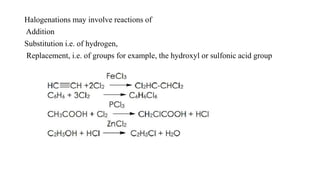
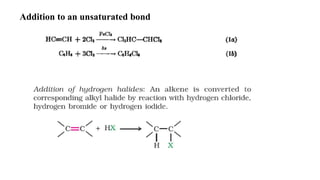
This document discusses halogenation reactions, which involve replacing a halogen atom with another substance or adding halogens to a substance. It describes three main types of halogenation reactions: addition to unsaturated bonds, substitution reactions where halogens replace hydrogen, and replacement reactions where halogens replace other groups. The document also discusses common halogenating agents used to catalyze these reactions like iron, antimony, and phosphorus compounds. Finally, it provides an overview of reaction kinetics and how rate equations are used to relate the rate of reaction to reactant concentrations and determine the order of reactions.











![• The rate equation (rate law) relates the concentrations of the reactants to
the rate of reaction.
• Consider: A + B → C + D
• Rate = Kr [A]a [B]b
• The rate is proportional to the concentrations of A and B raised to some
power (a and b)
• Where kr is a rate constant, and a and b have to be determined
experimentally.
• Values a and b are usually whole numbers, and are called the order of
the reaction.
• (a+b) is the overall order of a reaction. Kinetics and the Rate Equation](https://image.slidesharecdn.com/halogenation-231007050328-374f5cf5/85/HALOGENATION-pptx-14-320.jpg)
![• Consider the following reaction:
• CH3 -Br + -OH → CH3 -OH + Br-
• Experiments show that doubling the concentration of bromomethane,
doubles the rate of reaction. Also doubling the [- OH] doubles the rate.
• Therefore the rate is proportional to both [CH3Br] and [-OH], and the rate
equation would read:
• Rate = kr [CH3Br] [-OH]
• The rate is said to be first order w.r.t. bromomethane, and first order w.r.t.
hydroxide ion.
• The reaction is second order overall.](https://image.slidesharecdn.com/halogenation-231007050328-374f5cf5/85/HALOGENATION-pptx-15-320.jpg)
![• However, similar reactions can have different kinetic orders. (CH3
)3C-Br + -OH → (CH3 )3C-OH + Br-
• For this reaction, doubling the [t-butylbromide] doubles the rate, but
doubling [-OH] has no effect on the rate.
• Rate = kr [(CH3 )3C-Br]
• First order w.r.t. t-butylbromide, but zeroth order w.r.t. hydroxide ion.
The order has to be obtained experimentally.](https://image.slidesharecdn.com/halogenation-231007050328-374f5cf5/85/HALOGENATION-pptx-16-320.jpg)

