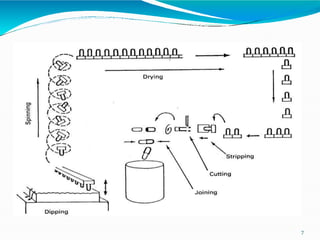
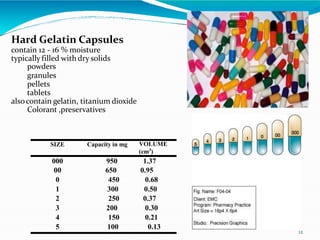
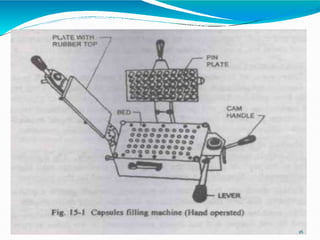
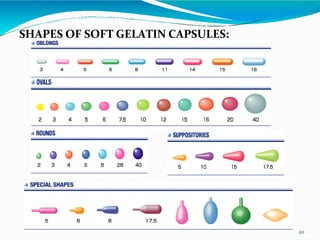
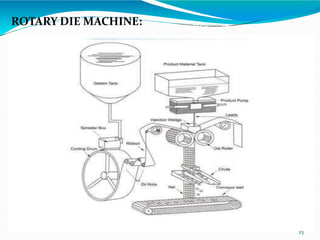
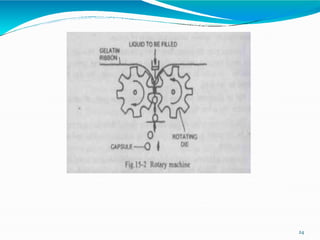
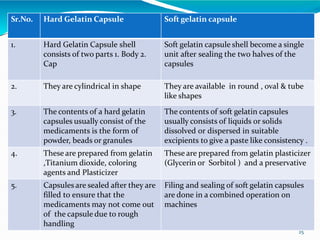
Capsules are solid dosage forms where the drug is enclosed within a shell, typically made of gelatin. There are two main types - hard gelatin capsules which contain powders, granules, or pellets and release their contents rapidly; and soft gelatin capsules which contain liquids or pastes and provide rapid release. Capsules offer advantages like masking unpleasant tastes and smells, easy swallowing, and sustained or delayed release depending on the formulation. They are manufactured through processes like dipping, spinning, drying, stripping, trimming, and joining. Finished capsules are evaluated for content uniformity, disintegration time, moisture content, and dissolution.