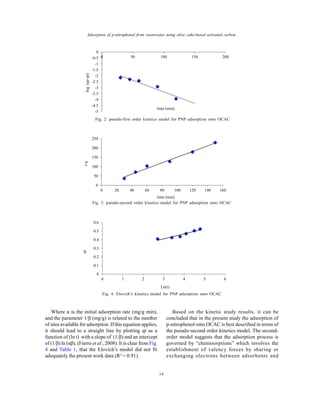
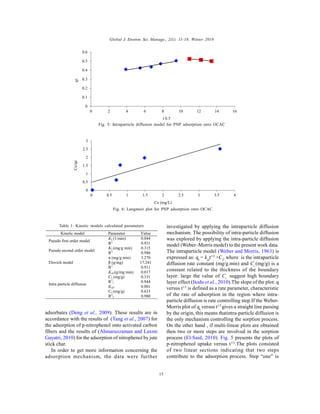
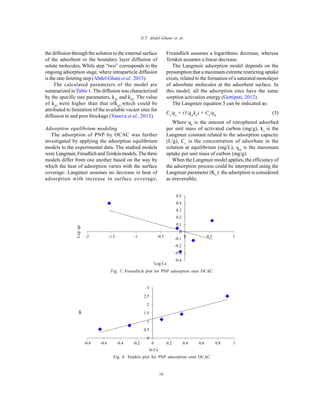
The study evaluates the removal of p-nitrophenol from wastewater using activated carbon derived from olive cake, achieving adsorption efficiency with an optimal surface area of 672 m2/g. The adsorption kinetics were best described by a pseudo-second-order model, indicating chemisorption processes, with a notable equilibrium time of 120 minutes. This research highlights olive cake-based activated carbon as an effective and sustainable adsorbent for organic pollutants.