This document provides an overview of dentin, including:
- A brief history of discoveries related to dentin structure.
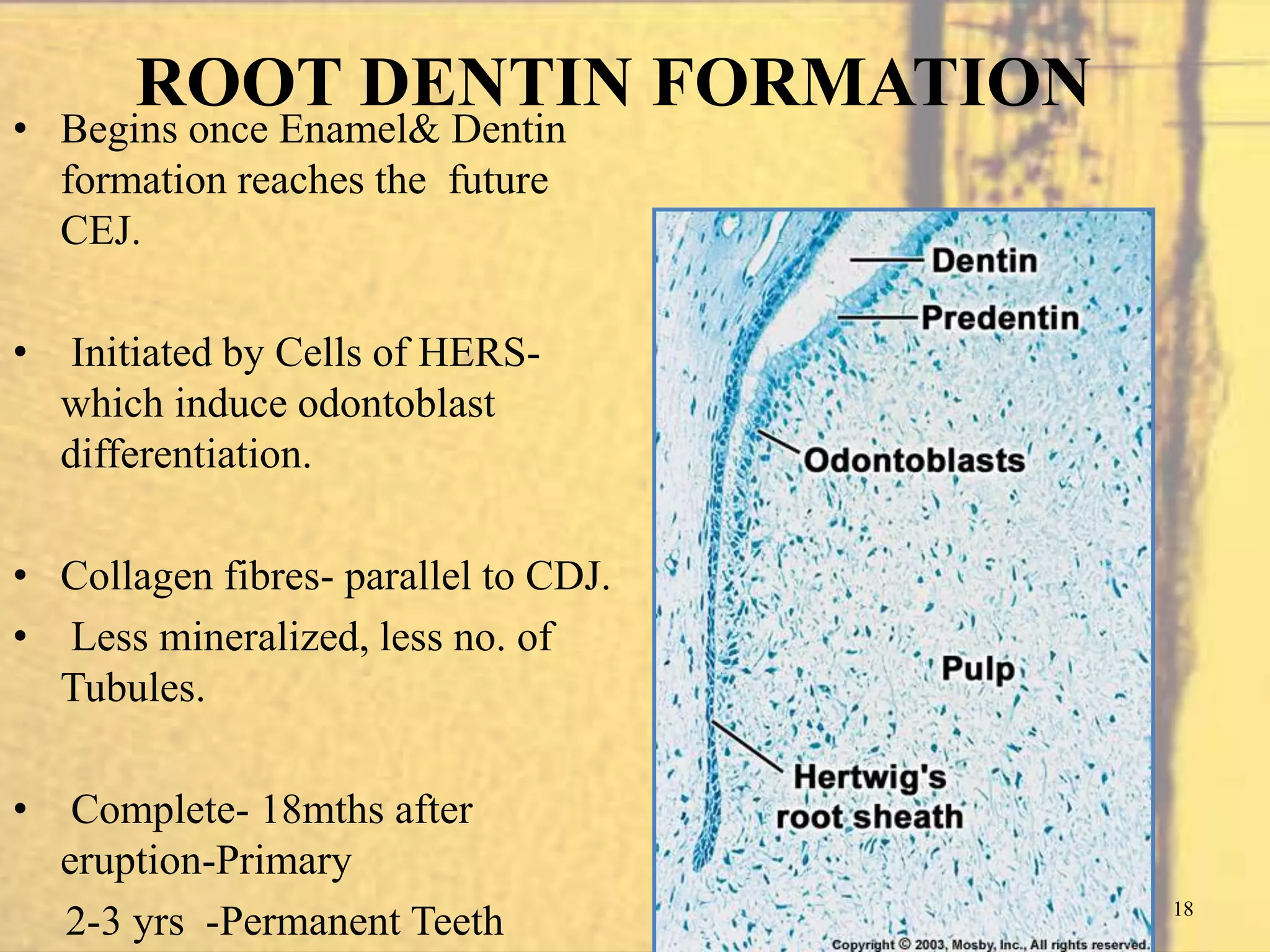
- Dentinogenesis, the process of dentin formation carried out by odontoblasts. Primary dentin formation beneath the enamel and root dentin formation are described.
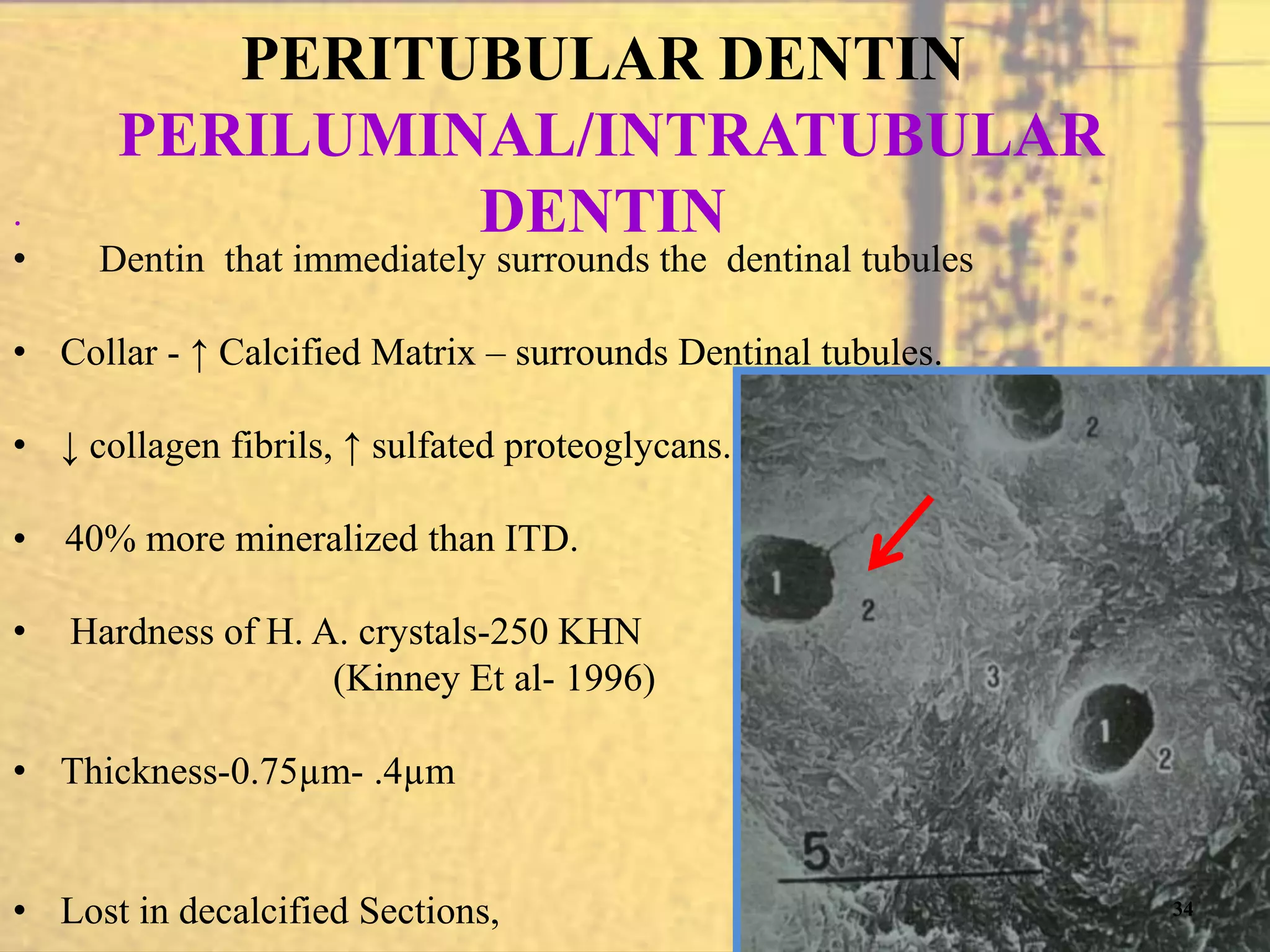
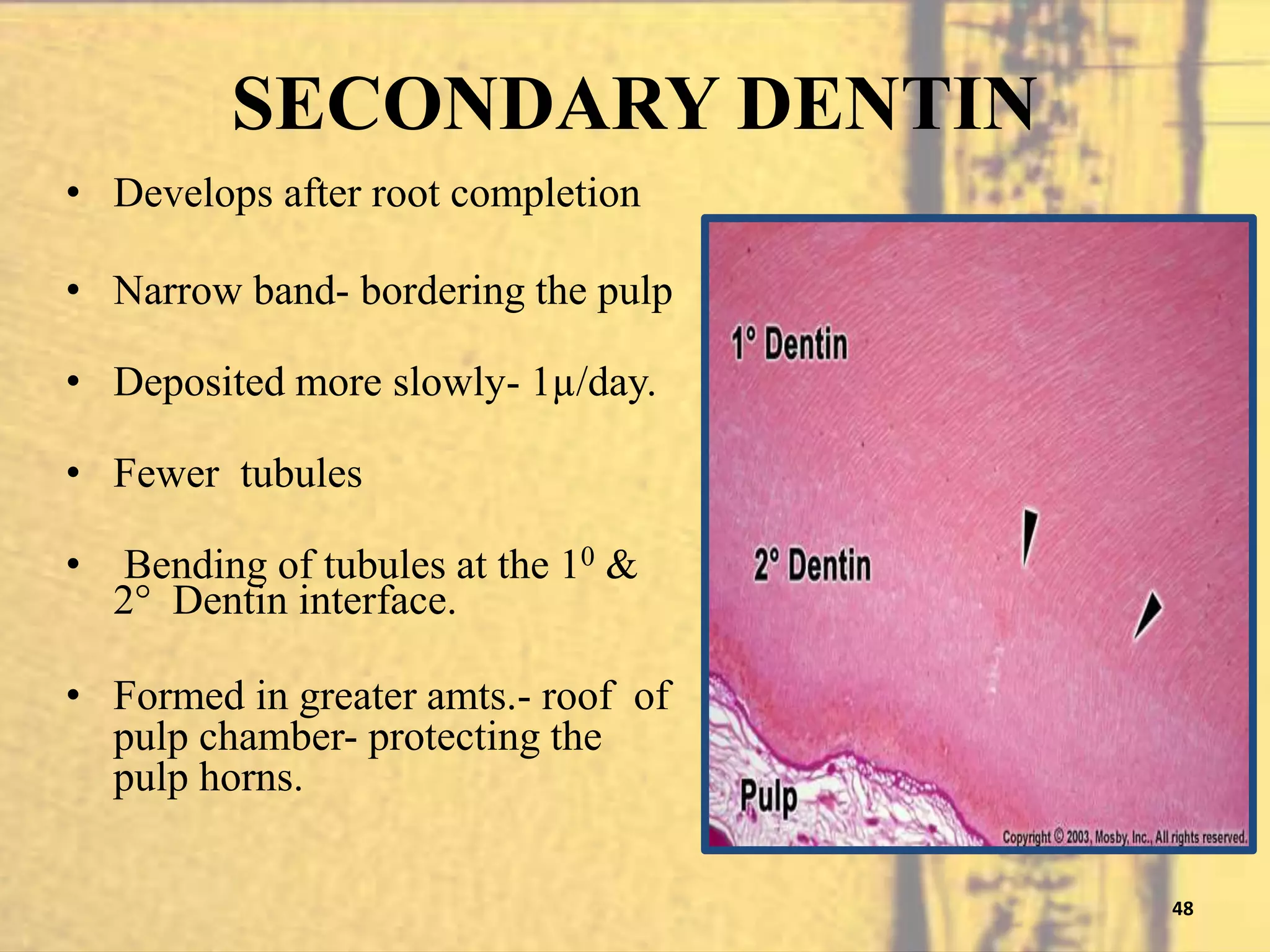
- The physical properties, chemical composition, and structural components of dentin including dentinal tubules, predentin, peritubular and intertubular dentin.
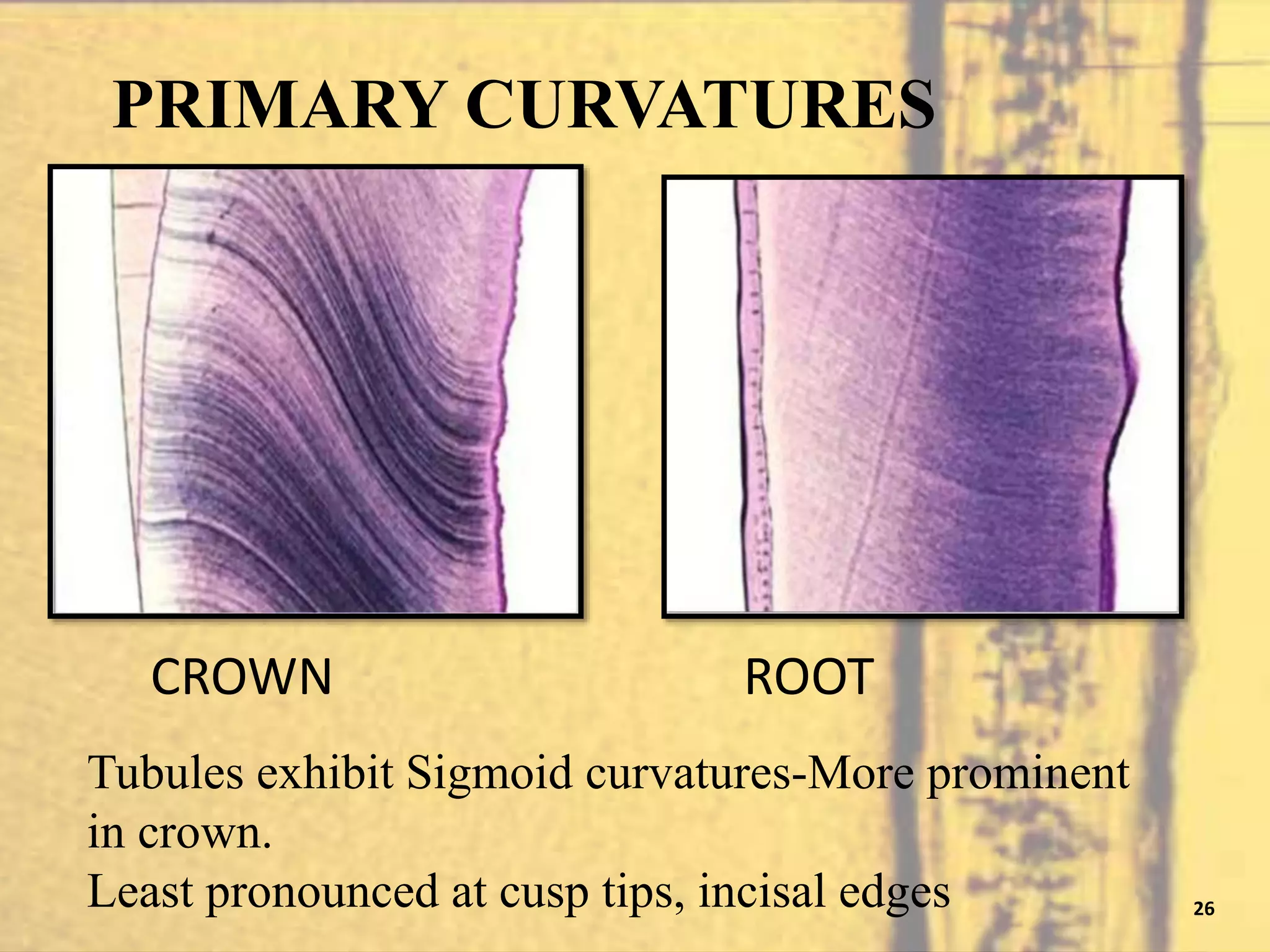
- Features such as von Ebner's lines, lines of Schreger, and contour lines of Owen which represent incremental growth patterns in dentin.























































































![RESPONSE OF DENTIN TO RESTORATIVE
PROCEDURE AND MATERIALS
1) SMEAR LAYER:
The smear layer is an amorphous , relatively smooth layer of
microcrystalline debris with a featureless surface that cannot
be seen with the naked eye [ Pashley DH 1984]
The cutting of dentin during cavity preparation produces
microcrystalline grinding debris that coats the dentin and
clogs the orifices of the dentinal tubules. This layer of debris
is termed as smear layer.](https://image.slidesharecdn.com/dentin-141211031221-conversion-gate02/75/Dentin-92-2048.jpg)










