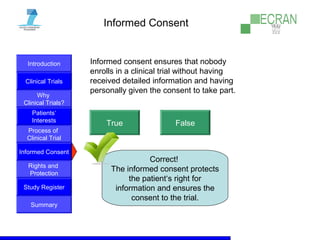
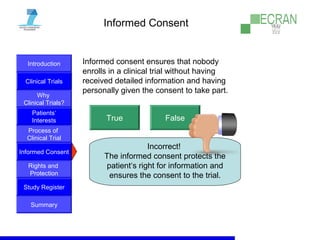
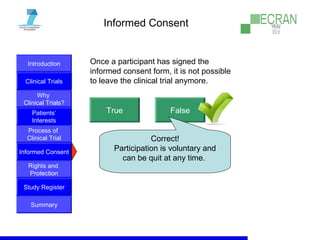
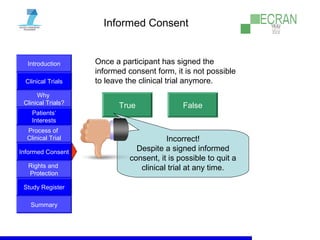
Clinical trials are essential for testing new medical treatments and ensuring their safety and efficacy. They involve dividing patients into groups that receive either an experimental treatment or the standard treatment in a controlled manner. The clinical trial process is carefully designed and regulated to obtain reliable results while protecting patients' rights and well-being. Large numbers of patients are needed to statistically prove whether a new treatment is better or worse than existing options. While new therapies may help future patients, there are no guarantees of success or improvement, so participation in clinical trials always involves some unknown risks.