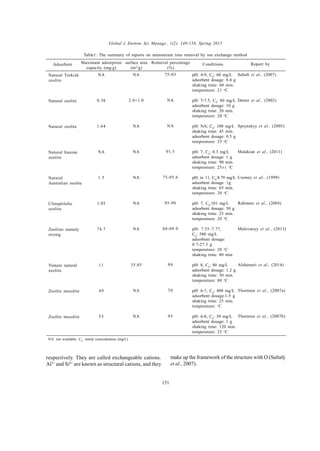
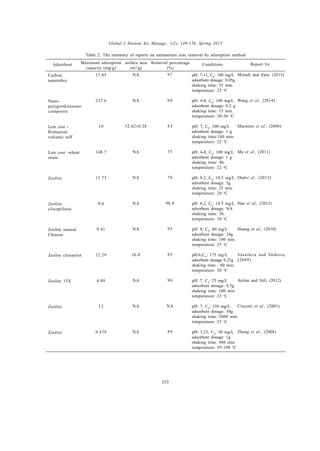
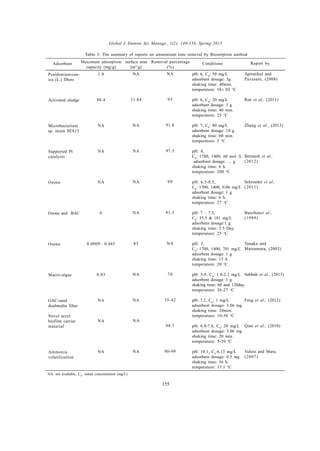
This document reviews methods for the removal of ammonium ions from wastewater, addressing the environmental challenges posed by these pollutants. Various techniques are evaluated, including ion exchange, adsorption, biosorption, and biological methods, with particular emphasis on natural zeolites and their effectiveness. The review compiles data from about 75 studies on these removal techniques and highlights the increasing research focus on low-cost and efficient adsorbents.