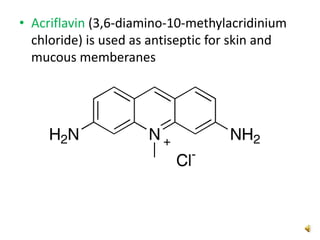
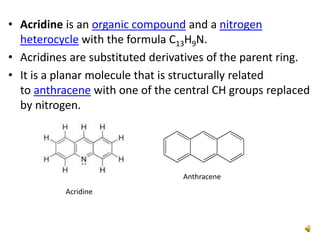
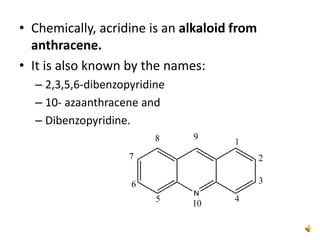
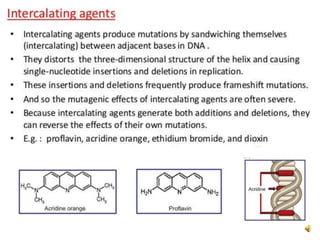
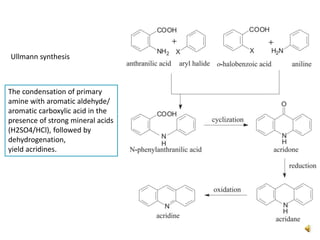
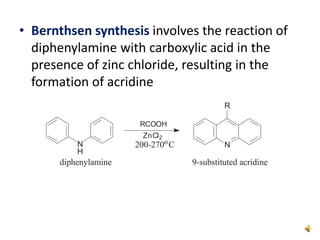
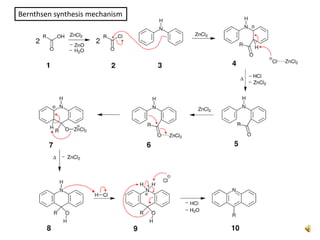
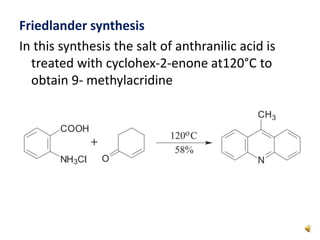
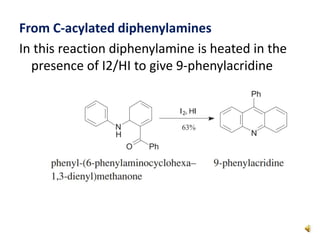
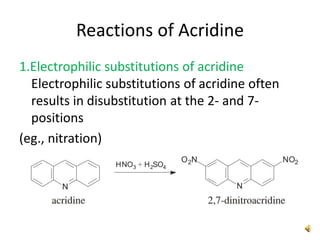
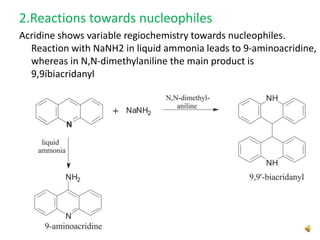
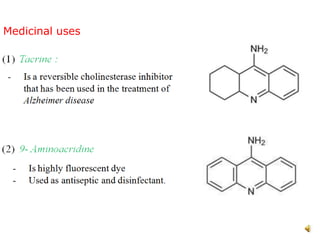
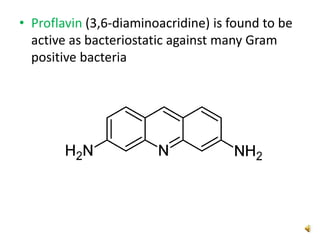
Acridine is an organic nitrogen heterocycle with various pharmaceutical applications, first isolated from coal tar in 1870. It exhibits antibacterial properties discovered in 1917 and has seen a resurgence in interest due to rising drug-resistant bacterial infections. The document discusses its chemical properties, structure, synthesis methods, and medicinal uses including anesthetics and antimalarial agents.







![• Bucricaine [butyl-(1,2,3,4-tetrahydroacridin-9-
yl)- amine] is used topically for surface
anesthesia of eye and given by injection for
infilteration anesthesia, peripheral nerve block
and spinal anesthesia.](https://image.slidesharecdn.com/fusedheterocycliccomponds-acridine-200709102043/85/Fused-heterocyclic-componds-acridine-24-320.jpg)
![• Quinacrine [2-methoxy-6-chloro-9-(1-diethylamino-
3-methylpropanamine)-acridine] is also known as
mepacrine. It acts as gametocytocide. It destroy the
sexual erythrocytic forms of plasmodia and act as
antimalarial agent](https://image.slidesharecdn.com/fusedheterocycliccomponds-acridine-200709102043/85/Fused-heterocyclic-componds-acridine-25-320.jpg)
![• Nitracrine [1-nitro-9-
(dimethylaminopropylamino)acridine] causes the
DNA damage and acts as an anticancer agent](https://image.slidesharecdn.com/fusedheterocycliccomponds-acridine-200709102043/85/Fused-heterocyclic-componds-acridine-27-320.jpg)