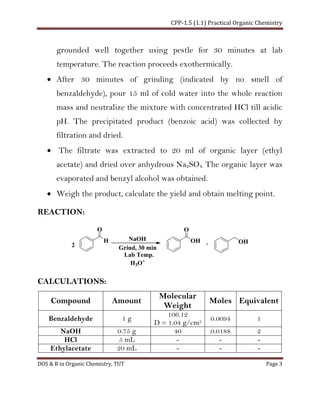
This document describes a laboratory experiment on Cannizzaro's reaction of benzaldehyde. Benzaldehyde undergoes a solvent-free disproportionation reaction in the presence of sodium hydroxide to produce benzoic acid and benzyl alcohol. The reaction is carried out by grinding benzaldehyde and sodium hydroxide together for 30 minutes. Benzoic acid precipitates out and is collected by filtration. Benzyl alcohol is extracted from the filtrate using ethyl acetate. The yields of benzoic acid and benzyl alcohol are calculated and their melting points determined and compared to theoretical values.