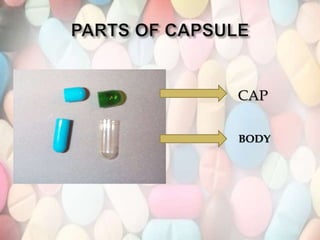
Capsules have several advantages over tablets like masking unpleasant tastes, being easy to swallow, and requiring fewer excipients. They consist of a shell made of gelatin enclosing the drug formulation. Gelatin capsules come in various sizes and are made through a process involving dipping, spinning, drying, stripping, trimming, and polishing. Stability testing ensures the integrity of the capsule shell and determines shelf life, while uniformity testing confirms consistent drug content between capsules. Dissolution and disintegration tests evaluate how quickly the drug is released from the capsule.