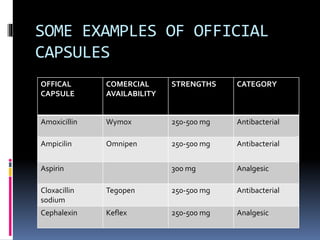
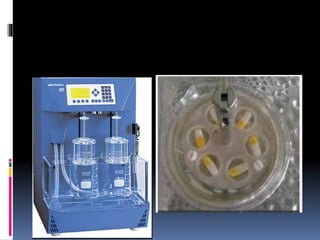
This document discusses quality control testing for capsules. It describes physical tests like disintegration testing, weight variation testing, and chemical tests like dissolution testing and content uniformity testing. Disintegration testing ensures capsules break down within a specified time in liquid. Weight variation testing checks that capsule weights are consistent. Dissolution testing measures how quickly the active drug is released. Content uniformity testing confirms the amount of drug in each capsule is consistent. These quality control tests are important to ensure capsules meet specifications and perform as intended.