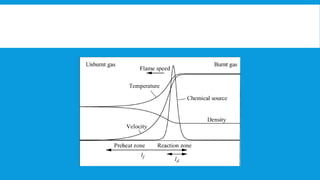
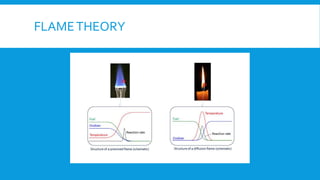
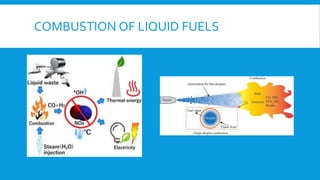
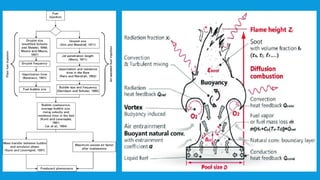
Flames are caused by highly exothermic combustion reactions occurring in a thin reaction zone. There are two main types of flames - luminous flames which emit light from glowing carbon particles, and non-luminous blue flames where combustion is more complete. The structure of a flame consists of distinct zones including unburnt fuel, the reaction zone, and zones of incomplete and complete combustion. Different types of combustion can occur depending on factors like whether the flame is stationary or pulsating, and whether combustion involves explosions.