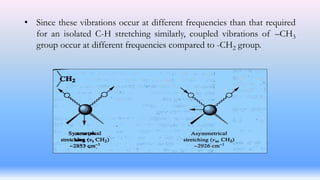
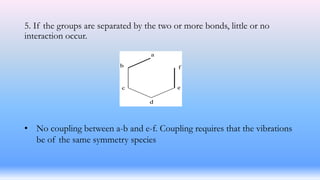
The document discusses coupling vibrations in infrared spectroscopy, highlighting the significance of shared atoms and bonds in achieving effective coupling between vibrating groups. It emphasizes that strong coupling occurs when vibrations of the same symmetry species share a common atom or bond, leading to shifts in absorption frequencies. Additionally, it notes that these variations in absorption position can aid in the identification of organic compounds.