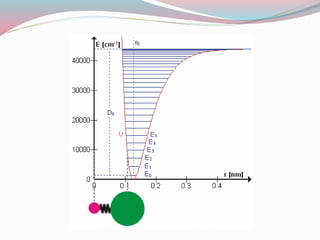
This document discusses diatomic molecules and their behavior as simple anharmonic oscillators. It begins by defining diatomic molecules as molecules composed of two atoms that can be the same or different elements. It then describes molecular vibration, noting that diatomic molecules have one normal mode of vibration involving periodic motion of the atoms. The motion is approximated as simple harmonic motion. However, real molecules are anharmonic oscillators as the restoring force is not perfectly proportional to displacement from equilibrium. The document explores vibrational energy levels and selection rules for infrared spectroscopy. It concludes by relating the potential energy curve of an oscillator to the period of oscillations at different energies.