Embed presentation
Downloaded 17 times
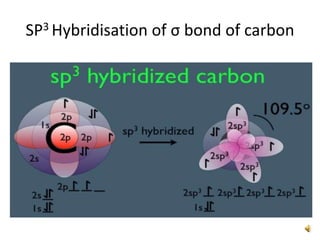
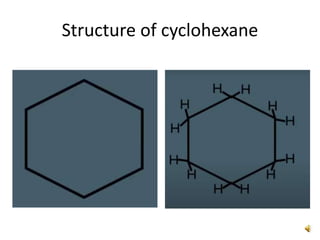

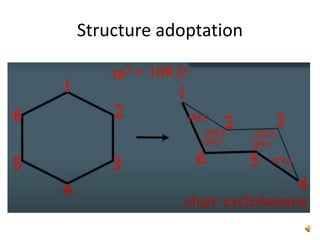
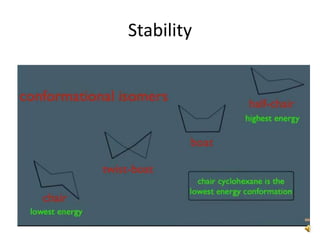
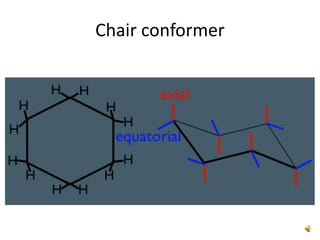
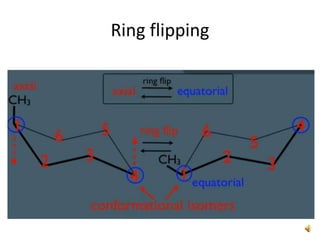
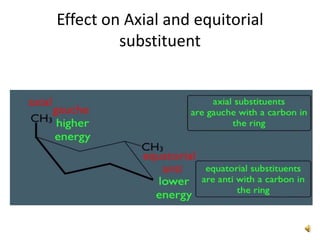
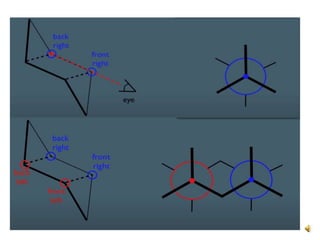
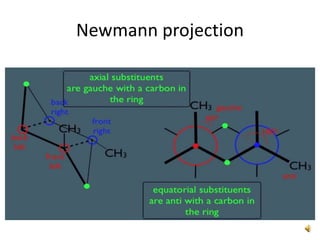
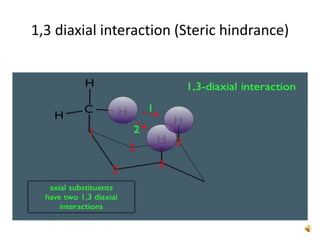
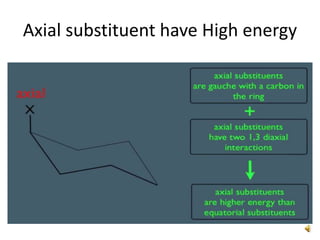





This document discusses the structure and properties of cyclohexane. Cyclohexane adopts a chair conformation with sp3 hybridized carbon atoms forming 120 degree bond angles. The chair structure is stable due to minimizing 1,3-diaxial interactions between axial substituents which experience more steric hindrance than equatorial substituents. Newman projections are used to illustrate axial and equatorial substituents in the chair conformation of cyclohexane.


















