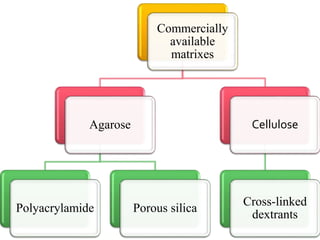
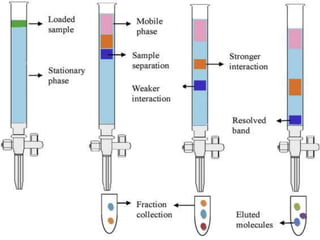
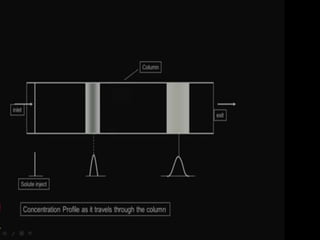
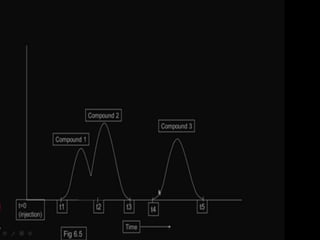
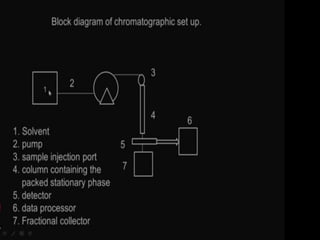
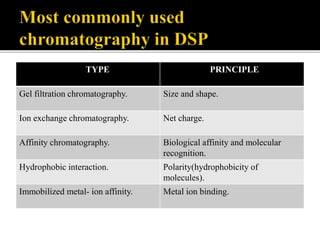
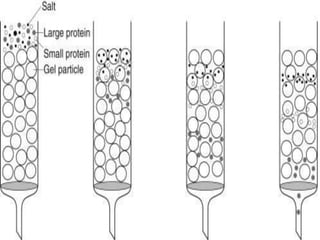
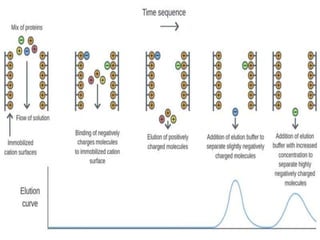
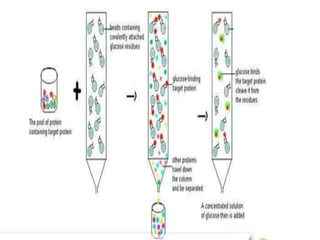
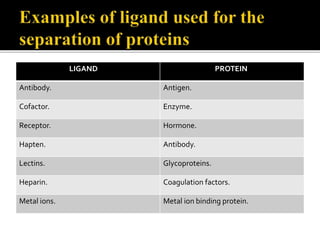
This document discusses downstream processing and chromatography techniques used in downstream processing. It begins by stating that downstream processing, which can account for up to 60% of production costs, is needed to separate and purify desired products after fermentation or enzyme reactions. Chromatography is commonly used for purification and separation in downstream processing. The document then describes the main types of chromatography used - gel filtration chromatography, ion exchange chromatography, and affinity chromatography - and explains the principles behind each technique. It provides examples of their industrial applications and summarizes the key objectives of learning about chromatography in downstream processing.