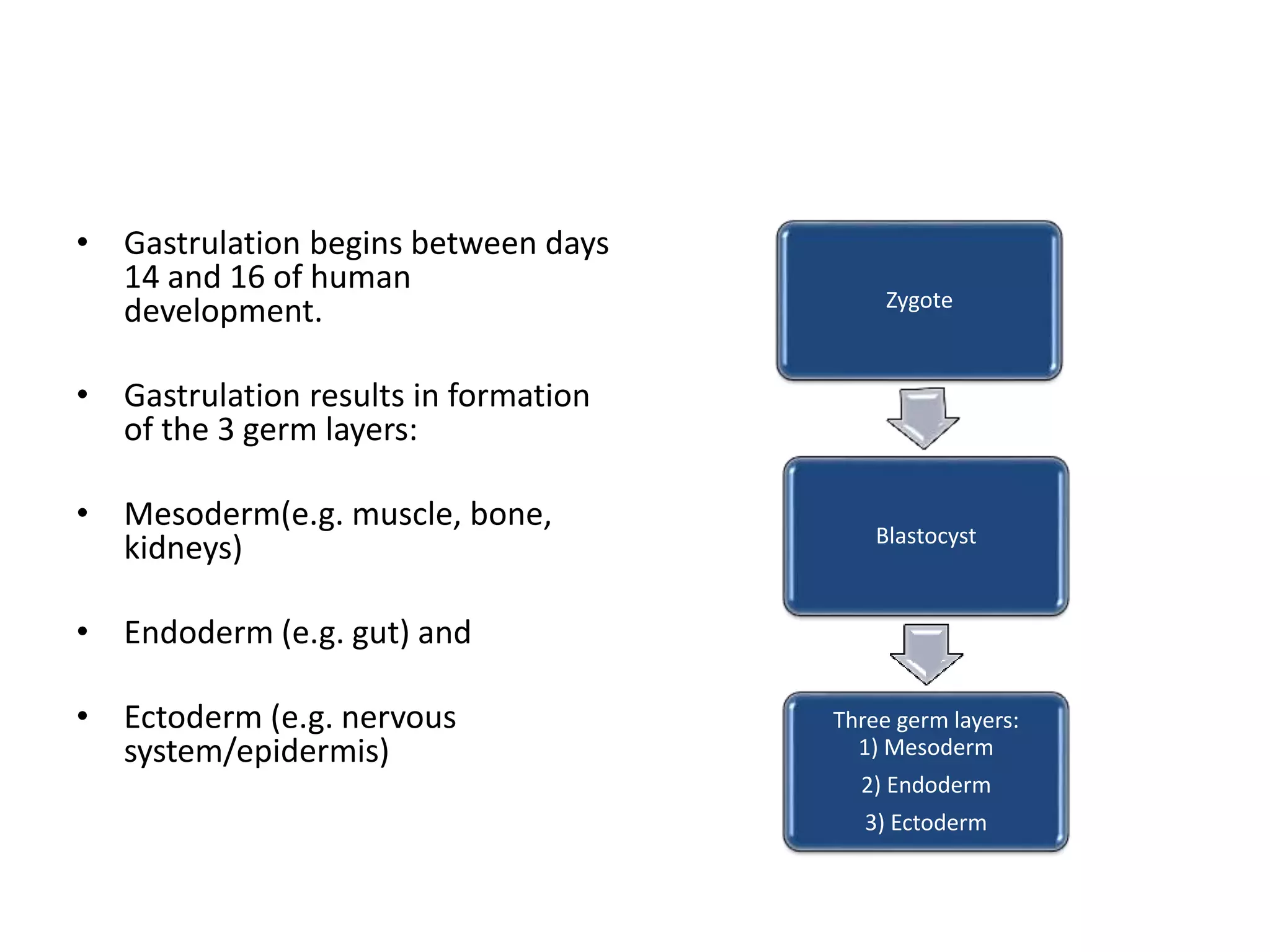
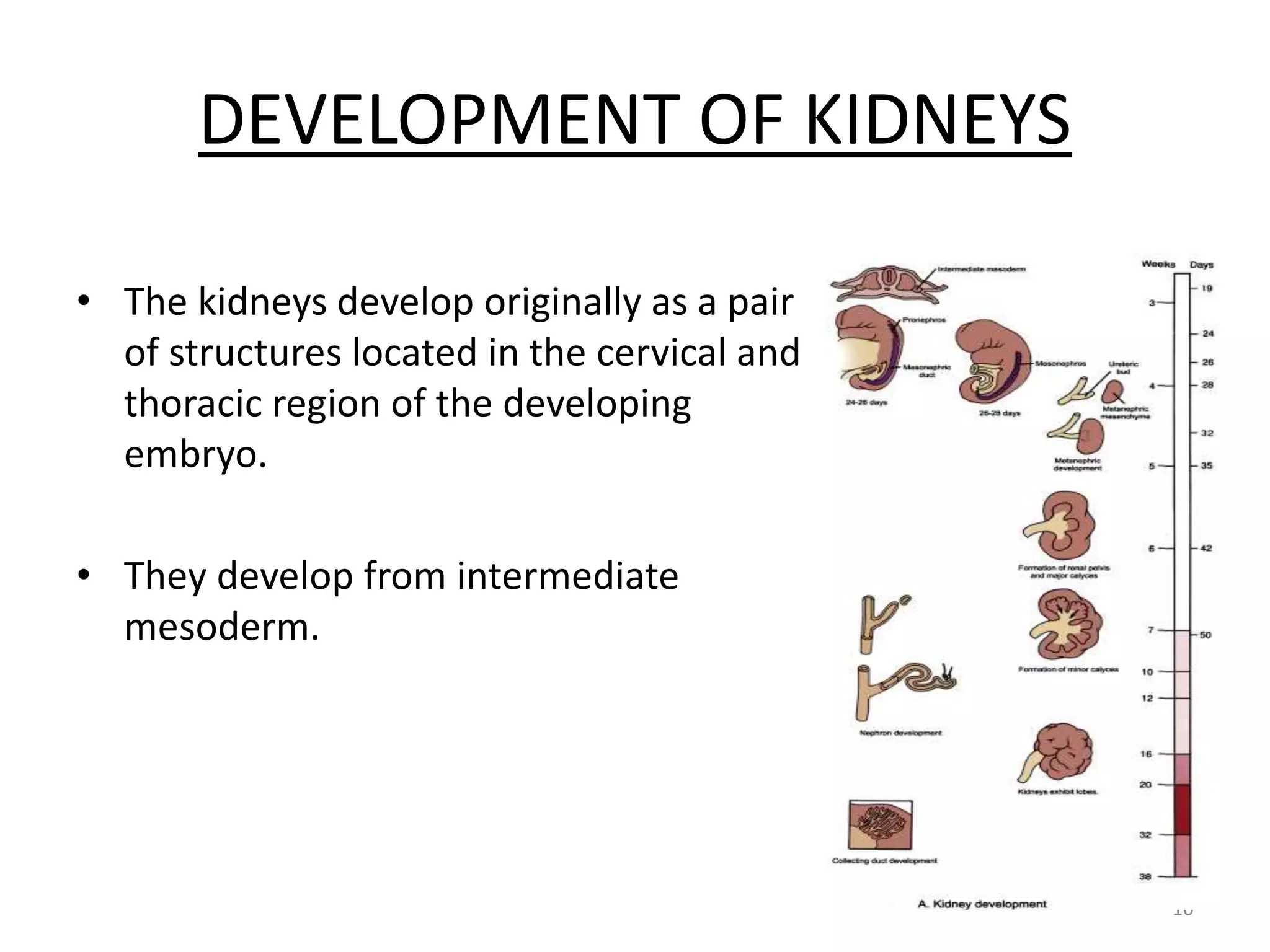
The document discusses kidney development from the formation of the three germ layers during gastrulation to the development of the pronephros, mesonephros, and metanephros. It describes how the intermediate mesoderm forms the nephrogenic cord which develops into the metanephros. The ureteric bud branches to form the collecting system and induces nephron formation from the surrounding metanephric mesenchyme. Congenital anomalies of kidney development including anomalies of number, ascent, form and fusion, rotation, and the collecting system and vasculature are also summarized.