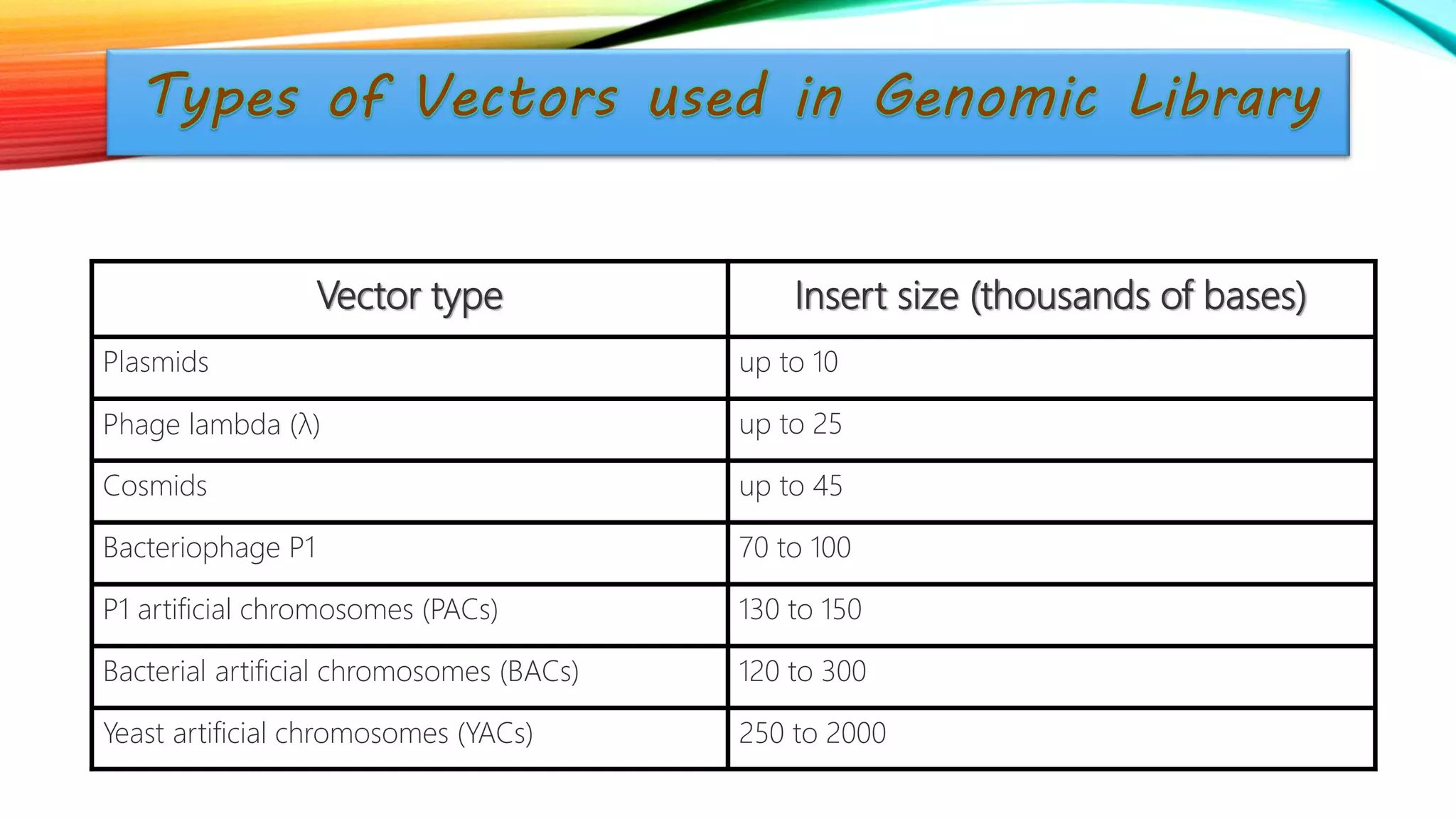
The document summarizes the process of constructing genomic and cDNA libraries. A genomic library contains total genomic DNA from an organism, including exons and introns. Genomic DNA is extracted, fragmented, and inserted into vectors which are then introduced into host cells. Each cell contains a different DNA fragment. A cDNA library contains only expressed genes as cDNA, excluding introns. mRNA is extracted and reverse transcribed to cDNA, which is then inserted into vectors. Both library types are useful tools for sequencing and studying gene expression.