? Interviews With People On site – i.e., What are the human behaviors at the site which might result in exposure? What are the contaminants of concern? Have effects been observed? What is the duration or frequency of exposure? Collection Of Samples For Analysis – i.e., What are the concentrations of contaminants in soils, sediments, water or tissue? Are there unexpected contaminants of concern? What is the spatial distribution of contaminants? More information will be provided regarding the data used in a risk assessment in the problem formulation section .
- 11. Problem Formulation The very first step in the risk assessment is the problem formulation. The problem formulation defines the problem, explicitly identifies the components and sets the stage for the Risk Assessment (Environment Canada 2003, Health Canada 2004). <<components on the next slide>>
- 12. Components Needed to Have Risk ( Chemical Hazard , Receptor and Pathway ) Who is being exposed? Workers? Families? Children? Are animals or fish exposed? Are people eating contaminated food? Are they inhaling contaminated dust? Are they coming in contact with the chemical via skin contact? What chemical is of concern? Receptor Pathway Chemical Hazard Risk
- 13. Hazard To determine if a Hazard exists, existing chemical data is screened against environmental quality guidelines (e.g., the CCME environmental quality guidelines and Health Canada drinking water guidelines). Concentration data is first summarized by calculating the mean, 95% upper confidence limit of the mean (UCLM), 95 th percentile and maximum concentration. The summary statistics are then screened against the environmental quality guidelines. In addition to statistical summaries of concentrations, other information may need to be collected (e.g., for soil data ).
- 14. Environmental Quality Guidelines Environmental Quality Guidelines are concentration limits for contaminants or environmental quality characteristics (such as dissolved oxygen or pH), which, if not met, may affect humans or the environment (CCME 2006). For the POPs program, the CCME Environmental Quality Guidelines for the protection of human health have been recommended as the principle guidelines for screening purposes. Like many environmental quality guidelines, the CCME Guidelines are based on scientific studies in which humans or animals were exposed to the contaminant in question at various concentrations and a toxic effect was observed. If an animal study is used to represent potential effects to humans, a scaling factor accounting for difference in body weight is used. In addition, safety factors are applied to account for uncertainty, such as the relative sensitivity of animals and humans to a contaminant (CCME 2006). In the case of guidelines protective of human health, once scaling and safety factors have been taken into consideration, the guideline is calculated from the toxicological reference value (TRV). This is the highest total dose of a given chemical that is predicted not to result in a toxic effect in humans. The TRV is also referred to as the tolerable daily intake (TDI), reference dose (RD) or acceptable daily intake (ADI). Guidelines are back calculated from the TRV, by making a number of assumptions about possible exposure scenarios and uptake efficiencies (CCME 2006). The CCME guidelines provide guidelines addressing most potential exposure scenarios. However CCME does not provide guidelines specific to potential contaminated groundwater exposure .
- 15. Screening Contaminants of Potential Concern (COPCs) are identified by screened environmental concentrations against the CCME Environmental Quality Guidelines for protection of human health, where possible. Where CCME human health guidelines are not available, other human health-based guidelines from reliable sources may be used, provided that guidelines for non-carcinogens are derived on the basis of 20% of the toxicological reference value (TRV). The CCME applies 20% of the TRV when setting guidelines for soil and other media. If no CCME human health-based environmental quality guideline for a particular contaminant exists, guidelines from other reputable sources can be used. One source is the U.S. Environmental Protection Agency’s preliminary remediation goals (PRGs)(U.S. Environmental Protection Agency [U.S. EPA], 2002). The PRG’s must be adjusted for non-carcinogens to reflect 20% of the U.S. EPA TRV (which the U.S. EPA calls a RfD). In the event that a contaminant has no corresponding health-based soil quality guideline, the contaminant should be included as a COPC for further risk assessment, unless the measured concentrations are consistent with natural or background concentrations (see Background Concentrations section).
- 16. Groundwater For contaminants in groundwater, the following screening approaches are recommended, depending on the potential exposure route. 1) If the groundwater may be used for drinking water purposes, the Health Canada Guidelines for Canadian Drinking Water Quality (http://www.hc-sc.gc.ca/hecs-sesc/water/index.htm) should be used for screening of COPCs. 2) If the groundwater is not used for drinking but may be used for other purposes, then the CCME water quality guidelines that best matches the intended purpose should be used (CCME 2006). 3) If the groundwater flows into a stream, lake or pond, it is often assumed that the groundwater will undergo a 10x dilution before discharging. Therefore for screening purposes, it is reasonable to screen groundwater concentrations in this scenario against CCME water quality criteria (for the protection of aquatic life) which have been multiplied by 10.
- 17. Background Concentrations Before a site is considered contaminated, concentrations of contaminants at the site, particularly natural elements (e.g., metals), should also be compared to background soil and groundwater concentrations (and surface water concentrations, if relevant), if data are available. If it is found that concentrations of contaminants at the site are representative of background levels, then the site may not be contaminated even though measured concentrations are greater than the guidelines. A further discussion of background levels is provided in Appendix A of the PQRA guidance document (Health Canada 2004).
- 18. Additional Data (e.g., Soil Data) For soil samples, the depth at which samples were collected should be indicated. A map of sampling locations is often helpful to determine if the collected samples reflect the distribution of contaminants across the property (Health Canada 2004).
- 19. Receptor Receptors are biological organisms that may be affected by exposure to a chemical hazard. Chemical-related risk assessments are generally divided into human health risk assessments and environmental risk assessments . Human health risk assessments focus on humans as receptors ; environmental risk assessments focus on all other biological organisms as individuals as well as ecological functioning.
- 20. Humans as Receptors Humans are often subdivided on the basis of age group (Health Canada 2004). Typical age groups are as follows: infants (0 to 6 months), toddlers (7 months to 4 years), child (5 years to 11 years), teen (12 – 19 years) and adults (20+ years). Age groups are assessed separately because many of the factors determining the degree of exposure are different. In addition, certain age groups are more susceptible to chemically mediated effects (i.e., infants, toddlers and pregnant woman are often the most sensitive to chemical exposure)(Health Canada 2004).
- 21. Exposure pathway An exposure pathway is the route a chemical hazard takes to reach (and potentially affect) a receptor (Environment Canada 2003, Health Canada 2004). Exposure pathways include: Physical mechanisms – for instance contaminated soil being washed into a nearby creek and potentially affecting sediment dwelling organisms. Human behavior – for instance contaminated material can be moved by people from one location to another; contaminated soil on a truck’s tires or people bring PCB containing oils home to be burned in cooking fires. Biological mechanisms – for instance dermal contact with contaminated soil, ingestion of contaminated food, inhalation of dust, etc.
- 22. Conceptual Diagram Once all three of the risk components have been identified, they are summarized in a Conceptual Diagram (Environment Canada 2003, Health Canada 2004). There are two types of conceptual diagram: a graphical conceptual diagram and a box and line conceptual diagram . Both are acceptable and the choice of which one is used depends on personal preference.
- 23. Conceptual Diagram – Graphical
- 24. Conceptual Diagram – Box and Line Medium Route Receptor Air Water Food Soil Dermal Ingestion Inhalation Human being
- 25. Exposure and Toxicity Analysis After the problem formulation is the exposure and toxicity analysis . The exposure and toxicity Analysis quantifies exposure (or dose) as well as the toxic potency of the chemical hazard(s). Information from the Exposure and Toxicity Analysis goes directly into the last step of the risk assessment, called a risk characterization (Health Canada 2004).
- 26. Exposure Analysis The exposure assessment attempts to quantify the exposure of a receptor to the hazard via a given pathway. The daily intake of a contaminant via individual pathways can then be summed to estimate the total daily intake (TDI) (Health Canada 2004). Exposure via individual exposure pathways can be calculated using contaminant concentrations in environmental samples , human behavior information from exposure surveys combined with standard exposure parameters . Alternatively, total exposure (for POPs) can be estimated by collecting human tissue samples for chemical analysis (i.e., blood and breast milk samples). Health Canada’s Preliminary Quantitative Risk Assessment (PQRA) guidance, provides an excel spread sheet-based model which simplifies the calculation of total daily intake (TDI) (Health Canada 2004). Dermal exposure Inhalation Diet (Dust)
- 29. Mass Transfer Models Models may be necessary to estimate the concentrations of contaminants in groundwater, surface water, air, fruits, vegetables, fish, meat or other environmental media through which receptors may be exposed. If used for a Preliminary Quantitative Risk Assessment, mass transfer models should be fairly simple. Estimates of the concentrations of volatile COPCs in indoor air should be derived from the methods presented by Williams et al. (1996) and the CCME (1996 - Appendix G). Likewise, estimating COPC concentrations in groundwater and in surface water may be obtained from the methods described by the CCME (1996). For estimating COPC concentrations in fruits or vegetables, methods presented by the CCME (1996) or the Oak Ridge National Laboratory (ORNL) (1998) may be used. For estimating COPC concentrations in fish and wildlife, simple bioaccumulation/biomagnification factors may be used (Health Canada 2004). More complicated modelling is usually not necessary, but may be used.
- 31. Blood/Breast Milk Sampling The assessment of concentration in blood and breast milk can be useful for contaminants that bioaccumulate (i.e., all POPs). These samples can be used to verify the total predicted exposure calculated from numerous different exposure pathways (Health Canada 2004). Unfortunately, these samples provide little information regarding the importance of individual exposure pathways and therefore the value to risk management decision making is limited.
- 32. Standard Exposure Parameters Calculation of exposure will require the inclusion of exposure parameters. Some of these numbers can be derived from the human exposure survey; however, many will not be easily available (Health Canada 2004). Fortunately, Health Canada provides a table of standard exposure parameters which can be used in the absence of information collected at the site. More… <<Please insert link to table 3 and 4 from part 1 of the PQRA manual, table 4 may need modifying to suit the SE Asian perspective>>
- 33. More… Other sources of Standard Exposure Parameters include: Richardson (1997), U.S. EPA Exposure Factors Manual (U.S. EPA, 1997).
- 35. More… Duration of Exposure Regarding the duration of exposure, inhalation exposures are generally based on the time spent in the contaminated environment, while soil ingestion exposures are not dependant of the time spent outdoors. Inhalation of Dust The inhalation of dust is usually insignificant relative to direct ingestion of soil and water, and to dermal absorption. However, in some cases the inhalation pathway can become important. In most cases, the concentration of a contaminant in the respirable airborne dust should be assumed to be equal to the concentration in surface soil (maximum or average). The average airborne concentration of respirable (≤ 10 μm aerodynamic diameter) particulate matter should be assumed to be 0.76 μg/m3 (based on U.S. EPA, 1992). For situations where vehicle traffic on contaminated unpaved roads can be a significant concern, a reasonable dust level created by vehicle traffic on unpaved roads is 250 μg/m3 (down-wind side of the road; Claiborn et al., 1995).
- 36. More… Dermal Exposure Toxicological reference values (TRVs) for the dermal exposure pathway are not commonly available. Therefore, dermal exposures are generally added to the ingested dose, once adjustments are made accounting for differences in absorption (see Relative Absorption Factors below). Exposure via Multiple Pathways In many cases the TRVs for oral, dermal and inhalation exposures are combined. In cases where TRVs for oral and inhalation exposures are separate, calculations for these pathways should be performed separately. In cases where only an oral TRV is available, exposures by all routes (oral, dermal, inhalation) should be summed for comparison to the oral TRV. Relative Absorption Factors (RAFs) If multiple exposure pathways will be summed for comparison to a single TRV, it will be necessary to apply relative absorption factors (RAFs) in exposure calculations. Oral exposures should always be assumed to have a relative absorption of 100% (RAF = 1). Where inhalation exposures are being summed with oral exposures, the inhalation RAF will generally default to 1 unless there is a good reason for respiratory absorption to be significantly less that 100%. Where dermal exposures are being summed with oral exposures, the RAF values presented in Table 6 should be applied. Other sources of RAF values include the Risk Assessment Information System (RAIS; http://risk.lsd.ornl.gov/rap_hp.shtml), Toxicological Profiles published by the Agency for Toxic Substances and Disease Registry (ATSDR; http://www.atsdr.cdc.gov/toxpro2.html), or other sources should be consulted.
- 37. Toxicity Assessment The toxicity assessment attempts to quantify the sensitivity of the receptor to the chemical hazard. In a Preliminary Quantitative Risk Assessment, the toxicity assessment typically consists of choosing the correct toxicity reference value ( TRV ) for a given exposure. TRVs generally can be used for multiple routes of exposure (i.e., dietary, inhalation or dermal contact), but for some contaminants, separate TRVs are provided for ingestion and inhalation pathways. <<Please provide link to Health Canada TRVs in: Health Canada PQRA Guidance Part II TRVs.pdf located at: V:atarojectsOP1406oolkitisk Assessment ModelsQRA>> The assessment takes into consideration the mode of action of the hazard, the toxic potency as observed in scientific studies and physiological/biochemical factors which might moderate the toxic potency for a given receptor. Generally, contaminants resulting in a chemical hazard are considered either as threshold or non-threshold contaminants. Each is assessed differently in a human health risk assessment (Health Canada 1994).
- 38. Threshold Contaminants A threshold response is characterized by a toxic effect occurring over a narrow range of exposure concentrations. Lower concentrations do not result in a toxic effect. Most environmental contaminants are threshold contaminants. The maximum allowable exposure concentrations, called the “Exposure Limits” (or “Toxicity Reference Values”) are based on the threshold determined from toxicity experiments. These experiments generally used animals, not humans. Usually the Exposure Limit incorporates a “Uncertainty Factor” (or “Safety factor”) to account for uncertainties in the estimate. The Exposure Limit for a Threshold Contaminant is generally presented as a Tolerable Daily Intake value (TDI; mg chemical/kg body weight/day) (Health Canada 1994). Threshold Concentration Chemical concentration Incidence of toxic response
- 39. Non-Threshold Contaminants The non-threshold response can occur over a very wide range of concentrations. Generally, the higher the concentration, the greater probability of an effect. Mutagens are generally considered non-threshold contaminants. The maximum allowable exposure concentrations, called the “Exposure Limits” are based on the probability of an incremental effect during a typical human life span. The maximum allowable incremental probability is generally an concentration resulting in a incremental probability increase of one in one million. The Exposure Limit for a Non-threshold Contaminant is generally presented as a Slope Factor [(mg chemical/kg body weight/day) -1 ] (Health Canada 1994). Chemical concentration Incidence of toxic response
- 40. TRVs Health Canada TRVs should be applied where available (Health Canada, 2003). For substances with no Health Canada TRVs, reference doses (RfDs), reference concentrations (RfCs), acceptable daily intakes (ADIs), or minimum risk levels (MRLs) should be obtained from the following agencies, in order of preference (Health Canada 2004): 1) U.S. EPA Integrated Risk Information System (IRIS); http://www.epa.gov/iriswebp/iris/index.html 2) World Health Organization (WHO); various sources including: http:// www.inchem.org / ; http:// jecfa.ilsi.org/index.htm ; http://www.who.dk/air/activities/20020620_1 ) 3) Netherlands National Institute of public Health and the Environment (RIVM); http://www.rivm.nl/bibliotheek/rapporten/711701025.pdf 4) Agency for Toxic Substances and Disease Registry (ATSDR) (U.S.); http://www.atsdr.cdc.gov/toxpro2.html
- 41. Risk Characterization The final component of the risk assessment is the risk characterization. The risk characterization integrates the information from the exposure and toxicity analysis to derive a quantitative estimate of human health risk. This is normally accomplished by calculating an exposure ratio called a hazard quotient (HQ) for threshold contaminants or a incremental life-time cancer risk (ILCR) for non-threshold contaminants. If risks are predicted at the risk characterization stage, the risk assessment process is repeated using additional data, refined assumptions and more complex equations.
- 42. Hazard Quotient For threshold contaminants , the risk to a human receptor from being exposed to a chemical via a single pathway can be expressed as an Exposure Ratio, commonly called a Hazard Quotient (HQ). HQ = Dose Rate/Reference Dose Reference Dose is commonly referred to as the Tolerable Daily Intake (TDI; mg/kg/day). A Hazard Index (HI) is the sum of HQ’s for all pathways and similar toxic effects. A HQ of <0.2 for any given pathway is often considered “acceptable”; while an HI of <1.0 is considered acceptable (Health Canada 2004).
- 43. Non-carcinogens: Single-Substance Exposures For substances presenting risks other than cancer, a Hazard Quotient (HQ; also called “exposure ratio” and “hazard ratio”) will be derived as the ratio of the estimated exposure (for each critical receptor) to the tolerable daily intake (TDI) or tolerable concentration (TC), as follows: Hazard Quotient = Estimated Exposure (μg/kg/day) Tolerable Daily Intake (μg/kg/day) OR, in the case of air-borne contaminants with a tolerable air concentration in (μg/m3) -1 : Hazard Quotient = Air Concentration (μg/m3) x Fraction of Time Exposed Tolerable Air Concentration (μg/m 3 ) Hazard Quotients for individual exposure pathways should be presented if pathway-specific TRVs are available. Where exposures via multiple pathways are being summed for comparison to a single TRV (for example, it is common to sum oral and dermal exposures for comparison to the oral TDI), it is necessary only to display the HQ for the summed exposure. For purposes of preliminary quantitative risk assessment, exposures associated with a HQ = 0.2 will be deemed negligible. This is consistent with the CCME (1996) and the OMEE (1996a), and has become accepted common practice (Health Canada 2004).
- 44. Carcinogens: Single-Substance Exposures For carcinogens, the estimated exposure will be multiplied by the appropriate Cancer Slope Factor or Unit Risk to derive an estimate of the potential incremental lifetime cancer risk (ILCR) associated with that exposure (Health Canada 2004). The ILCR is derived as: ILCR = Exposure (μg/kg/d) x Cancer Slope Factor (μg/kg/d) -1 OR, in the case of air-borne contaminants with a unit risk value in (μg/m3) -1 : ILCR = Air Concentration (μg/m3) x Fraction of Time Exposed x Cancer Unit Risk (μg/m3) -1 Where pathway-specific slope factors or unit risks exist, the risks via inhalation and the risks via oral + dermal exposure should be estimated separately. In other cases, the cancer risks posed by simultaneous inhalation/dermal/oral exposure will be estimated. Cancer risks will be deemed to be “essentially negligible” ( de minimus ) where the estimated ILCR is 1-in-100,000 (≤ 1 x 10 -5 )(Health Canada 2004).
- 45. Exposure to Mixtures For exposure to multiple chemicals, non-cancer Hazard Quotients should be assumed to be additive, and should be summed for those substances determined to have similar mechanisms of action. For the purposes of PQRAs, exposures associated with this total HQ # 0.2 will be deemed negligible. For carcinogens with the same target organ and form of cancer, the risks should be assumed to be additive and thus should be summed. The total cancer risk in such cases will be considered to be “essentially negligible” where the estimated total ILCR is # 1-in-100,000 (1 x 10 -5 ).
- 46. References Claiborn, C., et al. 1995. Evaluation of PM10 emission rates from paved and unpaved roads using tracer techniques. Atmos. Environ. 29(10): 1075-1089. Richardson, G.M., 1997. Compendium of Canadian Human Exposure Factors for Risk Assessment. Ottawa: O’Connor Associates Environmental Inc. Environment Canada, 2003. TABs on Contaminated Sites, Contaminated Sites Program - Federal Sites, Technical Assistance Bulletins (TABs) #15: Risk Assessment-Application & The Screening Process. Accessed on line on June 10, 2008 at http://www.on.ec.gc.ca/pollution/ecnpd/tabs/tab15-e.html. Health Canada, 1994. Human Health Risk Assessment for Priority Substances. ISBN: 0-662-22126-5, Cat. No.: En40-215/41E accessed on line on June 10, 2008 at http://www.hc-sc.gc.ca/ewh-semt/pubs/contaminants/approach/index-eng.php Health Canada, 2004. Federal Contaminated Site Risk Assessment in Canada Part I: Guidance on human health Preliminary Quantitative Risk Assessment (PQRA), Cat. H46-2/04-367E, ISBN 0-662-38244-7. Accessed on line on July 3, 2008 at http://www.hc-sc.gc.ca/ewh-semt/contamsite/risk-risque-eng.php United States Environmental Protection Agency (U.S. EPA). 1992. Risk Assessment Guidance for Superfund: Volume I – Human Health Evaluation Manual (Part B, Development of Risk-based Preliminary Remediation Goals). EPA/540/R-92/003, U.S. EPA, Washington, DC. United States Environmental Protection Agency (U.S. EPA). 1997. Exposure Factors Handbook, Volume I: General Factors; Volume II: Food Ingestion Factors; Volume III: Activity Factors. EPA/600/P-95/002Fa, U.S. EPA, Washington, DC. August 1997. United States Department of Energy (USDOE), 2008. Data Quality Objectives (DQO), Accessed on line on July 3, 2008 at: http://www.hanford.gov/dqo/index.html