Embed presentation
Download to read offline




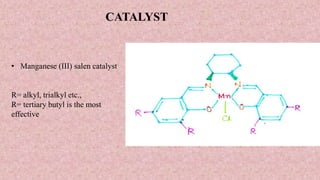
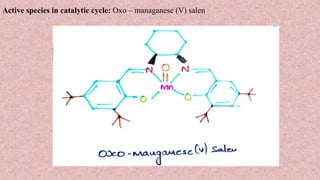
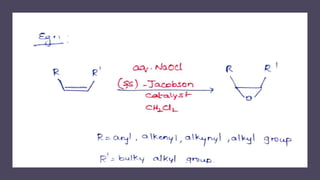
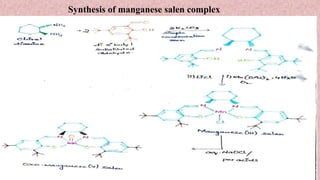
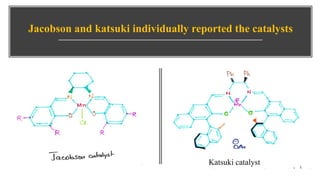
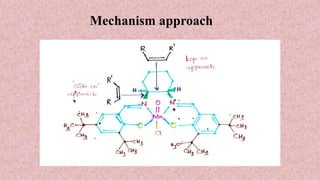



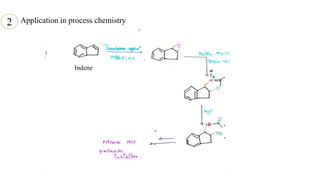


This document presents information about Jacobson asymmetric epoxidation. It discusses the history, catalysts, mechanism, and applications of Jacobson asymmetric epoxidation. The catalyst uses a manganese (III) salen complex where tertiary butyl is the most effective substituent. There are three major pathways for the mechanism: concerted, free radical, and metallo oxetane. Jacobson asymmetric epoxidation is useful for synthesizing pharmaceutical drugs by forming chiral epoxides from cyclic and acyclic alkenes with 100% enantiopurity.















