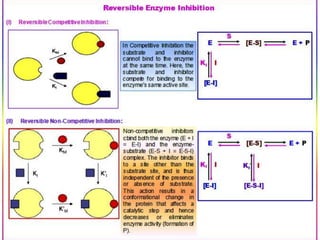
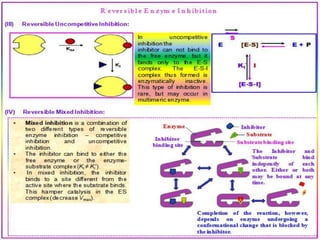
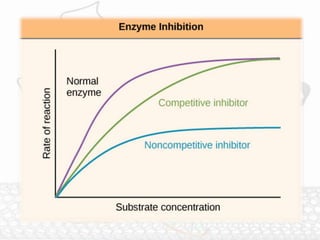
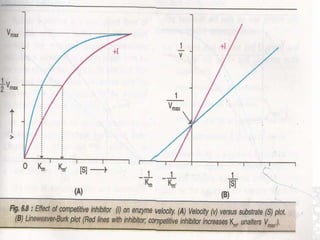
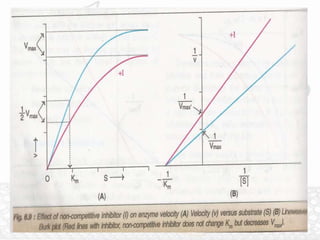
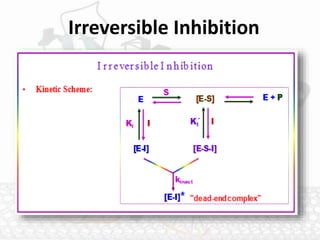
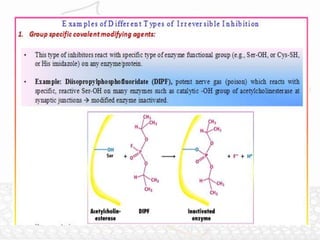
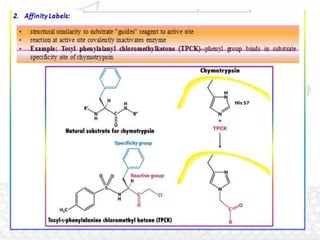
This document discusses enzymes and enzyme kinetics. It defines enzymes as biomolecules that catalyze chemical reactions and provides examples of enzyme units and the SI unit for enzyme activity, the katal. The document then summarizes Michaelis-Menten enzyme kinetics, including the key assumptions of the MM model. It describes parameters of the MM equation like Km, Vmax, kcat, and kcat/Km. Finally, it discusses enzyme inhibition, categorizing inhibitors as reversible or irreversible and describing different types of reversible and irreversible inhibition.




![Kinetics of enzyme catalyzed reaction
Model for enzyme catalyzed reaction is L.
Michaelis & M. Menten in 1913.
Used the enzyme invertase in their study.
Proposed the following scheme of Rxn.
What is MM Equation???
The equation describing the hyperbolic dependence of
the initial velocity, V0 , on the substrate concentration
[S].](https://image.slidesharecdn.com/enzymeandenzzymeinhibition-160315183225/85/Enzyme-and-enzyme-inhibition-6-320.jpg)
![1.The conc. of [S] is much greater than the
conc. Of of the [E].
2.The rate of formation of the ES = rate of
breakdown of ES {steady state assumption
given by the Briggs and Haldane}
3.very little accumulation of P, so the
formation of ES complex form E+P is
negligible.
Assumptions on which MM model is based on:=](https://image.slidesharecdn.com/enzymeandenzzymeinhibition-160315183225/85/Enzyme-and-enzyme-inhibition-7-320.jpg)

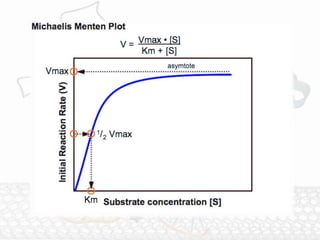


![Significance of Various Parameters in Michaelis-
Menten Equation
1. Significance of Km- Measure of Substrate affinity:
•From Michaelis-Menten equation: If v0 is set equal to 1/2
Vmax, then the relation Vmax /2 = Vmax[S]/Km + [S] can be
simplified to Km + [S] = 2[S], or Km = [S]. This means that at
one half of the maximal velocity, the substrate concentration
at this velocity will be equal to the Km.
•Km represents the substrate concentration at which half of
the enzyme active sites are filled with the substrate.
•The significance of Km change depends on the different rate
constants and which step is the slowest rate-limiting step. In
the simplest assumption, the rate of ES breakdown to product
(k2) is the rate-determining step of the reaction, so k-1 >>
k2 and Km = k-1/k1.](https://image.slidesharecdn.com/enzymeandenzzymeinhibition-160315183225/85/Enzyme-and-enzyme-inhibition-12-320.jpg)